Advanced Rhodium-Catalyzed Synthesis of Indolo[3,2-c]quinoline Compounds for Commercial Scale-Up
Advanced Rhodium-Catalyzed Synthesis of Indolo[3,2-c]quinoline Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex nitrogen-containing heterocycles, particularly those with significant biological potential. A groundbreaking development in this domain is detailed in patent CN110183443B, which discloses a novel synthesis method for indolo[3,2-c]quinoline compounds. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-pot oxidative coupling process. By utilizing a transition metal catalyst, specifically rhodium or indium, in a protic solvent under an oxygen-containing atmosphere, this method enables the direct series reaction between two molecules of 2-alkynyl aniline. The significance of this innovation lies not only in its operational simplicity but also in its ability to bypass the generation of hazardous halogenated byproducts, thereby aligning with modern green chemistry principles while delivering high-purity intermediates essential for drug discovery and development pipelines.
![General reaction scheme showing the conversion of 2-alkynyl aniline to indolo[3,2-c]quinoline via Rhodium catalysis](/insights/img/indolo-quinoline-synthesis-rhodium-pharma-supplier-20260303145958-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolo[3,2-c]quinoline skeleton has relied heavily on methodologies that necessitate the use of halogenated starting materials and reagents. These conventional routes often suffer from inherent inefficiencies, including poor atom economy and the generation of substantial quantities of chemical waste. The reliance on halogenated precursors not only increases the raw material costs due to the complexity of synthesizing these specific building blocks but also introduces significant downstream processing challenges. Separation and purification steps become arduous as chemists must remove residual halogens and associated byproducts to meet the stringent purity specifications required for pharmaceutical applications. Furthermore, the environmental footprint of these legacy processes is considerable, involving the disposal of toxic halogenated waste streams, which contradicts the increasing regulatory pressure for sustainable manufacturing practices in the global chemical supply chain.
The Novel Approach
In stark contrast to these legacy limitations, the novel approach described in the patent data utilizes a direct oxidative dimerization strategy that fundamentally simplifies the synthetic architecture. By employing readily available 2-alkynyl aniline compounds as the sole organic starting materials, the process achieves the formation of the target heterocycle in a single reaction vessel. The use of an oxygen-containing atmosphere serves as a benign and cost-effective oxidant, replacing expensive and hazardous chemical oxidants often found in older literature. This methodology not only streamlines the workflow by eliminating intermediate isolation steps but also drastically reduces the overall resource consumption. The reaction proceeds under relatively mild thermal conditions, typically around 120°C, which enhances energy efficiency and minimizes the risk of thermal degradation of sensitive functional groups, making it highly suitable for the production of diverse derivative libraries for medicinal chemistry optimization.
Mechanistic Insights into Rhodium-Catalyzed Oxidative Cyclization
Understanding the catalytic cycle is paramount for R&D directors aiming to optimize this process for specific API intermediates. The reaction initiates with the coordination of the Rhodium(III) catalyst to both the alkyne and amino groups of the 2-alkynyl aniline substrate, forming a key organometallic intermediate. This activation facilitates an intermolecular nucleophilic addition where a second molecule of the aniline attacks the activated alkyne, leading to the formation of an enamine intermediate. The presence of hexafluoroisopropanol (HFIP) as the solvent plays a dual role: it acts as a proton source to facilitate protonation steps and stabilizes charged intermediates through hydrogen bonding interactions. Subsequent intramolecular nucleophilic addition to the Rh-activated alkyne bond drives the cyclization forward, eventually liberating the Rhodium catalyst to re-enter the cycle. This elegant mechanism ensures high turnover numbers and maintains catalytic efficiency throughout the prolonged reaction times required for complete conversion.
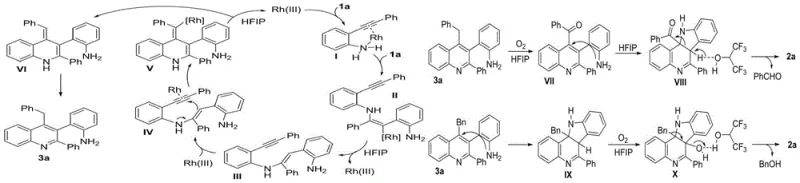
The final aromatization step is critically dependent on the oxidative environment provided by the air or oxygen atmosphere. Mechanistic studies indicate that the intermediate dihydro-species undergoes oxidation to form a ketone or imine species, which then participates in further intramolecular nucleophilic additions. This sequence ultimately leads to the loss of small molecules like benzaldehyde or benzyl alcohol, driven by the thermodynamic stability of the fully aromatic indolo[3,2-c]quinoline system. The detection of molecular ion peaks corresponding to benzaldehyde and benzyl alcohol via GC-MS analysis confirms this oxidative cleavage pathway. For quality control teams, understanding this mechanism is vital for impurity profiling, as incomplete oxidation could lead to the accumulation of partially reduced intermediates. However, the robustness of the Rhodium catalyst system, combined with the oxidative power of molecular oxygen, ensures that the reaction proceeds cleanly to the desired aromatic product with minimal side reactions, provided the oxygen supply is maintained.
