Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Pharmaceutical Intermediates
Scalable Iron-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Advanced Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing nitrogen-containing heterocycles, particularly quinazolinones, due to their pervasive presence in bioactive molecules. As illustrated in the structural diversity of known drugs, quinazolinone scaffolds serve as the core architecture for compounds exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. The introduction of a trifluoromethyl group into these heterocyclic systems further enhances their pharmacokinetic profiles by improving metabolic stability and lipophilicity. Patent CN111675662B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted quinazolinone compounds, utilizing a cost-effective iron-catalyzed cyclization strategy. This technological advancement addresses critical bottlenecks in the manufacturing of high-value pharmaceutical intermediates, offering a pathway that balances synthetic efficiency with economic viability.
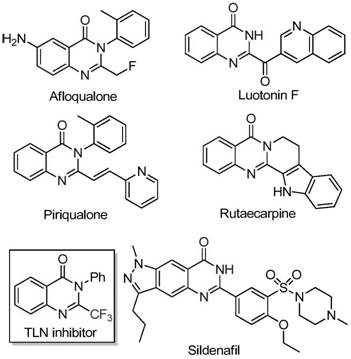
Quinazolinone derivatives represent a privileged scaffold in medicinal chemistry, underpinning the efficacy of numerous approved drugs and clinical candidates. The biological significance of this core structure is evident in its ability to interact with various enzymatic targets, making it a focal point for drug discovery programs targeting oncology and central nervous system disorders. The strategic incorporation of fluorine atoms, specifically the trifluoromethyl moiety, has become a standard tactic in modern drug design to modulate electronic properties and block metabolic hotspots. However, traditional synthetic routes to access these fluorinated architectures often rely on expensive reagents or harsh reaction conditions that limit their utility in large-scale production. The disclosed invention provides a timely solution by leveraging abundant iron catalysts to drive the formation of these complex heterocycles under relatively mild conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trifluoromethyl-substituted quinazolinones has been plagued by significant operational and economic challenges that hinder widespread adoption in industrial settings. Conventional methodologies typically depend on the cyclization of trifluoromethyl-containing synthons, such as trifluoroacetic anhydride or ethyl trifluoroacetate, with substrates like anthranilamide or isatoic anhydride. These classical approaches frequently suffer from severe reaction conditions, requiring extreme temperatures or pressures that pose safety risks and increase energy consumption. Furthermore, the substrate scope is often narrow, limiting the structural diversity accessible to medicinal chemists who need to explore structure-activity relationships extensively. The reliance on specialized and costly starting materials drives up the overall cost of goods, making these routes less attractive for the production of generic pharmaceutical intermediates where margin compression is a constant pressure.
The Novel Approach
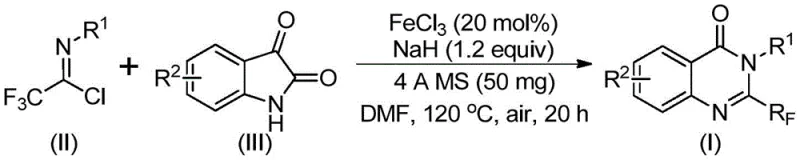
In stark contrast to legacy methods, the novel approach detailed in the patent utilizes readily available trifluoroethylimidoyl chloride and isatin as primary building blocks, catalyzed by inexpensive ferric chloride. This strategy represents a paradigm shift towards sustainable and economical chemical manufacturing by replacing precious metal catalysts with earth-abundant iron. The reaction proceeds through a tandem sequence involving alkali-promoted bond formation followed by iron-catalyzed decarbonylation and cyclization, effectively streamlining the synthetic pathway. By operating in common organic solvents like DMF and tolerating air exposure, the process simplifies the operational requirements, eliminating the need for rigorous inert atmosphere techniques that add complexity and cost to reactor operations. This accessibility makes the technology particularly appealing for contract development and manufacturing organizations aiming to optimize their production workflows.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the dual role of the reagents, where sodium hydride acts as a base to facilitate the initial nucleophilic attack, while ferric chloride orchestrates the subsequent cyclization and decarbonylation steps. The reaction initiates with the formation of a carbon-nitrogen bond between the trifluoroethylimidoyl chloride and the isatin substrate, generating a trifluoroacetamidine intermediate. This intermediate then undergoes an iron-mediated rearrangement where the carbonyl group is extruded, driving the thermodynamic equilibrium towards the formation of the stable quinazolinone ring system. The use of 4A molecular sieves plays a crucial role in sequestering moisture and potentially trapping byproducts, thereby pushing the reaction forward and ensuring high conversion rates. This mechanistic pathway avoids the generation of toxic waste streams associated with traditional coupling agents, aligning with green chemistry principles.
