Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-Ones for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-Ones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those with proven biological significance. Patent CN112538054B, published in September 2022, introduces a groundbreaking preparation method for 1,2,4-triazole-3-one compounds that addresses many longstanding synthetic challenges. This technology leverages a transition metal palladium-catalyzed carbonylation tandem cyclization reaction, utilizing readily available chlorohydrazones and sodium azide as starting materials. The significance of this development cannot be overstated, as the 1,2,4-triazole-3-one scaffold is a privileged structure found in numerous bioactive molecules ranging from antifungal agents like Itraconazole to potent antitumor and anticonvulsant drugs. 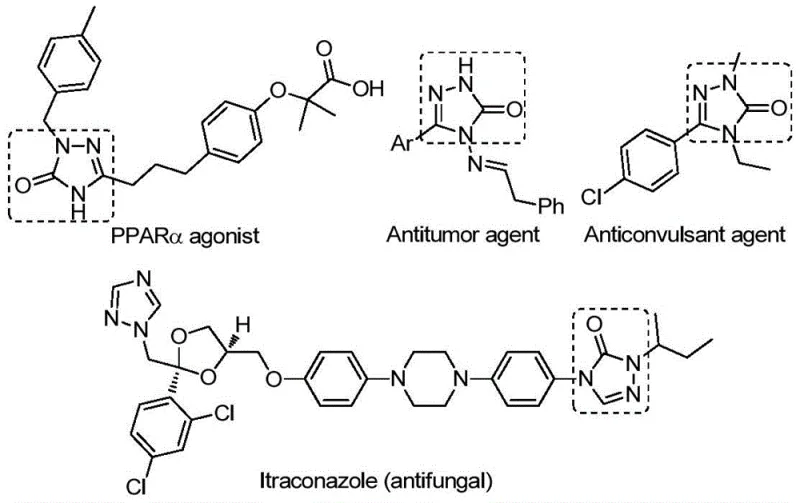 By establishing a reliable pathway to access these diverse structures, this patent offers a strategic advantage for any organization aiming to secure a reliable pharmaceutical intermediate supplier status. The method is characterized by its operational simplicity, high reaction efficiency, and exceptional substrate compatibility, allowing for the synthesis of derivatives substituted with various functional groups through rational substrate design.
By establishing a reliable pathway to access these diverse structures, this patent offers a strategic advantage for any organization aiming to secure a reliable pharmaceutical intermediate supplier status. The method is characterized by its operational simplicity, high reaction efficiency, and exceptional substrate compatibility, allowing for the synthesis of derivatives substituted with various functional groups through rational substrate design.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazole-3-one compounds has been fraught with significant technical hurdles that impede efficient commercial production. Traditional literature reports describe several common routes, such as the cyclization of benzoyl hydrazide with urea under the influence of potassium hydroxide, or the tandem cyclization of hydrazides with isocyanates. Other methods involve the condensation of thioamides with hydrazines at elevated temperatures or the reaction of acyl isocyanates with monosubstituted hydrazines. These conventional approaches suffer from severe limitations, including harsh reaction conditions that often require strong bases or extreme thermal energy, which can degrade sensitive functional groups. Furthermore, these processes typically involve complicated multi-step sequences, necessitating the pre-activation of reaction substrates, which increases both material costs and processing time. The narrow substrate scope of these older methods means that introducing diverse substituents often leads to drastically reduced yields or complete reaction failure, making them unsuitable for the rapid exploration of structure-activity relationships required in modern drug discovery.
The Novel Approach
In stark contrast to these legacy techniques, the novel methodology disclosed in the patent utilizes a sophisticated yet practical palladium-catalyzed system that streamlines the entire synthetic process. This approach employs chlorohydrazones and sodium azide as inexpensive and easily accessible starting materials, reacting them in the presence of a palladium catalyst, a specialized ligand, and a carbon monoxide substitute known as TFBen. The reaction proceeds smoothly in an organic solvent at moderate temperatures between 100°C and 120°C, eliminating the need for hazardous high-pressure carbon monoxide gas cylinders. This innovation not only enhances safety protocols within the manufacturing facility but also simplifies the engineering requirements for reactor setup. The method demonstrates remarkable versatility, accommodating a wide range of substituents on both the hydrazone and the nitrogen atom, thereby enabling the efficient production of a broad library of 1,2,4-triazole-3-one derivatives. This flexibility is crucial for cost reduction in API manufacturing, as it allows chemists to optimize molecular properties without being constrained by synthetic feasibility.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
To fully appreciate the technical sophistication of this process, one must delve into the proposed catalytic cycle that drives the transformation. The reaction is believed to initiate with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the solid carbon monoxide substitute, TFBen, undergoes thermal decomposition to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond, forming a critical acyl palladium intermediate. Subsequently, this intermediate reacts with sodium azide to produce an acyl azide species, which is prone to undergoing a Curtius rearrangement. This rearrangement generates an isocyanate intermediate, which then participates in an intramolecular nucleophilic addition reaction to close the ring, ultimately yielding the target 1,2,4-triazole-3-one compound.  Understanding this mechanism is vital for R&D directors focused on purity and impurity profiles, as it highlights the precise points where side reactions might occur and how they are mitigated by the specific choice of ligands and reaction conditions.
Understanding this mechanism is vital for R&D directors focused on purity and impurity profiles, as it highlights the precise points where side reactions might occur and how they are mitigated by the specific choice of ligands and reaction conditions.
