Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Scale-Up
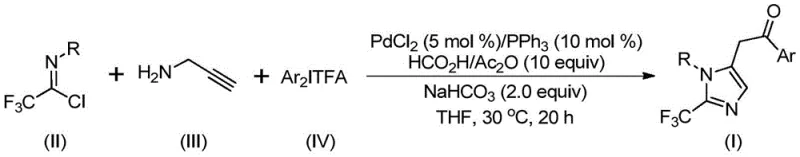
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to incorporate trifluoromethyl groups into heterocyclic scaffolds, driven by the profound impact of fluorination on metabolic stability and bioavailability. Patent CN111423381B discloses a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds that addresses many historical challenges in this domain. This technology leverages a transition metal palladium-catalyzed carbonylation series reaction, utilizing cheap and easily obtained starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salts. The process operates under remarkably mild conditions, specifically at 30°C for 18 to 20 hours, demonstrating high reaction efficiency and exceptional substrate compatibility. For R&D directors and process chemists, this represents a significant leap forward, offering a pathway to diverse molecular architectures without the need for extreme temperatures or pressures that often complicate scale-up efforts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has been fraught with significant operational and safety hurdles. Literature reports indicate that common methods often rely on reacting synthons directly with trifluoromethyl groups, frequently utilizing reagents like trifluorodiazoethane. While effective in specific contexts, these reagents can be unstable, hazardous to handle on a large scale, and limited in their commercial availability. Furthermore, conventional cyclization strategies often require harsh reaction conditions, including elevated temperatures and strong bases, which can lead to the decomposition of sensitive functional groups or the formation of complex impurity profiles. The reliance on high-pressure carbon monoxide gas for carbonylation steps also introduces severe safety risks and requires specialized equipment, creating bottlenecks for reliable pharmaceutical intermediate suppliers aiming for rapid commercialization.
The Novel Approach
The novel approach detailed in the patent fundamentally shifts the paradigm by employing a palladium-catalyzed cascade reaction that integrates C-N bond formation and carbonylation in a single pot. By using trifluoroethylimidoyl chloride and propargylamine as the core building blocks, the method bypasses the need for dangerous diazo compounds. A critical innovation is the use of a formic acid and acetic anhydride mixture as a safe, in-situ source of carbon monoxide, eliminating the hazards associated with storing and handling CO gas cylinders. This strategy not only enhances safety but also simplifies the reactor setup, making it highly attractive for cost reduction in API manufacturing. The reaction proceeds smoothly at 30°C, preserving the integrity of diverse substituents on the aryl rings, such as halogens, nitro groups, and alkyl chains, thereby expanding the utility of this method for generating libraries of biologically active molecules.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific targets. The reaction initiates with the formation of a trifluoroacetamidine compound through an intermolecular carbon-nitrogen bond promoted by the alkali additive, sodium bicarbonate. Following isomerization, the palladium catalyst, typically PdCl2 coordinated with triphenylphosphine, activates the alkyne moiety of the propargylamine derivative. This leads to a palladation event, generating an alkenyl palladium intermediate which subsequently isomerizes to a more stable alkyl palladium species. The introduction of the carbonyl group occurs via the insertion of carbon monoxide, released from the formic acid/acetic anhydride system, into the palladium-carbon bond to form an acyl palladium intermediate.
