Advanced Resolution Technology for Axially Chiral Oxindole-Substituted Styrenes in Pharmaceutical Manufacturing
The landscape of asymmetric synthesis is continually evolving, driven by the pharmaceutical industry's relentless demand for high-purity chiral intermediates. A significant breakthrough in this domain is detailed in patent CN111848322B, which discloses a novel class of axially chiral oxindole-substituted styrene compounds and an efficient resolution method for their production. These compounds are not merely structural curiosities; they represent a critical class of ligands and organocatalysts capable of inducing high stereoselectivity in complex organic transformations. The patent outlines a robust kinetic resolution strategy utilizing chiral phosphoric acid catalysts, marking a departure from traditional metal-dependent methodologies. This technological advancement offers a streamlined pathway to access structurally diverse and complex chiral scaffolds that were previously difficult to synthesize with high optical purity. For R&D teams focused on developing next-generation therapeutics, the ability to access these specific axially chiral motifs opens new avenues for drug design and catalyst development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has relied heavily on transition metal catalysis or harsh nucleophilic additions. Literature precedents often describe the use of palladium-catalyzed coupling reactions between aryl halides and hydrazones, or titanium-mediated reactions involving aldehydes and organozinc reagents. While effective in specific contexts, these conventional approaches suffer from significant drawbacks that hinder their widespread industrial adoption. The reliance on precious metals introduces stringent regulatory hurdles regarding residual metal limits in final API products, necessitating costly and time-consuming purification steps. Furthermore, many of these metal-catalyzed processes require elevated temperatures or strictly anhydrous conditions that are not always compatible with sensitive functional groups found in advanced intermediates. The substrate scope is often narrow, limiting the structural diversity of the accessible chiral library, and the atom economy can be poor due to the generation of stoichiometric metal waste.
The Novel Approach
In stark contrast, the methodology presented in the patent utilizes an organocatalytic kinetic resolution strategy that operates under remarkably mild conditions. By employing a chiral phosphoric acid catalyst in dichloromethane at low temperatures (0°C to 10°C), the process achieves high enantioselectivity without the need for toxic heavy metals. This approach leverages the subtle steric and electronic interactions between the catalyst and the racemic substrate to differentiate enantiomers effectively. The reaction tolerates a wide variety of substituents on the aromatic rings, including halogens, alkoxy groups, and trifluoromethyl moieties, demonstrating exceptional versatility. The operational simplicity is another key advantage; the reaction proceeds to completion with straightforward monitoring via TLC, and the workup involves simple filtration and concentration. This shift from metal catalysis to organocatalysis represents a paradigm shift towards greener, more sustainable, and cost-effective manufacturing of high-value chiral intermediates.
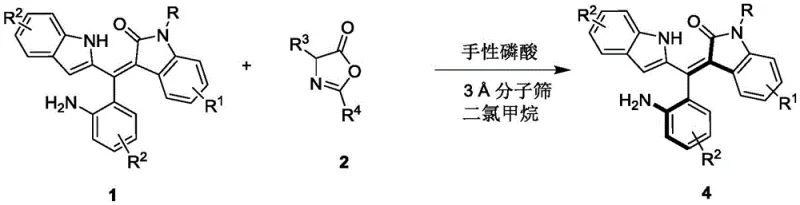
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
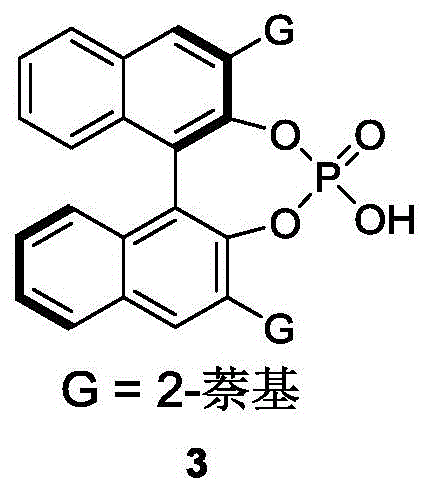
The core of this technological innovation lies in the precise molecular recognition facilitated by the chiral phosphoric acid catalyst. These catalysts, typically derived from BINOL or SPINOL backbones, function as Brønsted acids that activate the electrophilic species through dual hydrogen bonding interactions. In the context of this resolution, the catalyst likely forms a tight ion pair or hydrogen-bonded complex with the reacting species, creating a highly organized chiral environment. This confined space imposes severe steric constraints on the approaching nucleophile, effectively blocking one face of the prochiral center or differentiating between the enantiomers of the racemic starting material. The specific geometry of the catalyst, particularly the bulky substituents at the 3,3'-positions of the binaphthyl skeleton, plays a pivotal role in shielding one trajectory of the reaction, thereby enforcing the formation of a single enantiomer with high fidelity.
