Advanced Resolution Technology for Axially Chiral Oxindole-Substituted Styrene Compounds
The landscape of asymmetric synthesis is continually evolving, driven by the demand for highly enantioenriched building blocks that serve as critical ligands and catalysts in modern drug discovery. Patent CN111848322B introduces a groundbreaking methodology for the preparation of axially chiral oxindole-substituted styrene compounds, addressing a significant gap in the availability of diverse chiral scaffolds. This technology leverages a kinetic resolution strategy mediated by chiral phosphoric acid catalysts, offering a robust alternative to traditional transition-metal-dependent pathways. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and operationally simple protocols that do not compromise on stereochemical fidelity. The ability to access these complex architectures through a direct resolution of racemic precursors simplifies the supply chain for high-value chiral intermediates, ensuring a reliable source of material for downstream catalytic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has relied heavily on methodologies that present significant operational and economic challenges for large-scale manufacturing. Conventional approaches often involve the nucleophilic addition to alkynes or palladium-catalyzed coupling reactions between aryl halides and alkenes, which frequently necessitate the use of expensive and toxic transition metals. These metal-catalyzed processes often require stringent exclusion of air and moisture, specialized ligands, and rigorous post-reaction purification steps to remove trace metal residues that could poison downstream biological assays or catalytic cycles. Furthermore, the substrate scope in many prior art methods is notoriously narrow, limiting the structural diversity available to medicinal chemists who need to explore various substitution patterns on the aromatic rings to optimize binding affinity. The reliance on harsh reaction conditions also poses safety risks and increases the energy footprint of the manufacturing process, making these conventional routes less attractive for cost-sensitive commercial production.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes an organocatalytic kinetic resolution that operates under remarkably mild conditions, typically between 0°C and 10°C in dichloromethane. This approach employs readily available azlactone derivatives and racemic styrene precursors, reacting them in the presence of a chiral phosphoric acid to achieve high levels of enantioselectivity. The elimination of transition metals not only reduces the cost of goods sold by removing the need for precious metal catalysts but also drastically simplifies the work-up procedure, as there is no need for extensive metal scavenging protocols. The reaction demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkoxy groups, and trifluoromethyl moieties on the aromatic rings without significant loss in yield or selectivity. This versatility allows for the rapid generation of a library of structurally diverse axially chiral compounds, facilitating faster lead optimization cycles in drug discovery programs.
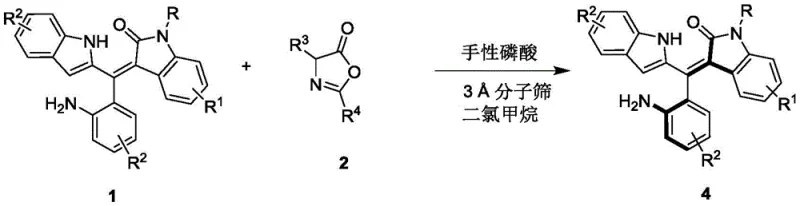
The core transformation involves the reaction of a racemic oxindole-substituted styrene derivative with an azlactone partner, mediated by a chiral Brønsted acid catalyst. As illustrated in the reaction scheme, the process effectively differentiates between the enantiomers of the starting material, converting one enantiomer into the desired product while leaving the other unreacted or transforming it into a separable byproduct. This kinetic resolution strategy is particularly powerful because it transforms a difficult asymmetric bond formation problem into a more manageable selectivity issue. The use of 3Å molecular sieves as an additive further enhances the reaction efficiency by sequestering water, which could otherwise deactivate the sensitive phosphoric acid catalyst or hydrolyze the azlactone reagent. The resulting products are obtained as yellow solids with high optical purity, ready for immediate use in subsequent synthetic transformations or biological evaluations.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
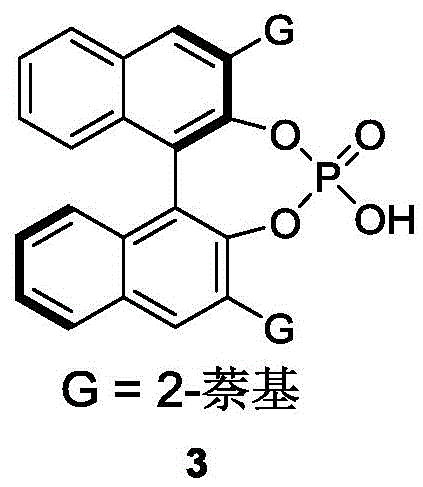
The success of this resolution protocol hinges on the precise three-dimensional arrangement of the chiral phosphoric acid catalyst, which creates a well-defined chiral pocket for the substrate binding. The catalyst, typically derived from a BINOL backbone substituted with bulky groups like 2-naphthyl, acts as a dual hydrogen-bond donor and acceptor. It activates the electrophilic azlactone through hydrogen bonding with the carbonyl oxygen while simultaneously orienting the nucleophilic styrene substrate via interactions with the indole nitrogen or other polar functionalities. This dual activation mode lowers the activation energy for the reaction of one specific enantiomer of the racemic mixture while sterically hindering the approach of the other, thereby enforcing high enantioselectivity. The rigidity of the catalyst scaffold ensures that the chiral information is effectively transferred to the product, resulting in the formation of the axially chiral bond with a defined configuration. Understanding this mechanistic nuance is crucial for process optimization, as slight modifications to the catalyst structure or reaction temperature can fine-tune the balance between reaction rate and stereoselectivity.
