Advanced Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Pharmaceutical Applications
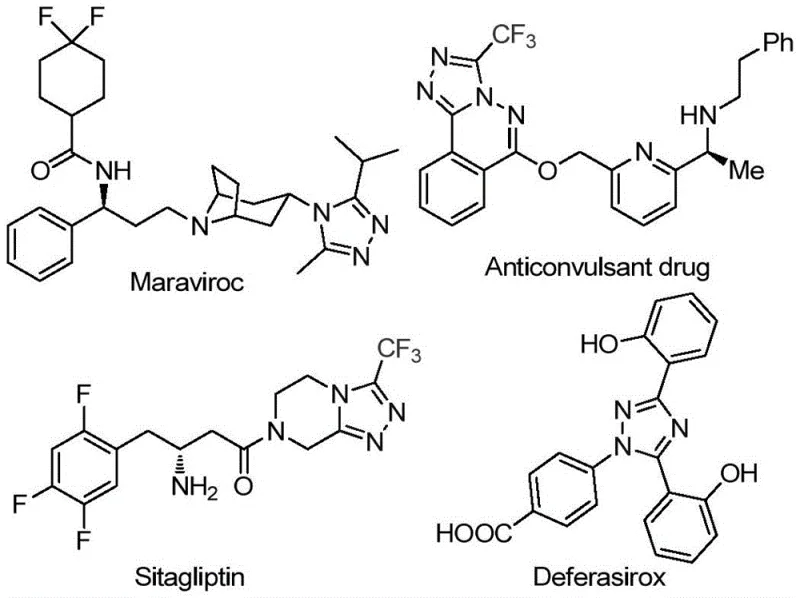
The pharmaceutical industry continuously seeks robust synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, which serve as critical scaffolds in numerous bioactive molecules. As highlighted in patent CN113105402B, a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds has been developed that addresses significant bottlenecks in traditional manufacturing. These triazole derivatives are not merely academic curiosities; they form the core structural backbone of major therapeutic agents such as Maraviroc, Sitagliptin, and Deferasirox, as illustrated in the structural diversity shown below. The introduction of a trifluoromethyl group into these heterocyclic systems is strategically vital, as it markedly enhances physicochemical properties including metabolic stability, lipophilicity, and bioavailability, thereby optimizing the drug-like characteristics of the final active pharmaceutical ingredient.
For procurement managers and supply chain directors, the significance of this patent lies in its potential to streamline the sourcing of high-purity pharmaceutical intermediates. By leveraging a non-metal promoted synthesis, manufacturers can bypass the stringent regulatory and logistical hurdles associated with heavy metal catalysts. This innovation represents a paradigm shift towards greener, more cost-efficient chemistry that aligns perfectly with modern sustainability goals while ensuring a reliable supply of complex heterocyclic building blocks for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, especially those bearing both trifluoromethyl and acyl groups, has been fraught with chemical and operational challenges. Conventional methodologies often rely heavily on transition metal catalysis, utilizing expensive and toxic metals such as copper or palladium to facilitate the cyclization steps. These metal-catalyzed processes typically demand rigorous exclusion of moisture and oxygen, necessitating specialized equipment like Schlenk lines or gloveboxes, which drastically increases capital expenditure and operational complexity. Furthermore, the removal of trace heavy metal residues from the final product to meet pharmacopeial standards requires additional purification steps, such as scavenging or extensive chromatography, leading to significant yield losses and extended production timelines. The reliance on sensitive catalysts also introduces variability in batch-to-batch consistency, posing a risk to supply chain reliability for critical API intermediates.
The Novel Approach
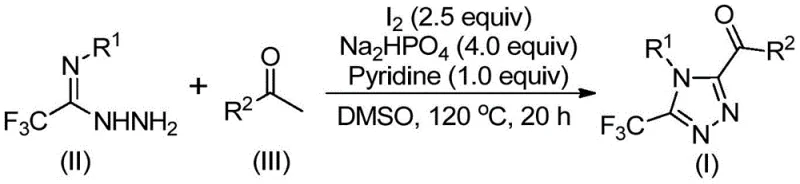
In stark contrast, the methodology disclosed in patent CN113105402B offers a streamlined, metal-free alternative that utilizes elemental iodine and dimethyl sulfoxide (DMSO) to drive the reaction. This approach eliminates the need for expensive transition metals and the associated removal protocols, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. The reaction proceeds through a tandem sequence involving iodination and Kornblum oxidation, followed by condensation and cyclization, all within a single pot. As depicted in the general reaction scheme below, the process tolerates a wide range of functional groups and does not require anhydrous or anaerobic conditions. This operational simplicity allows for easier handling and significantly lowers the barrier for commercial scale-up of complex pharmaceutical intermediates, making it an attractive option for large-scale production facilities aiming to optimize throughput and minimize waste.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation events that ensures high regioselectivity and yield. Initially, the aryl ethyl ketone undergoes an iodine-mediated alpha-iodination in DMSO, which subsequently facilitates a Kornblum oxidation to generate an aryl 1,2-diketone intermediate in situ. This reactive diketone species then undergoes a dehydration condensation with the trifluoroethylimide hydrazide to form a hydrazone intermediate. The final step involves an intramolecular oxidative cyclization promoted by the iodine base system, closing the triazole ring to afford the desired 3,4,5-trisubstituted product. This cascade mechanism is highly efficient because it avoids the isolation of unstable intermediates, thereby minimizing decomposition pathways and maximizing overall atom economy. The use of sodium dihydrogen phosphate and pyridine acts as a buffered base system, carefully controlling the pH to favor cyclization over hydrolysis, which is critical for maintaining high purity profiles.
