Advanced Synthesis of Axially Chiral Oxindole-Substituted Styrenes for Commercial Catalysis Applications
The landscape of asymmetric synthesis is continuously evolving, driven by the urgent demand for high-purity chiral building blocks that serve as the foundation for modern therapeutic agents. A significant breakthrough in this domain is documented in patent CN111848322B, which discloses a novel class of axially chiral oxindole-substituted styrene compounds and an efficient resolution method for their preparation. This technology addresses a critical gap in the availability of robust chiral ligands and catalysts, offering a pathway to structurally diverse molecules that were previously difficult to access with high stereochemical fidelity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this patented methodology is essential for securing a competitive edge in drug development pipelines. The innovation lies not merely in the final structure but in the elegant simplicity of the catalytic system that enables the construction of the axially chiral skeleton with exceptional control.
Furthermore, the implications of this technology extend beyond academic curiosity, presenting tangible opportunities for cost reduction in pharmaceutical intermediate manufacturing. By leveraging organocatalytic strategies rather than relying on scarce precious metals, the process inherently reduces raw material volatility and supply chain risks associated with metal mining and refining. The ability to generate these complex scaffolds under mild conditions also suggests a lower energy footprint and reduced safety hazards during production, aligning with the increasing global emphasis on green chemistry principles. As we delve deeper into the technical specifics, it becomes clear that this patent represents a versatile platform technology capable of supporting the commercial scale-up of complex polymer additives and fine chemicals alike, ensuring supply continuity for high-value applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has been fraught with significant challenges that hinder widespread industrial adoption and limit the diversity of accessible chemical space. Traditional methodologies often rely heavily on transition metal catalysis, such as palladium or titanium complexes, which introduce substantial complications regarding metal residue removal and regulatory compliance in pharmaceutical contexts. These metal-catalyzed processes frequently require harsh reaction conditions, including elevated temperatures or stringent anhydrous environments, which can degrade sensitive functional groups and lead to poor atom economy. Moreover, the reliance on nucleophilic addition reactions or coupling of aryl halides with alkenes often results in limited substrate scope, restricting the ability to introduce diverse substituents necessary for optimizing biological activity in drug candidates. The scarcity of efficient resolution methods for racemic mixtures further exacerbates the problem, forcing manufacturers to rely on inefficient separation techniques that drastically reduce overall yield and increase production costs.
The Novel Approach
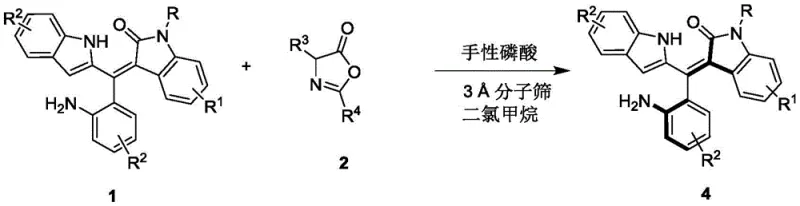
In stark contrast to these legacy limitations, the methodology outlined in the referenced patent introduces a paradigm shift through the application of chiral phosphoric acid catalysis for the kinetic resolution of racemic precursors. This organocatalytic approach operates under remarkably mild conditions, typically between 0°C and 10°C, utilizing dichloromethane as a solvent and molecular sieves to maintain anhydrous conditions without the need for extreme measures. The use of a binaphthyl-derived chiral phosphoric acid catalyst enables precise stereocontrol, achieving enantiomeric excess values as high as 96% ee, which is critical for ensuring the efficacy and safety of downstream pharmaceutical products. This novel strategy not only broadens the substrate scope to include a wide variety of substituted indoles and oxazolones but also simplifies the purification process, as the absence of heavy metals eliminates the need for costly scavenging steps. Consequently, this approach offers a streamlined, cost-effective route to high-purity OLED material precursors and agrochemical intermediates that was previously unavailable.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed Resolution
The core of this technological advancement lies in the sophisticated interaction between the chiral phosphoric acid catalyst and the prochiral or racemic substrates, which dictates the stereochemical outcome of the reaction. The catalyst, typically a BINOL-derived phosphoric acid with bulky substituents at the 3,3'-positions, creates a well-defined chiral pocket that differentiates between the enantiomers of the starting material through a network of hydrogen bonding interactions. During the reaction, the acidic proton of the catalyst activates the electrophilic species, likely the oxazolone derivative, while the basic phosphoryl oxygen may simultaneously coordinate with the nucleophile, facilitating a highly organized transition state. This dual activation mode ensures that the reaction proceeds through a specific trajectory that favors the formation of one axial atropisomer over the other, effectively resolving the racemic mixture into its constituent enantiomers with high fidelity. The presence of 3A molecular sieves plays a crucial role in this mechanism by sequestering water generated during the condensation process, thereby driving the equilibrium towards product formation and preventing catalyst deactivation.