Impurity control in this process is inherently managed by the high specificity of the organocatalyst. Unlike radical-based or metal-catalyzed processes that might generate a plethora of side products through non-selective pathways, this ionic mechanism is highly directed. The primary impurities are typically the unreacted starting materials or minor diastereomers, which are easily removed via standard silica gel column chromatography. The patent data indicates that the reaction proceeds with high atom economy, minimizing the generation of waste streams associated with stoichiometric chiral auxiliaries. Furthermore, the stability of the axially chiral bond in the product is ensured by the steric bulk of the ortho-substituents on the biaryl system, preventing racemization during isolation and storage. This robustness is essential for supply chain reliability, as it guarantees that the high enantiomeric excess achieved at the reactor scale is maintained until the material reaches the customer's laboratory.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
The synthesis of these high-value chiral intermediates is designed to be scalable and reproducible, utilizing standard laboratory equipment and commercially available reagents. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent moisture ingress, although the use of molecular sieves provides a buffer against minor humidity fluctuations. The careful control of temperature is paramount; maintaining the reaction at 0°C ensures optimal selectivity, preventing thermal erosion of the chiral induction provided by the phosphoric acid. Following the reaction completion, the removal of the solid additives and solvent evaporation yields a crude residue that is highly amenable to purification. The detailed standardized synthesis steps below outline the precise molar ratios, solvent volumes, and purification techniques required to replicate the high yields and ee values reported in the patent literature.
- Prepare the reaction mixture by combining the racemic styrene precursor (Compound 1) and the azlactone derivative (Compound 2) in dichloromethane solvent with 3Å molecular sieves.
- Add the chiral phosphoric acid catalyst (preferably a BINOL derivative like Compound 3) at a loading of 10 mol% and maintain the temperature between 0°C and 10°C.
- Stir the reaction until TLC indicates completion, then filter off the molecular sieves, concentrate the filtrate, and purify via silica gel column chromatography to isolate the high-enantiopurity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic resolution technology offers substantial strategic benefits beyond mere technical performance. The shift away from precious metal catalysts directly translates to significant cost reductions in raw material procurement, as chiral phosphoric acids, while specialized, are used in catalytic amounts and do not carry the volatile price tags of palladium or rhodium complexes. Moreover, the simplified downstream processing eliminates the need for expensive metal scavenger resins and the associated validation testing for heavy metal limits, streamlining the quality control workflow. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to a reduced carbon footprint and aligning with corporate sustainability goals. These factors combined create a more resilient supply chain that is less susceptible to geopolitical disruptions affecting the supply of critical transition metals.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver and simplifies the purification process, leading to substantial savings in both material and operational expenses. By avoiding the use of stoichiometric chiral reagents and expensive metal scavengers, the overall cost of goods is significantly optimized, making the final chiral intermediate more competitive in the global market. The high yield and selectivity further reduce waste disposal costs, enhancing the overall economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The reliance on robust organocatalysts and readily available starting materials ensures a stable and continuous supply of critical intermediates. Unlike processes dependent on scarce metals, this methodology mitigates the risk of supply shortages and price volatility, guaranteeing consistent delivery schedules for downstream customers. The operational simplicity of the reaction also reduces the likelihood of batch failures due to equipment sensitivity, further securing the supply continuity for long-term projects.
- Scalability and Environmental Compliance: The mild conditions and absence of toxic heavy metals make this process highly scalable from gram to multi-kilogram quantities without significant re-engineering. The reduced environmental burden facilitates easier regulatory compliance and permits acquisition for manufacturing facilities, accelerating the time-to-market for new drug candidates. The green chemistry profile of this method aligns with increasingly stringent environmental regulations, future-proofing the production process against evolving legislative standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and scope of protection defined in the patent, providing clarity on the practical aspects of adopting this methodology. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their existing pipelines.
Q: What are the primary advantages of this resolution method over traditional metal-catalyzed couplings?
A: Unlike traditional methods relying on palladium or titanium catalysts which often require harsh conditions and generate heavy metal waste, this organocatalytic approach operates at mild temperatures (0-10°C) using metal-free chiral phosphoric acids, significantly simplifying purification and reducing environmental impact.
Q: What level of enantiomeric excess (ee) can be achieved with this process?
A: The patented methodology demonstrates exceptional stereocontrol, consistently achieving enantiomeric excess values up to 96% across a broad range of substrates, ensuring the production of high-purity chiral intermediates suitable for sensitive pharmaceutical applications.
Q: Can the resolved styrene compounds be further functionalized?
A: Yes, the resolved axially chiral styrenes serve as versatile precursors; they can be efficiently derivatized into complex chiral tertiary amine catalysts, which have been proven effective in subsequent asymmetric [4+2] cyclization reactions with high yields and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results observed in the laboratory can be seamlessly translated to industrial manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to verify the enantiomeric excess of every batch we produce. Our commitment to quality assurance means that you receive material that is not only chemically pure but also stereochemically defined, ready for immediate use in your most demanding catalytic applications.
We invite you to collaborate with us to leverage this advanced resolution technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can optimize your budget. Please contact us to request specific COA data for our catalog of chiral styrenes or to discuss route feasibility assessments for your proprietary targets. Together, we can accelerate your drug discovery timeline with reliable, high-quality chemical solutions.
