Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Compounds for Commercial Scale-up
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Compounds for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing nitrogen-containing heterocycles, particularly those with proven biological significance. A recent technological breakthrough detailed in patent CN112538054B introduces a highly efficient preparation method for 1,2,4-triazole-3-one compounds, a privileged scaffold found in numerous bioactive molecules ranging from antifungal agents to antitumor drugs. This novel approach leverages a transition metal palladium-catalyzed carbonylation tandem cyclization reaction, utilizing readily available chlorohydrazones and sodium azide as starting materials. Unlike traditional methods that often suffer from harsh conditions and limited substrate scope, this invention provides a streamlined pathway that operates under relatively mild thermal conditions while maintaining exceptional reaction efficiency. For R&D directors and process chemists, this represents a significant opportunity to optimize synthetic routes for complex API intermediates, ensuring higher purity profiles and reduced impurity burdens in the final drug substance.
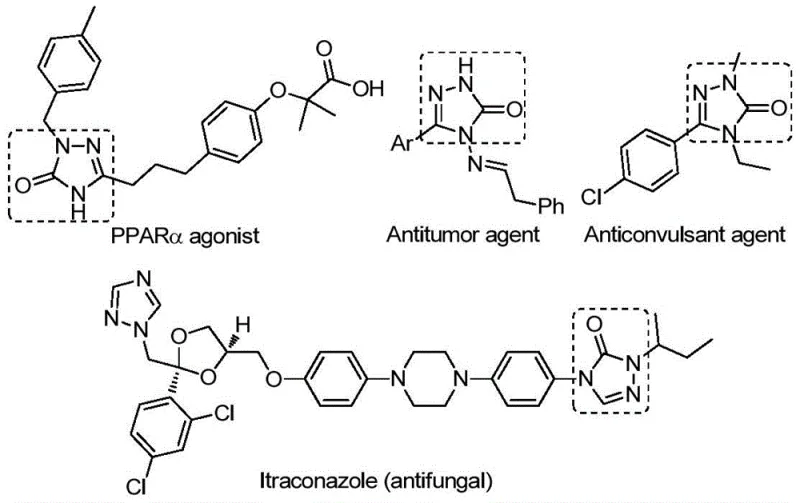
The structural versatility of the 1,2,4-triazole-3-one core is exemplified by its presence in diverse therapeutic classes, as illustrated in the provided chemical structures. These compounds serve as critical pharmacophores in PPARα agonists, antitumor agents, anticonvulsants, and potent antifungals like Itraconazole. The ability to access these scaffolds through a unified, catalytic strategy allows medicinal chemists to rapidly explore structure-activity relationships (SAR) without being bottlenecked by cumbersome synthesis. By employing a palladium catalyst system combined with a ligand and a solid carbon monoxide substitute, the method described in CN112538054B effectively bypasses the logistical and safety challenges associated with gaseous CO, making it an attractive candidate for both laboratory discovery and industrial production environments where safety and throughput are paramount concerns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazole-3-one derivatives has been plagued by significant operational drawbacks that hinder their widespread adoption in large-scale manufacturing. Conventional literature methods typically rely on the cyclization of benzoyl hydrazides with urea under strong basic conditions, or the tandem reaction of hydrazides with isocyanates, which often require stringent moisture control and expensive reagents. Other approaches involve the condensation of thioamides with hydrazines at elevated temperatures, leading to poor atom economy and difficult purification processes due to the formation of sulfur-containing byproducts. Furthermore, many existing protocols necessitate the pre-activation of substrates, adding extra synthetic steps that cumulatively reduce overall yield and increase waste generation. These traditional pathways frequently exhibit narrow substrate compatibility, failing to tolerate sensitive functional groups such as halogens or electron-withdrawing moieties, which limits their utility in the synthesis of diversified pharmaceutical libraries required for modern drug discovery programs.
The Novel Approach
In stark contrast to these legacy methods, the innovative process disclosed in the patent utilizes a palladium-catalyzed carbonylation strategy that fundamentally simplifies the synthetic architecture. By reacting chlorohydrazones directly with sodium azide in the presence of a palladium catalyst and TFBen (a solid CO source), the method achieves a tandem cyclization in a single operational step. This approach eliminates the need for handling toxic carbon monoxide gas, replacing it with a safe, easy-to-handle solid surrogate that releases CO in situ under heating. The reaction proceeds efficiently in common organic solvents like 1,4-dioxane at 100°C, demonstrating remarkable tolerance for a wide array of substituents including alkyl, aryl, and halogen groups. As shown in the general reaction scheme below, the transformation is direct and high-yielding, offering a practical solution for producing high-purity intermediates with minimal downstream processing requirements.

Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
The mechanistic pathway of this transformation is a sophisticated sequence of organometallic steps that ensures high selectivity and conversion. The cycle initiates with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the TFBen additive undergoes thermal decomposition to release carbon monoxide, which subsequently inserts into the carbon-palladium bond to form an acyl-palladium species. This acyl intermediate then reacts with sodium azide to generate an acyl azide compound in situ. Crucially, this acyl azide undergoes a Curtius rearrangement, a thermally driven process that releases nitrogen gas and forms an isocyanate intermediate. The final step involves an intramolecular nucleophilic addition where the hydrazine nitrogen attacks the electrophilic carbon of the isocyanate, closing the ring to furnish the desired 1,2,4-triazole-3-one product. This elegant cascade minimizes side reactions and ensures that the carbonyl group is incorporated precisely where needed within the heterocyclic framework.
