Scalable Terminal Alkyne Synthesis via Inorganic Base Elimination for Global Pharma Supply Chains
Scalable Terminal Alkyne Synthesis via Inorganic Base Elimination for Global Pharma Supply Chains
The landscape of organic synthesis for critical pharmaceutical intermediates is undergoing a significant transformation, driven by the urgent need for safer, more economical, and environmentally sustainable manufacturing processes. Patent CN101967075A introduces a groundbreaking methodology for the synthesis of terminal alkyne compounds utilizing 3-aryl-2,3-dibromopropionic acid as the primary starting material. This innovation represents a paradigm shift away from traditional, hazard-prone organometallic routes towards a robust protocol employing readily available inorganic bases. For R&D directors and procurement strategists in the global fine chemical sector, this technology offers a compelling value proposition by drastically simplifying reaction conditions while maintaining high purity standards essential for downstream drug development. The ability to generate high-value terminal alkynes without the need for cryogenic temperatures or exotic reagents positions this method as a cornerstone for next-generation supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of terminal alkynes has been dominated by the Corey-Fuchs reaction and its variants, which rely heavily on the use of extremely strong bases such as n-butyllithium (nBuLi) operating at cryogenic temperatures of -78°C. These conventional methodologies impose severe logistical and financial burdens on manufacturing facilities, necessitating specialized low-temperature equipment and rigorous safety protocols to handle pyrophoric reagents. Furthermore, alternative approaches like the Ohira-Bestmann reaction, while popular in academic settings, often involve the generation of phosphine oxide byproducts that are environmentally unfriendly and economically inefficient to dispose of on a commercial scale. The reliance on such苛刻 (harsh) conditions limits substrate scope and increases the risk of side reactions, ultimately compromising the overall yield and purity profile required for GMP-compliant API intermediate production.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages a double elimination and decarboxylation strategy using inexpensive inorganic bases such as cesium carbonate (Cs2CO3), potassium carbonate (K2CO3), or cesium fluoride (CsF). By reacting 3-aryl-2,3-dibromopropionic acid in polar aprotic solvents like dimethyl sulfoxide (DMSO) or N,N-dimethylformamide (DMF) at temperatures merely exceeding 25°C, the process achieves efficient conversion to the desired terminal alkyne. This methodology eliminates the need for energy-intensive cooling systems and allows for the use of standard stainless steel reactors, thereby significantly lowering the barrier to entry for commercial scale-up. The versatility of this system is evidenced by its successful application across a diverse range of substrates, including electron-deficient and electron-rich aryl rings, demonstrating a level of robustness that is rarely seen in traditional alkyne synthesis.
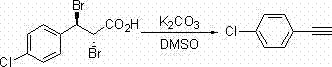
Mechanistically, this transformation proceeds through a concerted sequence of dehydrobromination events facilitated by the inorganic base. The initial step likely involves the abstraction of an acidic proton adjacent to the bromine atoms, leading to the formation of a vinyl bromide intermediate. Subsequent elimination of the second bromine atom, coupled with the loss of carbon dioxide (decarboxylation), drives the equilibrium towards the formation of the thermodynamically stable triple bond. This pathway is particularly advantageous because it avoids the generation of highly reactive carbanionic species that are prone to nucleophilic attack on other functional groups, thus ensuring a cleaner impurity profile. For quality control teams, this means a simplified purification workflow, often requiring only standard extraction and column chromatography to achieve analytical grade purity, which is critical for reducing lead times in process development.

Mechanistic Insights into Inorganic Base Mediated Double Elimination
The catalytic efficiency of this system relies heavily on the choice of base and solvent polarity to stabilize the transition states involved in the elimination steps. Solvents like DMSO and DMF are not merely inert media; their high dielectric constants facilitate the dissociation of the inorganic salts, increasing the availability of the basic anions required for proton abstraction. The patent data indicates that the molar ratio of the dibromopropionic acid to the base can vary widely from 0.1:1 to 1:1, suggesting a flexible stoichiometric window that allows process chemists to optimize reagent consumption based on cost constraints. This flexibility is a key differentiator, as it permits the use of cheaper bases like potassium carbonate for less demanding substrates while reserving more potent bases like cesium carbonate for sterically hindered or electronically deactivated systems, ensuring consistent performance across a broad portfolio of target molecules.
From an impurity control perspective, the absence of organolithium reagents virtually eliminates the risk of metal-halogen exchange side reactions, which are common pitfalls in halogenated substrate processing. In traditional methods, the presence of halogens on the aromatic ring often leads to unwanted coupling or decomposition when exposed to nBuLi. However, the mild basicity of carbonates and fluorides ensures that sensitive functionalities, such as the chloro-group in p-chlorophenylacetylene or the nitro-group in 4-nitrophenylacetylene, remain intact throughout the reaction. This chemoselectivity is paramount for the synthesis of complex pharmaceutical intermediates where orthogonal protection strategies add unnecessary steps and cost. By preserving the integrity of the molecular scaffold, this method streamlines the synthetic route, directly contributing to a reduced cost of goods sold (COGS) and a faster time-to-market for new drug candidates.