How to Synthesize Indolo[3,2-c]quinoline Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and purity. The standard protocol involves charging a reaction vessel with the 2-alkynyl aniline substrate and the Rhodium catalyst, typically [RhCp*Cl2]2, at a molar ratio ranging from 1:0.025 to 1:0.1. Hexafluoroisopropanol is the preferred solvent due to its unique ability to promote the reaction kinetics, although other protic solvents like methanol or trifluoroethanol can be used with varying degrees of success. The reaction mixture must be sealed under an air atmosphere to ensure a continuous supply of oxygen, which is non-negotiable for the success of the oxidative cyclization. Heating the mixture to 120°C for approximately 20 hours allows the reaction to reach completion, after which standard workup procedures involving aqueous extraction and silica gel chromatography yield the pure product. The detailed standardized synthesis steps are outlined in the guide below.
- Prepare the reaction mixture by combining 2-alkynyl aniline substrate with a Rhodium catalyst such as [RhCp*Cl2]2 in a protic solvent like hexafluoroisopropanol.
- Seal the reaction vessel under an oxygen-containing atmosphere, such as air or pure oxygen, to facilitate the oxidative cyclization process.
- Heat the mixture to a temperature between 70°C and 150°C, preferably 120°C, and stir for approximately 20 hours to achieve high conversion yields.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of halogenated reagents translates directly into a simplified supply chain, as the sourcing of specialized halogenated building blocks is no longer necessary. This reduction in raw material complexity mitigates the risk of supply disruptions often associated with niche chemical intermediates. Furthermore, the one-pot nature of the reaction significantly reduces the number of unit operations required, leading to lower labor costs and reduced equipment occupancy time. The use of air as the oxidant is perhaps the most significant cost-saving factor, removing the need for purchasing, storing, and handling expensive stoichiometric oxidants. These factors collectively contribute to a more resilient and cost-effective manufacturing process that can better withstand market volatility.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the drastic simplification of the synthetic route and the use of commodity chemicals. By avoiding the use of expensive halogenated starting materials and stoichiometric oxidants, the direct material costs are significantly lowered. Additionally, the high atom economy of the dimerization reaction means that a larger proportion of the input mass is converted into the final product, reducing waste disposal costs. The ability to use air as the oxidant further eliminates a major cost center associated with chemical oxidants. Consequently, the overall cost of goods sold (COGS) for the indolo[3,2-c]quinoline intermediate is optimized, allowing for more competitive pricing in the global marketplace without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on widely available 2-alkynyl aniline precursors rather than specialized halogenated derivatives. These starting materials are often easier to source from multiple vendors, reducing dependency on single-source suppliers. The robustness of the reaction conditions, which tolerate a wide range of substituents including electron-donating and electron-withdrawing groups, ensures that variations in raw material quality do not critically impact the process outcome. This flexibility allows procurement teams to negotiate better terms and secure long-term contracts with greater confidence. Moreover, the simplified process flow reduces the likelihood of batch failures due to complex multi-step handling, ensuring a steady and reliable flow of intermediates to downstream API manufacturing facilities.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is facilitated by the mild reaction conditions and the absence of hazardous reagents. The use of oxygen from air eliminates the safety risks associated with handling strong chemical oxidants on a large scale, making the process inherently safer for plant operators. From an environmental perspective, the reduction in halogenated waste streams aligns with increasingly stringent global environmental regulations, minimizing the burden on wastewater treatment facilities. The high selectivity of the reaction reduces the formation of difficult-to-separate byproducts, simplifying the purification process and reducing solvent consumption during workup. These attributes make the technology highly attractive for companies aiming to enhance their sustainability profiles while expanding production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages and operational parameters defined by the intellectual property, ensuring that stakeholders have accurate information for decision-making processes regarding technology transfer and process validation.
Q: What are the primary advantages of this synthesis method over traditional halogenated routes?
A: This method eliminates the need for halogenated starting materials and reagents, significantly improving atom economy and reducing environmental pollution associated with halogen waste disposal.
Q: Is the reaction sensitive to atmospheric conditions?
A: Yes, the presence of an oxygen-containing atmosphere is critical for the oxidative aromatization step; inert atmospheres lead to different intermediates and lower yields of the target quinoline.
Q: What types of substituents are tolerated on the substrate?
A: The process demonstrates wide substrate applicability, tolerating various groups including alkyl, alkoxy, halogens, and trifluoromethyl groups on both the aniline and alkyne moieties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolo[3,2-c]quinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the Rhodium-mediated oxidative coupling described in CN110183443B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of transition metal catalysis, including rigorous containment protocols and advanced purification capabilities. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of indolo[3,2-c]quinoline intermediate meets the highest industry standards for pharmaceutical applications. Our commitment to quality and consistency makes us the ideal partner for securing your supply chain against technical and logistical uncertainties.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient methodology. We encourage potential partners to request specific COA data and route feasibility assessments to validate the performance of this technology against your current benchmarks. Together, we can accelerate the development of next-generation therapeutics by ensuring a reliable, high-quality supply of critical heterocyclic intermediates.