The catalytic cycle concludes with the oxidative addition of the diaryl iodonium salt to the acyl palladium species, forming a transient tetravalent palladium intermediate. This high-energy species undergoes reductive elimination to forge the final carbon-carbon bond, releasing the 2-trifluoromethyl-substituted imidazole product and regenerating the active palladium catalyst. This intricate dance of organometallic steps ensures high atom economy and selectivity. From an impurity control perspective, the mild conditions minimize side reactions such as polymerization of the alkyne or hydrolysis of the imidoyl chloride. The use of sodium bicarbonate as a mild base further buffers the reaction environment, preventing acid-catalyzed degradation pathways that are common in harsher acidic or basic media, thus ensuring a cleaner crude product profile prior to purification.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The operational simplicity of this protocol makes it highly amenable to both laboratory discovery and pilot plant production. The procedure involves charging a reactor with the palladium catalyst system, the CO source components, and the specific substrates in an aprotic organic solvent like tetrahydrofuran (THF). The reaction mixture is stirred at a controlled temperature of 30°C, allowing the multi-component coupling to proceed to completion over a period of 16 to 24 hours. Post-reaction processing is straightforward, involving filtration to remove inorganic salts followed by standard silica gel column chromatography to isolate the pure product. For detailed standard operating procedures and specific stoichiometric ratios tailored to your target molecule, please refer to the synthesis guide below.
- Combine palladium chloride, triphenylphosphine, sodium bicarbonate, acetic anhydride, and formic acid in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryl iodonium salt to the reaction mixture under stirring.
- Maintain the reaction at 30°C for 16-24 hours, then filter and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic and logistical implications of this patent are substantial. The shift towards milder conditions and safer reagents translates directly into reduced operational expenditures and enhanced supply continuity. By eliminating the need for specialized high-pressure equipment and hazardous gas handling infrastructure, capital expenditure for new production lines is significantly lowered. Furthermore, the reliance on commodity chemicals ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with supply chain disruptions for exotic reagents.
- Cost Reduction in Manufacturing: The utilization of inexpensive starting materials such as aromatic amines (precursors to imidoyl chlorides) and propargylamine drives down the direct material costs. Additionally, the in-situ generation of carbon monoxide removes the logistical burden and safety costs associated with purchasing and storing high-pressure CO gas. The mild reaction temperature of 30°C drastically reduces energy consumption for heating and cooling compared to traditional reflux conditions, leading to substantial utility savings over long production runs. The high conversion rates observed across various substrates minimize waste generation and maximize the yield of valuable intermediates per batch.
- Enhanced Supply Chain Reliability: The starting materials, including trifluoroethylimidoyl chlorides and diaryl iodonium salts, can be synthesized from widely available commercial precursors like aryl boronic acids and aryl iodides. This decentralization of raw material sourcing means that manufacturers are not dependent on a single supplier for a niche reagent. The robustness of the reaction against various functional groups allows for flexibility in sourcing; if one specific substituted aniline is unavailable, alternative analogs can often be substituted without re-optimizing the entire process, ensuring continuous production schedules for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The protocol's compatibility with gram-level synthesis and its potential for industrial large-scale production make it an ideal candidate for technology transfer. The use of THF as a solvent is well-established in the industry, with mature recovery and recycling protocols that align with green chemistry principles. The avoidance of heavy metal contaminants beyond the catalytic amount of palladium, which can be effectively scavenged, simplifies the purification process and ensures the final product meets stringent regulatory limits for residual metals in APIs. This streamlined workflow reduces the environmental footprint and accelerates the time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for their specific projects.
Q: What is the primary advantage of this synthesis method regarding reaction conditions?
A: The primary advantage is the exceptionally mild reaction temperature of 30°C, which significantly reduces energy consumption compared to traditional high-temperature heterocycle syntheses and minimizes thermal degradation of sensitive functional groups.
Q: How is the carbon monoxide source managed in this protocol?
A: Instead of using hazardous high-pressure carbon monoxide gas, this method utilizes a safe in-situ generation system where formic acid and acetic anhydride react to release CO, greatly enhancing operational safety for industrial scale-up.
Q: What represents the key structural innovation in the starting materials?
A: The method utilizes readily available trifluoroethylimidoyl chlorides and propargylamines, avoiding the need for specialized or unstable trifluoromethyl synthons like trifluorodiazoethane, thereby improving supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed methodology for developing next-generation therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl imidazole intermediate meets the highest global standards for quality and consistency required by top-tier pharmaceutical companies.
We invite you to leverage our technical expertise to optimize this synthesis for your specific pipeline needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines while maximizing value.
