Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles Using DMF as Dual Solvent and Reactant
The pharmaceutical and agrochemical industries continuously seek efficient pathways to construct nitrogen-rich heterocyclic scaffolds, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. A pivotal advancement in this domain is disclosed in patent CN114920707B, which details a novel preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift by utilizing the ubiquitous organic solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source in an iodine-promoted tandem cyclization. The significance of this scaffold cannot be overstated, as evidenced by its presence in high-value therapeutic agents such as Factor IXa inhibitors and GlyT1 inhibitors, where the trifluoromethyl group plays a critical role in modulating physicochemical properties. For R&D directors and procurement strategists, understanding this methodology offers a pathway to more sustainable and cost-effective manufacturing of complex pharmaceutical intermediates.
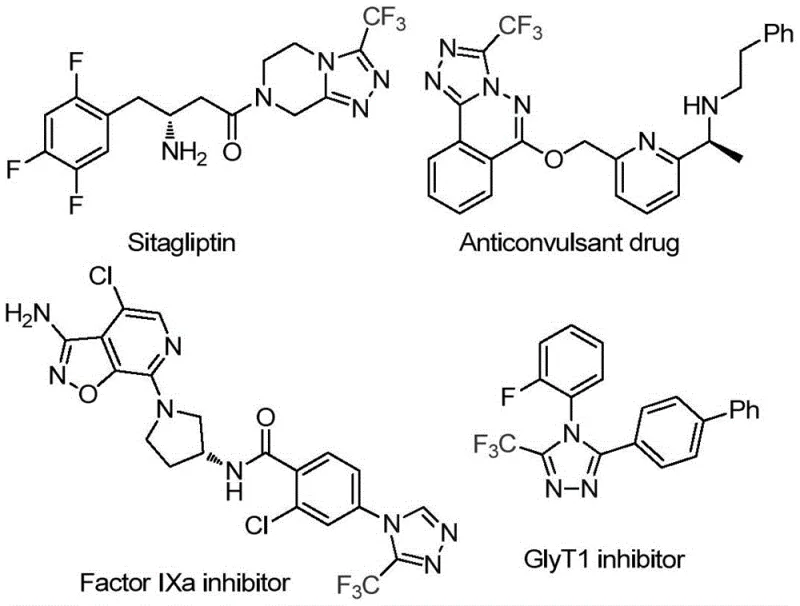
Traditional synthetic routes for constructing 1,2,4-triazole rings often suffer from significant limitations that hinder large-scale production. Conventional methods typically rely on multi-step sequences involving hazardous reagents, expensive transition metal catalysts, or strictly anhydrous and anaerobic conditions to prevent side reactions. These requirements impose a heavy burden on operational expenditure (OPEX) due to the need for specialized equipment, inert gas purging, and complex waste treatment protocols associated with heavy metal removal. Furthermore, the use of distinct carbon donors separate from the solvent system increases the stoichiometric complexity and raw material costs. In contrast, the novel approach described in the patent leverages the intrinsic reactivity of DMF, a solvent already present in vast quantities in most organic synthesis facilities. By activating DMF with molecular iodine, the process eliminates the need for external carbon sources and transition metals, thereby streamlining the reaction profile. This innovation directly addresses the pain points of process chemists who struggle with low atom economy and difficult purification steps in traditional heterocycle synthesis.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The core of this technological breakthrough lies in the unique dual-functionality of DMF under oxidative conditions facilitated by molecular iodine. Mechanistically, the reaction can proceed through two potential pathways depending on which fragment of the DMF molecule acts as the carbon donor. In the first pathway, the formyl group of DMF undergoes condensation with the trifluoroethyliminohydrazide substrate to generate a hydrazone intermediate. This is followed by an intramolecular cyclization event that eliminates a molecule of dimethylamine, yielding the desired 3-trifluoromethyl-1,2,4-triazole product. Alternatively, the N-methyl group of DMF can serve as the carbon source. In this scenario, DMF initially reacts with iodine to form an amine salt species. Subsequent elimination of hydrogen iodide and nucleophilic addition with the hydrazide generates an azadiene intermediate, which then undergoes intramolecular nucleophilic addition and oxidative aromatization. This mechanistic flexibility ensures high conversion rates across a diverse range of substrates.
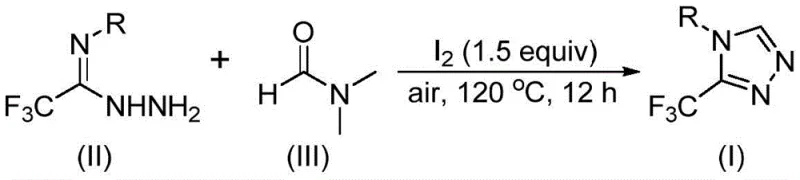
From an impurity control perspective, the use of molecular iodine as a promoter rather than a stoichiometric oxidant or catalyst offers distinct advantages. The reaction conditions are mild enough to prevent the decomposition of sensitive functional groups while being robust enough to drive the cyclization to completion within 10 to 15 hours at temperatures between 110°C and 130°C. The absence of transition metals means that the final product is free from heavy metal contamination, a critical quality attribute for pharmaceutical intermediates intended for human use. This simplifies the downstream purification process, often reducing it to simple filtration and column chromatography, thereby enhancing the overall yield and purity profile of the final API intermediate. The ability to run the reaction under air atmosphere further mitigates the risk of oxidation-sensitive byproducts that might form under harsher oxidative conditions.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The operational simplicity of this synthesis makes it highly attractive for both laboratory-scale optimization and industrial manufacturing. The procedure involves combining readily available starting materials—trifluoroethyliminohydrazide, molecular iodine, and DMF—in a standard reaction vessel. The molar ratio of trifluoroethyliminohydrazide to iodine is optimized at approximately 1:1.5, ensuring sufficient promotion of the cyclization without excessive reagent waste. The reaction mixture is heated to 120°C, a temperature easily achievable with standard heating mantles or oil baths, and stirred for 12 hours. Detailed standardized synthesis steps, including precise workup procedures and purification parameters, are outlined in the guide below to ensure reproducibility and safety compliance.
