Advanced Resolution of Axially Chiral Oxindole Styrenes for High-Purity Pharmaceutical Intermediates
The landscape of asymmetric synthesis is continually evolving, driven by the urgent need for efficient methods to construct complex chiral scaffolds essential for modern drug discovery. Patent CN111848322B introduces a groundbreaking approach to the resolution of axially chiral oxindole-substituted styrene compounds, addressing a significant gap in the availability of high-purity chiral ligands and catalysts. This technology leverages a sophisticated organocatalytic strategy that bypasses the limitations of traditional transition metal methodologies, offering a robust pathway to structurally diverse intermediates. For R&D directors and process chemists, this represents a pivotal shift towards more sustainable and operationally simple synthetic routes that maintain rigorous stereochemical control without the burden of heavy metal contamination.
Furthermore, the versatility of this resolution method extends beyond mere isolation; the resulting axially chiral compounds serve as potent precursors for next-generation organocatalysts. The patent details a seamless derivatization process that transforms these resolved styrenes into highly active chiral tertiary amine catalysts, capable of driving complex asymmetric transformations with remarkable efficiency. This dual utility—acting both as a high-value intermediate and a catalyst precursor—significantly enhances the commercial viability of the process, making it an attractive option for supply chain managers looking to consolidate sourcing strategies for specialized fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has relied heavily on narrow and often cumbersome synthetic pathways that pose significant challenges for scale-up and purity control. Conventional literature describes methods primarily centered on the nucleophilic addition of alkynes or the coupling of aryl halides with alkenes, frequently requiring expensive transition metal catalysts such as palladium or titanium complexes. These metal-mediated processes often demand stringent anhydrous conditions, elevated temperatures, and complex workup procedures to remove trace metal residues, which is a critical bottleneck in pharmaceutical manufacturing where regulatory limits on heavy metals are exceedingly strict. Additionally, the substrate scope for these traditional methods is frequently limited, restricting the structural diversity accessible to medicinal chemists exploring new chemical space for drug candidates.
The Novel Approach
In stark contrast, the methodology disclosed in CN111848322B utilizes a chiral phosphoric acid-catalyzed resolution strategy that operates under exceptionally mild and practical conditions. By employing a binaphthyl-derived chiral phosphoric acid catalyst in dichloromethane at temperatures ranging from 0°C to 10°C, the process achieves high enantioselectivity without the need for toxic metals. This organocatalytic approach not only simplifies the reaction setup but also drastically reduces the environmental footprint and safety hazards associated with handling pyrophoric or toxic metal reagents. The reaction tolerates a wide array of functional groups on the substrate, allowing for the generation of a diverse library of axially chiral products with high atom economy.
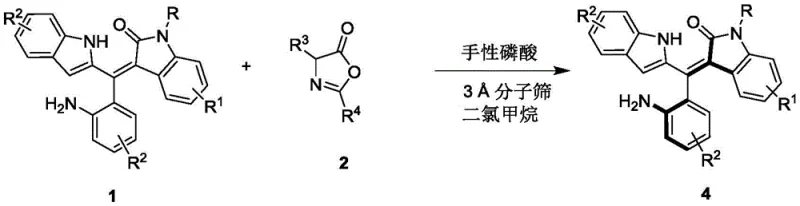
The core innovation lies in the dynamic kinetic resolution or selective transformation facilitated by the chiral phosphoric acid, which discriminates between enantiomers through a precise hydrogen-bonding network. This mechanism ensures that the desired axially chiral configuration is obtained with enantiomeric excess values reaching up to 96%, as demonstrated in the experimental data. Such high levels of stereocontrol are achieved while maintaining moderate to good yields across various substituted derivatives, proving the robustness of the method for generating reliable pharmaceutical intermediates on a commercial scale.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
The success of this resolution process is rooted in the unique ability of the chiral phosphoric acid catalyst to activate the reactants through a dual hydrogen-bonding interaction. The catalyst, typically a sterically hindered binaphthyl phosphate derivative, acts as a Brønsted acid that simultaneously activates the electrophilic and nucleophilic components of the reaction mixture. This bifunctional activation creates a highly organized chiral environment within the transition state, effectively lowering the activation energy for the formation of one specific axial atropisomer while suppressing the other. The presence of 3A molecular sieves in the reaction mixture plays a crucial role by sequestering water, thereby preventing catalyst deactivation and shifting the equilibrium towards product formation, which is vital for maintaining high turnover numbers and consistent batch-to-batch reproducibility.