Beyond the primary catalytic cycle, the mechanism also incorporates robust impurity control features that are vital for maintaining the integrity of the final product in a commercial setting. The mild reaction temperature of 0°C suppresses side reactions such as polymerization or decomposition of the sensitive styrene double bond, which are common pitfalls in conventional high-temperature syntheses. Furthermore, the specific choice of solvent and the stoichiometric ratio of reactants (1:0.5 to 1:0.6) are optimized to minimize the formation of byproducts, ensuring that the crude reaction mixture is clean and amenable to straightforward purification. This level of control over the impurity profile is essential for meeting the stringent purity specifications required by regulatory bodies for active pharmaceutical ingredients and high-value fine chemicals. By understanding these mechanistic details, process chemists can better optimize the reaction parameters for large-scale production, ensuring consistent quality and reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to a precise sequence of steps that maximize yield and enantioselectivity while maintaining safety standards. The process begins with the careful preparation of the reaction vessel, ensuring it is dry and inert, followed by the addition of the specific molar ratios of the indole-based substrate and the oxazolone coupling partner in dichloromethane. The detailed standardized synthesis steps see the guide below, which outlines the exact addition rates, temperature ramping profiles, and workup procedures necessary to replicate the high performance reported in the patent literature. Adhering to these protocols is critical for achieving the reported 96% ee and ensuring that the resulting chiral tertiary amine catalysts derived from these intermediates perform optimally in downstream asymmetric transformations.
- Prepare the reaction mixture by combining Formula 1 and Formula 2 compounds in dichloromethane solvent with 3A molecular sieves.
- Add the chiral phosphoric acid catalyst (10 mol%) and maintain the reaction temperature between 0°C and 10°C while stirring.
- Monitor reaction progress via TLC, then filter, concentrate, and purify the resulting Formula 4 compound using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this organocatalytic resolution method offers profound advantages that directly impact the bottom line and operational resilience of chemical manufacturing enterprises. By eliminating the dependence on expensive transition metal catalysts such as palladium or titanium, companies can significantly reduce raw material costs and mitigate the risks associated with the volatile pricing of precious metals. The simplified workup procedure, which avoids complex metal scavenging steps, translates into shorter processing times and lower consumption of auxiliary materials, contributing to substantial cost savings in the overall manufacturing process. Additionally, the mild reaction conditions enhance operational safety by reducing the risk of thermal runaways and pressure buildups, thereby lowering insurance premiums and minimizing downtime related to safety incidents. These factors collectively enhance supply chain reliability, ensuring a steady flow of high-quality intermediates even in fluctuating market conditions.
- Cost Reduction in Manufacturing: The transition from metal-catalyzed to organocatalytic processes fundamentally alters the cost structure of producing axially chiral compounds by removing the need for costly metal salts and ligands. This shift not only lowers the direct material cost but also reduces the environmental burden associated with metal waste disposal, leading to further indirect savings through reduced regulatory compliance costs. The high atom economy of the reaction ensures that a greater proportion of the starting materials are converted into the desired product, minimizing waste generation and maximizing resource efficiency. Furthermore, the ability to recover and potentially recycle the chiral phosphoric acid catalyst adds another layer of economic benefit, making the process increasingly cost-effective as production volumes scale up.
- Enhanced Supply Chain Reliability: Utilizing commercially available and stable starting materials ensures that the supply chain is not vulnerable to the geopolitical and logistical disruptions that often affect the sourcing of specialized metal catalysts. The robustness of the reaction conditions means that production can be maintained consistently across different manufacturing sites without the need for highly specialized equipment or extreme environmental controls. This flexibility allows for a more agile supply chain capable of responding quickly to changes in demand, reducing lead time for high-purity pharmaceutical intermediates and ensuring that downstream drug development projects remain on schedule. The stability of the intermediates also facilitates easier storage and transportation, further enhancing the reliability of the supply network.
- Scalability and Environmental Compliance: The simplicity of the reaction setup and workup makes this process highly amenable to scale-up from gram to ton quantities without significant re-engineering of the process parameters. The use of common solvents and the absence of toxic heavy metals simplify waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations globally. This environmental compatibility not only protects the company from potential fines and reputational damage but also aligns with the sustainability goals of major pharmaceutical partners who prioritize green chemistry in their supply chains. The scalability ensures that the technology can support the commercial scale-up of complex polymer additives and fine chemicals as market demand grows.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this axially chiral oxindole-substituted styrene synthesis technology. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the practical aspects of adopting this method. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this chemistry into their existing production workflows or R&D pipelines.
Q: What represents the primary advantage of this resolution method over traditional metal-catalyzed approaches?
A: The primary advantage lies in the utilization of organocatalysis via chiral phosphoric acids, which eliminates the need for expensive and toxic transition metal catalysts like palladium or titanium, thereby simplifying downstream purification and reducing heavy metal residue risks in pharmaceutical intermediates.
Q: How does the process ensure high enantiomeric excess (ee) for sensitive substrates?
A: High enantiomeric excess is achieved through the precise steric environment provided by the binaphthyl-derived chiral phosphoric acid catalyst, which facilitates highly selective kinetic resolution under mild cryogenic conditions ranging from 0°C to 10°C.
Q: Is this synthetic route scalable for industrial production of chiral tertiary amine catalysts?
A: Yes, the route is highly scalable due to the use of common solvents like dichloromethane, commercially available starting materials, and straightforward workup procedures involving filtration and standard column chromatography, making it suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technologies described in patent CN111848322B and are uniquely positioned to bring these advanced chiral intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of axially chiral oxindole-substituted styrene meets the exacting standards required for pharmaceutical and fine chemical applications. Our commitment to quality and consistency makes us a trusted partner for companies seeking to leverage this innovative chemistry for their next-generation products.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how switching to this organocatalytic route can optimize your budget and improve your process efficiency. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this technology for your projects. Let us collaborate to drive innovation and efficiency in your supply chain, ensuring you stay ahead in the competitive landscape of fine chemical manufacturing.