Furthermore, the control of impurities in this system is achieved through the careful selection of the ligand system, specifically the combination of Pd2(dba)3 and Xantphos. This ligand architecture stabilizes the palladium center throughout the catalytic cycle, preventing the formation of palladium black and ensuring high turnover numbers. The use of aprotic solvents like 1,4-dioxane is also critical, as protic solvents or polar aprotic solvents like DMSO were found to inhibit the reaction or lower efficiency significantly. The substrate scope is impressively broad, tolerating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups like halogens and trifluoromethyl groups on the aromatic rings.  This tolerance ensures that the final products maintain high structural integrity and purity, which is a prerequisite for high-purity pharmaceutical intermediates intended for downstream coupling reactions. The ability to synthesize compounds with bulky groups like tert-butyl or naphthyl further underscores the robustness of this catalytic system against steric hindrance.
This tolerance ensures that the final products maintain high structural integrity and purity, which is a prerequisite for high-purity pharmaceutical intermediates intended for downstream coupling reactions. The ability to synthesize compounds with bulky groups like tert-butyl or naphthyl further underscores the robustness of this catalytic system against steric hindrance.
How to Synthesize 1,2,4-Triazole-3-One Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize yield and safety. The process begins with the precise weighing of the palladium catalyst, ligand, and the carbon monoxide source, followed by their dissolution in the chosen organic solvent. The chlorohydrazone and sodium azide are then introduced, and the mixture is heated under controlled conditions. It is essential to monitor the reaction progress to determine the optimal endpoint, typically falling within the 16 to 30-hour window depending on the specific substrate electronics. Detailed standardized synthesis steps see the guide below.
- Combine palladium catalyst Pd2(dba)3, ligand Xantphos, carbon monoxide substitute TFBen, chlorohydrazone substrate, and sodium azide in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 100-120°C and maintain stirring for 16 to 30 hours to ensure complete conversion of the starting materials.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative benefits that directly impact the bottom line and operational resilience. The shift away from complex, multi-step traditional syntheses to a direct, one-pot carbonylation strategy significantly reduces the number of unit operations required. This consolidation of steps translates to substantial cost savings by minimizing solvent consumption, reducing labor hours, and lowering energy demands associated with heating and cooling cycles across multiple stages. Moreover, the reliance on commercially available and inexpensive reagents such as sodium azide and simple chlorohydrazones ensures a stable and predictable supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized starting materials that often plague specialty chemical manufacturing.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and hazardous reagents often used in older methods, such as isocyanates or activated acyl chlorides that require strict handling protocols. By utilizing a solid CO surrogate like TFBen, the process avoids the capital expenditure and safety costs associated with high-pressure gas infrastructure. Additionally, the high reaction efficiency and broad substrate compatibility mean that fewer batches are rejected due to low yields or impurity issues, leading to a more consistent and cost-effective production output. The simplified post-treatment process, involving basic filtration and standard column chromatography, further reduces the operational overhead compared to complex purification sequences required by alternative routes.
- Enhanced Supply Chain Reliability: Securing a steady flow of raw materials is critical for maintaining production schedules, and this method excels in that regard. The primary building blocks, chlorohydrazones, can be rapidly synthesized from corresponding acid chlorides and hydrazines, which are commodity chemicals available from multiple global vendors. This redundancy in the supply base prevents bottlenecks and ensures that production can continue even if one supplier faces disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain volatility. For supply chain heads, this translates to reduced lead time for high-purity pharmaceutical intermediates and greater confidence in meeting delivery commitments to downstream API manufacturers.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, this process aligns well with environmental sustainability goals. The use of 1,4-dioxane, while requiring careful handling, allows for efficient recycling and recovery, minimizing waste generation. The absence of heavy metal waste streams typical of stoichiometric oxidation or reduction methods simplifies wastewater treatment and disposal. The method has been demonstrated to be scalable, with the patent noting successful expansion to mmol scales and implying readiness for larger batch sizes. This scalability ensures that the transition from laboratory discovery to commercial scale-up of complex pharmaceutical intermediates is seamless, allowing manufacturers to respond quickly to market demand without extensive re-engineering of the process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity for potential partners and licensees. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production lines.
Q: What are the key advantages of this Pd-catalyzed method over traditional synthesis?
A: Unlike traditional methods requiring harsh conditions like strong bases or high temperatures, this novel approach utilizes mild conditions (100°C) and avoids pre-activation of substrates, resulting in higher yields and broader functional group tolerance.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is scalable and operationally simple. The use of commercially available reagents like sodium azide and stable chlorohydrazones facilitates supply chain reliability for commercial scale-up.
Q: What is the role of TFBen in this reaction mechanism?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide substitute. Under heating conditions, it releases CO in situ, which inserts into the carbon-palladium bond to form the necessary acyl palladium intermediate without requiring hazardous high-pressure CO gas.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-One Supplier
The technological advancements detailed in patent CN112538054B represent a significant leap forward in the synthesis of valuable heterocyclic scaffolds, yet realizing their full potential requires a partner with deep technical expertise and manufacturing capacity. NINGBO INNO PHARMCHEM stands ready to leverage this innovative chemistry, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch meets the exacting standards required by the global pharmaceutical industry. We understand that consistency is key, and our state-of-the-art facilities are designed to handle complex catalytic reactions with precision and safety.
We invite you to explore how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a reliable supply of high-quality intermediates that will accelerate your drug development timelines and strengthen your market position.