Furthermore, the choice of the specific catalyst structure is critical for optimizing the enantiomeric excess. The patent highlights the efficacy of a specific BINOL-derived phosphoric acid (Formula 3) bearing 2-naphthyl groups. These extended aromatic systems enhance the pi-stacking interactions with the substrate, further stabilizing the transition state leading to the desired product. The mechanism avoids the formation of stable metal-ligand complexes that can sometimes lead to catalyst deactivation or product inhibition. Instead, the dynamic nature of the hydrogen bonding allows for rapid turnover while maintaining strict stereochemical control. This mechanistic understanding is vital for process chemists aiming to scale the reaction, as it suggests that maintaining strict temperature control and solvent dryness is essential to preserve the integrity of the hydrogen-bonding network that drives the enantioselectivity.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
The synthesis protocol described in the patent is designed for reproducibility and scalability, making it highly attractive for process development teams. The procedure begins with the preparation of the reaction vessel containing activated 3Å molecular sieves to ensure anhydrous conditions, which are crucial for the performance of the phosphoric acid catalyst. The racemic styrene precursor and the oxazolone coupling partner are dissolved in dichloromethane, and the mixture is cooled to 0°C before the addition of the catalyst. This low-temperature initiation is a critical parameter that suppresses background non-catalyzed reactions which could erode enantiopurity. Following the reaction period, typically around 5 hours, the mixture is filtered to remove the desiccant, and the crude product is isolated via standard silica gel chromatography. For a detailed, step-by-step standardized operating procedure including exact molar ratios and purification gradients, please refer to the technical guide below.
- Prepare the reaction mixture by combining the racemic styrene precursor (Formula 1) and the oxazolone reactant (Formula 2) in dichloromethane solvent with molecular sieves.
- Add the chiral phosphoric acid catalyst (Formula 3) at a loading of 10 mol% and maintain the reaction temperature strictly at 0°C to ensure optimal enantioselectivity.
- Monitor reaction progress via TLC, then filter off the molecular sieves, concentrate the filtrate, and purify the resulting axially chiral product (Formula 4) using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this resolution technology offers compelling advantages that directly address the pain points of procurement and supply chain management in the fine chemical sector. The elimination of transition metals such as palladium and titanium removes a significant cost driver associated with both raw material acquisition and waste disposal. Heavy metal scavengers and specialized filtration equipment are no longer required, leading to a drastically simplified downstream processing workflow. This simplification translates into reduced cycle times and lower operational expenditures, allowing for more competitive pricing of the final chiral intermediate. Additionally, the use of commodity solvents like dichloromethane and readily available starting materials ensures a robust and resilient supply chain that is less susceptible to geopolitical fluctuations affecting rare earth or precious metal markets.
- Cost Reduction in Manufacturing: The transition to an organocatalytic system fundamentally alters the cost structure of producing these complex chiral molecules. By removing the dependency on expensive noble metal catalysts, the direct material costs are significantly lowered. Moreover, the mild reaction conditions (0°C) reduce the energy consumption required for heating or cryogenic cooling compared to processes requiring extreme temperatures. The high atom economy of the addition reaction minimizes waste generation, further reducing the costs associated with environmental compliance and waste treatment. These factors combine to create a manufacturing process that is inherently more economical, allowing for substantial cost savings that can be passed down the supply chain or reinvested into further R&D efforts.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents enhances the overall reliability of the supply chain. Unlike specialized metal catalysts that may have long lead times or single-source suppliers, the chiral phosphoric acids and organic substrates used in this process are accessible from multiple global vendors. This diversification of the supplier base mitigates the risk of production stoppages due to raw material shortages. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, ensuring consistent batch-to-batch quality. This stability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of large-scale pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with simple unit operations that translate easily from gram-scale laboratory synthesis to multi-kilogram pilot plant production. The absence of toxic heavy metals simplifies the regulatory approval process for new drug applications, as the burden of proving low residual metal levels is removed. This aligns perfectly with the industry's growing emphasis on green chemistry and sustainability. The reduced environmental footprint, characterized by lower energy usage and simpler waste streams, positions this technology as a future-proof solution for manufacturing high-purity pharmaceutical intermediates in an increasingly regulated global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing a clear picture of the method's capabilities and limitations. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What is the primary advantage of this resolution method over traditional metal-catalyzed couplings?
A: This organocatalytic method eliminates the need for expensive and toxic transition metals like palladium or titanium, significantly reducing heavy metal impurity risks and simplifying downstream purification for pharmaceutical applications.
Q: What level of enantiomeric excess (ee) can be achieved with this protocol?
A: The patented process demonstrates exceptional stereocontrol, consistently achieving enantiomeric excess values up to 96% across a broad range of substrate derivatives, ensuring high optical purity for chiral drug synthesis.
Q: Can the resulting chiral styrene compounds be further derivatized?
A: Yes, the resolved axially chiral styrenes serve as versatile precursors that can be chemically modified into chiral tertiary amine catalysts, which have been proven effective in subsequent asymmetric [4+2] cyclization reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing high-quality chiral building blocks for the development of innovative therapeutics. Our team of expert process chemists has thoroughly analyzed this patented resolution technology and is fully equipped to execute the synthesis of these complex axially chiral oxindole-substituted styrenes at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the high enantiomeric excess values promised by this novel method.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project requirements. Whether you need custom synthesis of specific derivatives or large-scale production of the core scaffold, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume needs. We are prepared to provide specific COA data from our pilot batches and comprehensive route feasibility assessments to demonstrate how we can optimize your supply chain for these critical pharmaceutical intermediates.