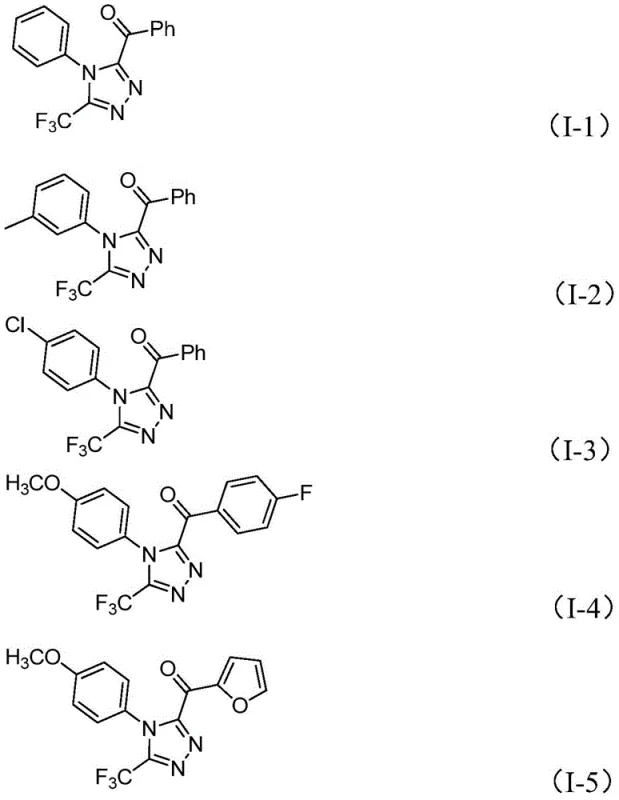
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed variants. The absence of metal centers eliminates the risk of metal-ligand complexation side reactions that often generate difficult-to-remove impurities. Furthermore, the mild reaction temperatures (110-130°C) and the specific stoichiometry of reagents (e.g., iodine at 2.5 equivalents) are optimized to suppress over-oxidation or polymerization of the hydrazide component. The broad substrate scope demonstrated in the patent, covering various substituted aryl and heteroaryl groups as shown in the examples below, confirms the robustness of this mechanistic pathway. Whether the substrate contains electron-withdrawing groups like chlorine or trifluoromethyl, or electron-donating groups like methoxy, the reaction maintains consistent efficiency, ensuring a stable impurity profile regardless of the specific derivative being synthesized.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory R&D and pilot plant operations. The protocol begins with the dissolution of the aryl ketone and iodine in DMSO, followed by a controlled heating phase to generate the oxidized intermediate. Subsequent addition of the hydrazide and base components triggers the cyclization. The workup procedure is equally straightforward, involving simple filtration and silica gel treatment followed by column chromatography, avoiding complex aqueous extractions or distillation steps that can degrade sensitive products. For detailed operational parameters, stoichiometry, and specific temperature ramps required to replicate these results, please refer to the standardized synthesis guide provided below.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours to complete the cyclization, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, this patented process offers compelling strategic advantages that extend beyond mere chemical novelty. By shifting away from precious metal catalysis, the manufacturing cost structure is fundamentally altered, removing the volatility associated with rhodium, palladium, or copper prices. The use of commodity chemicals like elemental iodine and DMSO ensures that raw material availability remains high, mitigating the risk of supply disruptions that often plague specialty catalyst markets. Additionally, the simplified purification train reduces solvent consumption and waste generation, aligning with environmental compliance standards and lowering disposal costs. These factors collectively enhance the economic viability of producing these valuable heterocycles at scale.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a significant cost center from the bill of materials. Furthermore, the avoidance of rigorous anhydrous conditions reduces energy consumption related to solvent drying and inert gas purging. The simplified downstream processing, which does not require specialized metal scavenging resins, further drives down the cost per kilogram, allowing for more competitive pricing of the final pharmaceutical intermediate without compromising margin.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk commodity reagents such as aryl ketones and iodine ensures a resilient supply chain. Unlike proprietary ligands or sensitive organometallic complexes that may have single-source suppliers, the inputs for this process are globally sourced and stable. This redundancy minimizes the risk of production halts due to raw material shortages, ensuring consistent delivery schedules for downstream API manufacturers who depend on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively at the gram level with clear pathways to kilogram and tonne scales. The absence of heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site. This ease of scale-up, combined with reduced hazardous waste generation, facilitates faster regulatory approval for new drug applications and supports sustainable manufacturing initiatives increasingly demanded by global health authorities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and scope of the method for potential licensees or manufacturing partners. Understanding these nuances is essential for evaluating the fit of this technology within existing production portfolios.
Q: Does this synthesis method require expensive heavy metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a promoter instead of toxic or expensive transition metal catalysts, significantly reducing raw material costs and simplifying purification.
Q: What are the reaction conditions regarding moisture and oxygen sensitivity?
A: Unlike many conventional heterocyclic syntheses, this process does not require strict anhydrous or oxygen-free conditions, making it highly suitable for standard industrial reactor setups without specialized inert atmosphere equipment.
Q: Can this method be scaled for commercial production of API intermediates?
A: Yes, the patent explicitly states that the method can be easily expanded to the gram level and beyond, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-promoted synthesis for the next generation of triazole-based therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this DMSO-based process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this advanced chemistry for your pipeline. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this metal-free route can optimize your COGS. Please contact our technical procurement team today to request specific COA data for our triazole library and to discuss route feasibility assessments for your custom synthesis projects.