From an impurity control perspective, the selectivity of the iron catalyst is paramount in ensuring the purity of the final API intermediate. The specific coordination environment provided by the ferric species favors the desired intramolecular cyclization over potential intermolecular side reactions that could lead to oligomerization or polymerization. The tolerance of various substituents on the aromatic rings, including electron-withdrawing halogens and electron-donating alkyl groups, suggests a robust catalytic cycle that is not easily poisoned by steric or electronic variations. This broad substrate compatibility is critical for process chemists who must validate the synthesis across a library of analogues during the drug development phase. The ability to achieve high yields with minimal byproduct formation simplifies the downstream purification process, reducing the burden on chromatographic separation and crystallization steps.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis involves a straightforward protocol that can be adapted for both laboratory discovery and pilot plant operations. The procedure begins with the precise weighing of ferric chloride and sodium hydride, which are added to a reaction vessel along with 4A molecular sieves to maintain anhydrous conditions. Trifluoroethylimidoyl chloride and isatin derivatives are then introduced into the organic solvent, typically DMF, and the mixture is stirred to ensure homogeneity. The reaction profile involves a two-stage temperature program, starting at a moderate 40°C to allow for initial adduct formation, followed by heating to 120°C to drive the cyclization to completion. Detailed standardized synthesis steps for this specific transformation are provided in the guide below.
- Mix ferric chloride, sodium hydride, 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin in an organic solvent like DMF.
- React the mixture at 40°C for 8-10 hours, then heat to 120°C for 18-20 hours under air atmosphere.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to obtain the final quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this iron-catalyzed methodology offers tangible benefits that directly impact the bottom line and operational resilience. The shift away from proprietary or scarce reagents towards commodity chemicals like isatin and ferric chloride mitigates the risk of supply disruptions caused by geopolitical factors or single-source dependencies. The simplified workup procedure, which relies on standard filtration and chromatography rather than complex extraction or distillation sequences, reduces the turnaround time for batch processing. This efficiency gain allows manufacturing facilities to increase their throughput without requiring significant capital investment in new equipment, thereby optimizing asset utilization rates across the production floor.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with ferric chloride results in a drastic reduction in raw material costs, as iron salts are among the most affordable catalysts available in the chemical market. Additionally, the elimination of stringent inert atmosphere requirements lowers the operational expenditure related to nitrogen or argon consumption and specialized glovebox maintenance. The high atom economy of the reaction minimizes waste generation, which in turn reduces the costs associated with waste disposal and environmental compliance monitoring. These cumulative savings contribute to a more competitive cost structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The starting materials, including various substituted isatins and aromatic amines required for the imidoyl chloride precursor, are widely commercially available from multiple global suppliers. This multi-sourcing capability ensures that production schedules are not held hostage by the inventory levels of a single vendor, providing a buffer against market volatility. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in reagent quality, further stabilizing the supply chain. Consequently, manufacturers can offer more reliable delivery commitments to their downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The use of DMF as a solvent, while requiring careful handling, is well-understood in industrial settings with established recovery and recycling protocols that minimize environmental impact. The absence of heavy metal residues in the final product simplifies the regulatory filing process, as extensive data on metal clearance is not required compared to palladium or rhodium-catalyzed routes. The process has been demonstrated to be scalable, moving seamlessly from milligram scale in research to gram and potentially kilogram scale in production, ensuring that the technology can support the growing demand for these bioactive scaffolds as drugs progress through clinical trials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity for technical teams evaluating the process. Understanding these nuances is essential for making informed decisions about process adoption and integration into existing manufacturing portfolios.
Q: What are the advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: Ferric chloride is significantly cheaper and more earth-abundant than precious metal catalysts, reducing raw material costs and simplifying heavy metal removal processes in pharmaceutical manufacturing.
Q: Can this synthesis method tolerate diverse functional groups on the substrate?
A: Yes, the method demonstrates excellent functional group tolerance, accommodating substituents such as halogens, alkyl groups, and methoxy groups on both the isatin and imidoyl chloride components.
Q: Is this process suitable for large-scale industrial production?
A: The process uses readily available starting materials and simple post-treatment procedures like filtration and chromatography, making it highly scalable from gram to kilogram levels for commercial application.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the fast-paced pharmaceutical landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to bulk manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl quinazolinone meets the highest standards required for API synthesis. Our commitment to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking reliable sources of complex heterocyclic intermediates.
We invite you to leverage our technical expertise to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your project goals. Let us collaborate to bring your next-generation therapeutics to market faster and more efficiently.