- Combine molecular iodine, trifluoroethyliminohydrazide, and organic solvent DMF in a reaction vessel under air atmosphere.
- Heat the reaction mixture to a temperature range of 110-130°C and maintain stirring for 10-15 hours to ensure complete conversion.
- Upon completion, perform post-treatment including filtration, washing, drying, and purification via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DMF-based synthesis route offers compelling economic and logistical benefits. The primary driver for cost reduction is the elimination of expensive and specialized reagents. By utilizing DMF, a commodity chemical available in bulk quantities at low prices, as both the solvent and the reactant, the process drastically reduces the bill of materials. There is no need to procure costly transition metal catalysts or specialized carbon donors, which often carry high price volatility and supply risks. Furthermore, the simplified post-treatment process reduces the consumption of silica gel and eluents during purification, leading to substantial savings in consumables and waste disposal costs. The qualitative improvement in process efficiency translates directly to a lower cost per kilogram of the final intermediate.
- Cost Reduction in Manufacturing: The integration of the solvent and reactant functions into a single molecule (DMF) fundamentally alters the cost structure of the synthesis. Traditional methods often require stoichiometric amounts of expensive formylating agents or methylating agents in addition to the solvent. By removing these extra reagents, the process achieves a higher atom economy. Additionally, the avoidance of transition metal catalysts eliminates the need for expensive scavenging resins or complex extraction protocols designed to meet strict residual metal limits (e.g., ICH Q3D guidelines). This streamlining of the chemical process results in a leaner manufacturing operation with significantly reduced variable costs.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis—iodine, DMF, and aromatic amines (precursors to the hydrazide)—are commodity chemicals with robust global supply chains. Unlike specialized catalysts that may be sourced from single suppliers with long lead times, these inputs are widely available from multiple vendors. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the reaction's tolerance to air and moisture means that storage and handling requirements for the reaction mixture are less stringent, reducing the logistical complexity and cost associated with maintaining inert atmospheres during transport and storage of intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and safety. This method operates at moderate temperatures (110-130°C) and does not involve highly exothermic steps or unstable intermediates that pose explosion risks. The use of DMF, while requiring proper handling due to its reproductive toxicity, is a well-understood solvent in the industry with established recovery and recycling protocols. The absence of heavy metals simplifies the environmental compliance landscape, as wastewater treatment does not require specialized precipitation steps for metal removal. This facilitates faster regulatory approval for new manufacturing sites and ensures long-term sustainability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the method's capabilities and limitations for potential adopters.
Q: What is the primary advantage of using DMF in this synthesis?
A: DMF serves a dual role as both the reaction solvent and the carbon source (providing either the formyl or N-methyl group), which significantly simplifies the reaction system and reduces raw material costs compared to traditional methods requiring separate carbon donors.
Q: Does this process require strict anhydrous or anaerobic conditions?
A: No, one of the key operational benefits of this patented method is that it proceeds efficiently under air atmosphere without the need for rigorous anhydrous or oxygen-free environments, greatly facilitating industrial scale-up.
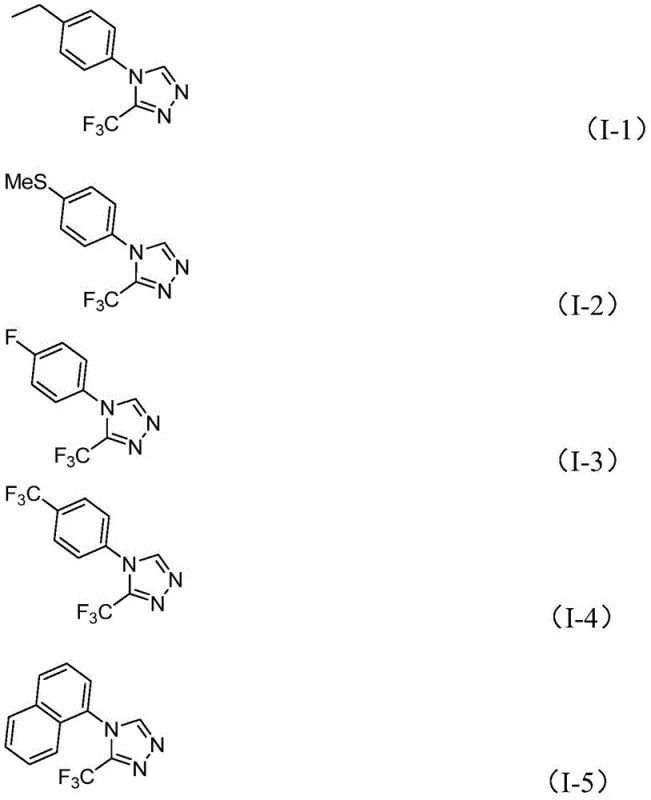
Q: What types of substituents are tolerated on the aromatic ring?
A: The method demonstrates broad substrate scope, successfully accommodating various substituents including alkyl, alkoxy, alkylthio, halogens (fluorine, chlorine), and trifluoromethyl groups at ortho, meta, or para positions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
The technological potential of using DMF as a carbon source for heterocycle synthesis represents a significant opportunity for optimizing the production of high-value pharmaceutical intermediates. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like this are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole meets the exacting standards required for downstream API synthesis. We understand the critical nature of supply continuity and quality consistency in the global pharmaceutical market.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this method can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our existing inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in driving efficiency and innovation in your supply chain.
