Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Palladium-Catalyzed Carbonylation for Commercial Production
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones via Palladium-Catalyzed Carbonylation for Commercial Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. A significant breakthrough in this domain is detailed in patent CN112480015B, which discloses a highly efficient multi-component one-pot method for synthesizing 2-trifluoromethyl substituted quinazolinones. This class of compounds is critically important due to its prevalence in bioactive molecules exhibiting antifungal, antibacterial, antiviral, and anticancer properties. The introduction of the trifluoromethyl group is particularly strategic, as it significantly enhances the metabolic stability, lipophilicity, and bioavailability of the parent molecule, making these intermediates highly desirable for drug discovery programs. By leveraging a palladium-catalyzed carbonylation cascade, this technology offers a streamlined route that bypasses many of the logistical and safety hurdles associated with traditional heterocycle synthesis.
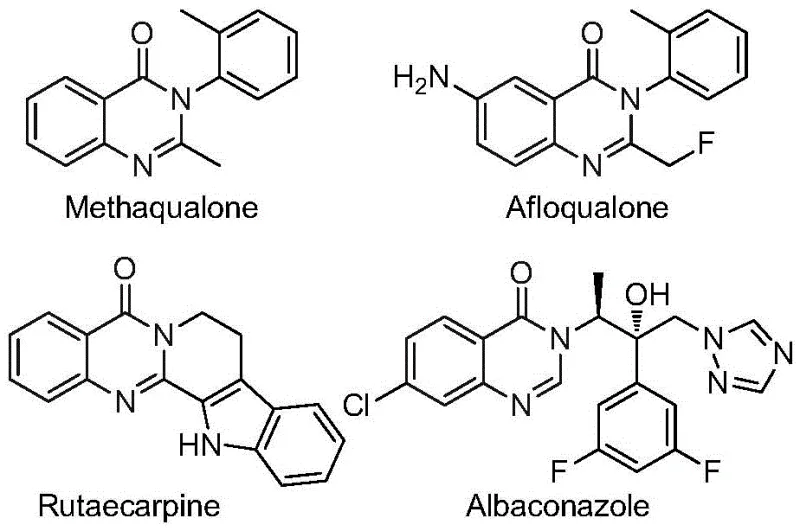
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has been fraught with synthetic challenges that impede efficient commercial manufacturing. Conventional strategies often rely on harsh reaction conditions, such as high-pressure carbon monoxide atmospheres, which require specialized and expensive reactor equipment, thereby increasing capital expenditure and operational risk. Furthermore, many established protocols necessitate the use of precious metal catalysts like ruthenium or platinum, which not only drive up raw material costs but also introduce complexities in downstream purification to meet stringent residual metal specifications for pharmaceutical ingredients. Other methods involve the use of pre-activated substrates, such as acid anhydrides or bromo-formylanilines, which add extra synthetic steps, reduce overall atom economy, and generate additional waste streams. These factors collectively result in lower yields, narrow substrate scope, and a supply chain that is vulnerable to bottlenecks and cost volatility.
The Novel Approach
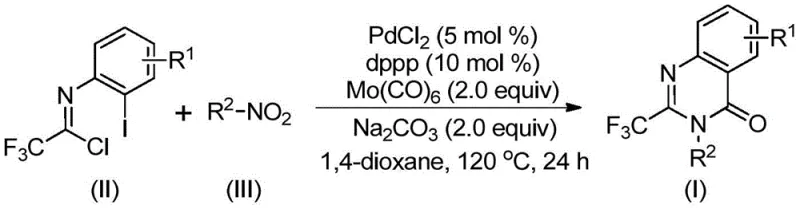
In stark contrast, the methodology outlined in patent CN112480015B presents a paradigm shift towards simplicity and efficiency. This novel approach utilizes readily available and inexpensive nitro compounds alongside trifluoroethylimidoyl chloride as starting materials, effectively removing the need for pre-functionalized precursors. The reaction proceeds via a transition metal palladium-catalyzed carbonylation cascade in a single pot, utilizing molybdenum hexacarbonyl as a safe, solid surrogate for toxic carbon monoxide gas. This innovation allows the reaction to be conducted under atmospheric pressure at a moderate temperature of 120°C, drastically improving operational safety and reducing energy consumption. The system demonstrates exceptional functional group tolerance, accommodating various substituents on both the aromatic ring and the nitrogen atom, which enables the rapid generation of diverse chemical libraries for structure-activity relationship studies without the need for protecting group strategies.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic elegance of this transformation lies in its seamless integration of reduction, coupling, and cyclization steps within a single reaction vessel. The process initiates with the reduction of the nitro compound to the corresponding amine by molybdenum hexacarbonyl, which simultaneously acts as the carbon monoxide source. Following this reduction, a base-promoted intermolecular carbon-nitrogen bond coupling occurs between the generated amine and the trifluoroethylimidoyl chloride, yielding a trifluoroacetamidine derivative in situ. The palladium catalyst, specifically palladium chloride coordinated with the dppp ligand, then inserts into the carbon-iodine bond of the imidoyl chloride moiety to form a reactive divalent palladium intermediate. As the reaction temperature is maintained at 120°C, the molybdenum hexacarbonyl decomposes to release carbon monoxide, which subsequently inserts into the carbon-palladium bond to generate a key acyl-palladium species.