From an impurity control perspective, this mechanism offers distinct advantages over non-catalytic thermal cyclizations. Because the reaction is driven by a well-defined catalytic cycle rather than brute-force thermal energy alone, the formation of random polymeric byproducts or decomposition species is significantly suppressed. The use of sodium azide in slight excess ensures complete consumption of the acyl-palladium intermediate, preventing the accumulation of unreacted starting materials that could complicate purification. Moreover, the choice of ligands such as Xantphos stabilizes the palladium center, preventing catalyst aggregation and deactivation, which is a common issue in carbonylation reactions. This stability translates to consistent batch-to-batch reproducibility, a critical factor for regulatory compliance in pharmaceutical manufacturing. The ability to tune the electronic properties of the chlorohydrazone substrate without disrupting the catalytic cycle further underscores the robustness of this method for generating diverse chemical libraries.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and safety. The protocol dictates the use of a palladium source such as tris(dibenzylideneacetone)dipalladium (Pd2(dba)3) paired with a bidentate phosphine ligand like Xantphos to facilitate the oxidative addition and CO insertion steps. The reaction is typically conducted in an aprotic solvent such as 1,4-dioxane, which provides the optimal polarity for dissolving both the organic substrates and the inorganic sodium azide. Heating the mixture to 100°C for a duration of 16 to 30 hours allows sufficient time for the slow release of CO from TFBen and the subsequent rearrangement steps to reach completion. Detailed standardized synthesis steps for this procedure are provided in the guide below.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), CO substitute (TFBen), chlorohydrazone, and sodium azide in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 100°C and maintain stirring for 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this palladium-catalyzed methodology presents a compelling value proposition centered on cost efficiency and risk mitigation. The primary raw materials, chlorohydrazones and sodium azide, are commodity chemicals that are inexpensive and readily available from multiple global suppliers, reducing dependency on single-source vendors. The replacement of high-pressure carbon monoxide cylinders with the solid surrogate TFBen drastically reduces the capital expenditure required for specialized high-pressure reactors and gas handling infrastructure. This shift not only lowers the barrier to entry for manufacturing but also significantly enhances workplace safety by eliminating the risk of CO leaks, a critical consideration for insurance and liability management in chemical production facilities. Furthermore, the simplified workup procedure, which often involves simple filtration and chromatography, reduces solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable production model.
- Cost Reduction in Manufacturing: The elimination of expensive pre-activated reagents and the use of cheap, bulk-available starting materials like sodium azide lead to substantial raw material cost savings. Additionally, the high atom economy of the carbonylation step means less waste is generated per kilogram of product, lowering the overall cost of goods sold (COGS). The mild reaction conditions also reduce energy consumption compared to high-temperature pyrolysis methods traditionally used for triazole synthesis, further driving down operational expenses.
- Enhanced Supply Chain Reliability: By utilizing a robust catalytic system that tolerates a wide range of functional groups, manufacturers can source diverse substrates without needing custom synthesis for each variant. The stability of the reagents allows for long-term storage and just-in-time inventory management, minimizing the risk of supply disruptions. The scalability of the process from milligram to multi-kilogram scales ensures that supply can be ramped up quickly to meet market demand without requiring extensive process re-validation or equipment changes.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of benign salts and spent catalyst that can be recovered or treated easily. The absence of sulfur-containing byproducts or heavy metal contaminants (beyond the trace palladium which is removable) simplifies the environmental permitting process. This green chemistry profile aligns with increasingly stringent global environmental regulations, ensuring long-term operational continuity and reducing the risk of regulatory fines or shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for project managers evaluating the integration of this route into existing production lines or new product development pipelines.
Q: What is the advantage of using TFBen over carbon monoxide gas?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) acts as a solid carbon monoxide surrogate, eliminating the need for high-pressure CO gas equipment, thereby significantly enhancing operational safety and simplifying reactor requirements for scale-up.
Q: What is the substrate scope of this palladium-catalyzed method?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituents including alkyl groups (t-Bu, i-Pr), aryl groups (phenyl, naphthyl), and halogens (F, Br), yielding products with efficiencies up to 96%.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available reagents, operates at moderate temperatures (100°C), and avoids hazardous high-pressure gases, making it highly amenable to commercial scale-up and continuous flow processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocycle synthesis in the development of next-generation therapeutics. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant. We are committed to delivering high-purity 1,2,4-triazole-3-one intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel analogs or large-scale supply of established scaffolds, our flexible manufacturing capabilities are designed to support your specific project timelines and quality requirements.
We invite you to collaborate with us to leverage this advanced palladium-catalyzed technology for your upcoming projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your drug development timeline while optimizing your manufacturing budget. Let us be your partner in turning complex chemical challenges into commercial successes.