How to Synthesize Terminal Alkynes Efficiently
To implement this synthesis effectively, process engineers must adhere to specific operational parameters regarding temperature control and workup procedures to maximize yield and safety. The general protocol involves charging the reactor with the 3-aryl-2,3-dibromopropionic acid precursor, the selected inorganic base, and the appropriate polar solvent, followed by heating the mixture to a range between 95°C and 140°C depending on the substrate reactivity. Detailed standardized operating procedures regarding exact stoichiometry, agitation rates, and quenching protocols are essential for reproducibility and are outlined in the technical documentation below for immediate adoption by manufacturing teams.
- Charge the reactor with 3-aryl-2,3-dibromopropionic acid, an inorganic base such as cesium carbonate or potassium carbonate, and a polar solvent like DMSO.
- Stir the reaction mixture at temperatures greater than 25°C, typically between 95°C and 140°C, for a duration of 1 to 24 hours to ensure complete conversion.
- Quench the reaction with water, extract the organic layer with ether, wash with brine, dry over sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this inorganic base-mediated synthesis route offers transformative economic benefits that extend far beyond simple reagent substitution. By shifting away from cryogenic organometallic chemistry, manufacturers can realize substantial cost reductions through the elimination of expensive specialty gases, low-temperature infrastructure, and hazardous waste disposal fees associated with lithium residues. The use of commodity chemicals like potassium carbonate and cesium carbonate, which are available in bulk quantities from multiple global suppliers, mitigates the risk of supply chain disruptions and price volatility often seen with niche organometallic reagents. This stability in raw material sourcing ensures continuous production schedules and protects margins against market fluctuations, making it an ideal strategy for long-term contract manufacturing agreements.
- Cost Reduction in Manufacturing: The economic impact of replacing nBuLi and phosphine reagents with inorganic salts cannot be overstated, as it removes the need for specialized handling equipment and reduces the overall energy consumption of the plant. Without the requirement for maintaining -78°C environments, facilities can operate with standard heating jackets, leading to drastic simplifications in utility usage and a significant decrease in the carbon footprint of the manufacturing process. Furthermore, the high yields reported in the patent examples suggest that raw material utilization is highly efficient, minimizing waste generation and maximizing the output per batch, which directly translates to improved profitability for large-scale production runs.
- Enhanced Supply Chain Reliability: Sourcing 3-aryl-2,3-dibromopropionic acid and inorganic bases is logistically straightforward compared to the complex supply chains required for air-sensitive organolithiums. This accessibility allows for the establishment of robust inventory buffers and reduces the lead time for raw material acquisition, ensuring that production lines remain operational even during periods of global logistics stress. The stability of the reagents also simplifies storage requirements, as they do not demand inert atmosphere containment or refrigerated warehousing, further lowering overhead costs and enhancing the overall agility of the supply network to respond to sudden spikes in demand from pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental profile of this method aligns perfectly with modern green chemistry initiatives, as it avoids the generation of toxic phosphine oxides and lithium salts that require complex remediation. The aqueous workup described in the patent allows for easy separation of inorganic salts, which can often be recycled or disposed of with minimal environmental impact, facilitating compliance with increasingly stringent international environmental regulations. This ease of waste management, combined with the inherent safety of operating at atmospheric pressure and moderate temperatures, makes the process highly scalable from pilot plant to multi-ton commercial production without the need for extensive re-engineering of safety systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this terminal alkyne synthesis technology, providing clarity for stakeholders evaluating its integration into their existing portfolios. These insights are derived directly from the experimental data and claims within the patent, ensuring that all information provided is grounded in verified scientific evidence rather than theoretical speculation. Understanding these nuances is critical for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this method over the Corey-Fuchs reaction?
A: Unlike the Corey-Fuchs reaction which requires cryogenic conditions (-78°C) and hazardous organolithium reagents like nBuLi, this patented method utilizes inexpensive inorganic bases at moderate temperatures, significantly reducing operational complexity and safety risks.
Q: Can this synthesis method tolerate sensitive functional groups?
A: Yes, the mild basic conditions and absence of超强碱 (superbases) allow for better tolerance of various functional groups on the aryl ring, including halogens and nitro groups, as demonstrated by the successful synthesis of p-chlorophenylacetylene and 4-nitrophenylacetylene.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Absolutely. The use of common solvents like DMSO and DMF, combined with the elimination of cryogenic cooling requirements and expensive catalysts, makes this route highly amenable to kilogram-to-ton scale production with simplified waste treatment protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Terminal Alkyne Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of reliable access to high-quality terminal alkyne intermediates for the development of next-generation therapeutics and functional materials. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of project phase. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this inorganic base methodology, backed by rigorous QC labs that enforce stringent purity specifications to meet the exacting standards of the global pharmaceutical industry.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this innovative synthesis route for their specific projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time-to-market with our proven expertise in complex intermediate manufacturing.