This acyl-palladium intermediate undergoes an intramolecular nucleophilic attack facilitated by the base, promoting the formation of a palladium-nitrogen bond and closing the seven-membered cyclic palladium intermediate. The final step involves a reductive elimination that releases the desired 2-trifluoromethyl substituted quinazolinone product and regenerates the active palladium catalyst for the next cycle. This intricate dance of organometallic steps ensures high atom efficiency and minimizes the formation of side products. From an impurity control perspective, the one-pot nature of the reaction reduces the exposure of reactive intermediates to the environment, thereby limiting degradation pathways. The use of a robust ligand system like 1,3-bis(diphenylphosphino)propane (dppp) stabilizes the palladium center, preventing the formation of palladium black and ensuring consistent catalytic turnover throughout the 16 to 30-hour reaction window, which is critical for achieving the reported high yields ranging from 69% to 96%.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction parameters to maximize throughput and purity. The protocol dictates a specific molar ratio of catalyst to ligand to base, typically utilizing palladium chloride at 5 mol% and dppp at 10 mol%, with sodium carbonate serving as the base additive. The reaction is carried out in an aprotic solvent, with 1,4-dioxane identified as the optimal medium due to its ability to dissolve all reagents effectively and promote high conversion rates. While the standard procedure targets a 24-hour reaction time at 120°C, the window can be adjusted between 16 to 30 hours depending on the specific electronic nature of the substrates involved. Detailed standardized operating procedures for executing this synthesis, including workup and purification guidelines, are provided below to ensure reproducibility at scale.
- Combine palladium chloride, dppp ligand, sodium carbonate, Mo(CO)6, trifluoroethylimidoyl chloride, and nitro compound in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 120°C and stir for 16 to 30 hours to allow the carbonylation cascade and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material portfolio; by utilizing commodity nitro compounds and avoiding exotic, pre-activated building blocks, the supply chain becomes more resilient and less susceptible to vendor lock-in. The elimination of high-pressure gas cylinders and the associated safety infrastructure significantly lowers the barrier to entry for manufacturing partners, allowing for a broader base of qualified suppliers who can produce these intermediates without specialized high-pressure facilities. This accessibility translates directly into improved negotiation leverage and potential cost reductions in API manufacturing, as the operational overhead is markedly lower compared to traditional carbonylation processes.
- Cost Reduction in Manufacturing: The economic profile of this process is highly favorable due to the use of earth-abundant starting materials and the avoidance of expensive noble metals like ruthenium or platinum. By substituting toxic carbon monoxide gas with solid molybdenum hexacarbonyl, the process eliminates the need for costly gas handling systems and rigorous safety monitoring, leading to substantial operational savings. Furthermore, the high reaction efficiency and broad substrate scope mean that fewer batches are rejected due to poor yield or impurity profiles, optimizing the overall cost of goods sold. The simplified post-treatment process, involving basic filtration and standard column chromatography, reduces solvent consumption and labor hours associated with complex purification sequences.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the commercial availability of all key reagents, including the palladium catalyst, ligands, and nitro substrates, which are standard stock items in the global chemical market. The robustness of the reaction conditions, tolerating a wide range of functional groups such as halogens, alkyls, and trifluoromethyl groups, ensures that supply disruptions caused by the inability to source specific niche precursors are minimized. This flexibility allows manufacturers to pivot quickly between different analogues within the quinazolinone series without retooling the entire production line, ensuring continuous supply continuity for downstream drug development projects.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this one-pot method aligns well with green chemistry principles by reducing the number of isolation steps and associated waste generation. The ability to scale the reaction from milligram to gram levels, as demonstrated in the patent data, indicates a clear pathway to kilogram and ton-scale production with minimal process re-engineering. The use of standard organic solvents like dioxane, which can be recovered and recycled, further enhances the environmental profile. This ease of scale-up ensures that as clinical demand grows, the manufacturing capacity can be expanded rapidly to meet commercial launch timelines without compromising on quality or regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and limitations. Understanding these details is essential for R&D teams evaluating this route for their specific pipeline candidates and for procurement teams assessing vendor qualifications.
Q: What are the key advantages of this one-pot synthesis method over traditional routes?
A: This method eliminates the need for high-pressure carbon monoxide gas and expensive pre-activated substrates. It utilizes cheap nitro compounds and operates under atmospheric pressure with high substrate compatibility and yields up to 96%.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the patent explicitly states the method can be expanded to the gram level and beyond. The use of standard solvents like dioxane and commercially available catalysts facilitates easy scale-up for industrial production.
Q: What is the role of Molybdenum Hexacarbonyl in this reaction?
A: Mo(CO)6 serves as a solid, safe substitute for toxic carbon monoxide gas. Under heating conditions, it releases CO in situ, which inserts into the carbon-palladium bond to form the necessary acyl-palladium intermediate for ring closure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has extensively evaluated the technology described in patent CN112480015B and possesses the technical capability to implement this palladium-catalyzed carbonylation route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from preclinical research to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone delivered meets the highest industry standards for potency and impurity control.
We invite you to collaborate with us to leverage this advanced synthetic platform for your next-generation therapeutic programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target timelines. We encourage you to reach out today to discuss your project needs,索取 specific COA data for our reference standards, and review our comprehensive route feasibility assessments. Let us help you secure a reliable supply of high-quality pharmaceutical intermediates while optimizing your overall development budget.