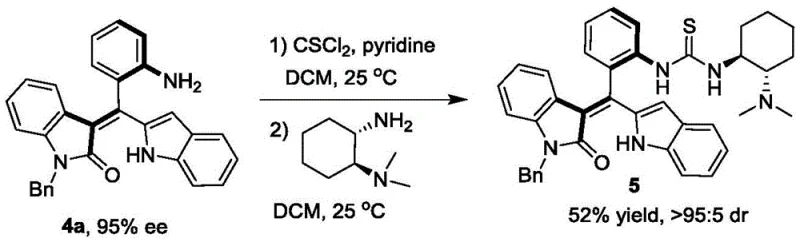
Beyond the initial resolution, the utility of the resulting axially chiral styrene is amplified through its conversion into a chiral tertiary amine catalyst. This derivatization involves reacting the resolved styrene with thiophosgene and a chiral diamine to construct a thiourea-based organocatalyst framework. This secondary transformation preserves the axial chirality established in the first step and translates it into a new catalytic function. The resulting catalyst exhibits superior performance in asymmetric [4+2] cyclization reactions, outperforming many commercially available analogues. This mechanistic continuity—from resolution to catalyst application—demonstrates a deep understanding of structure-activity relationships, providing R&D teams with a validated platform for developing custom catalytic solutions for complex bond-forming reactions.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for producing these high-value chiral intermediates. The process begins with the preparation of the reaction vessel under inert atmosphere, followed by the addition of the racemic starting material and the specific reactant in dry dichloromethane. The careful control of temperature and the precise loading of the chiral catalyst are critical parameters that dictate the final optical purity of the product. Detailed standard operating procedures for scaling this reaction from milligram to kilogram quantities rely on maintaining these specific stoichiometric ratios and thermal conditions to ensure consistent quality.
- Prepare the reaction mixture by combining the racemic Formula 1 compound and the Formula 2 reactant in dichloromethane solvent with 3A molecular sieves.
- Add 10 mol% of the chiral phosphoric acid catalyst (Formula 3) and stir the mixture at a controlled temperature between 0°C and 10°C.
- Monitor reaction progress via TLC, then filter off the molecular sieves, concentrate the filtrate, and purify the resulting axially chiral product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic resolution technology offers substantial strategic advantages over traditional metal-dependent syntheses. The elimination of precious metal catalysts such as palladium or titanium directly translates to significant cost reductions in raw material procurement, as these metals are subject to volatile market pricing and supply constraints. Furthermore, the absence of heavy metals simplifies the purification workflow, removing the need for expensive scavenging resins or complex extraction protocols designed to meet strict residual metal specifications. This streamlining of the downstream processing phase not only accelerates production timelines but also reduces the overall waste generation, aligning with increasingly stringent environmental regulations and corporate sustainability goals.
- Cost Reduction in Manufacturing: The shift from transition metal catalysis to organocatalysis fundamentally alters the cost structure of producing these complex intermediates. By utilizing readily available chiral phosphoric acids instead of proprietary metal-ligand complexes, manufacturers can achieve drastic savings on catalyst costs. Additionally, the mild reaction conditions (0°C to 10°C) reduce energy consumption for heating or cryogenic cooling, contributing to lower operational expenditures. The simplified workup procedure, which avoids heavy metal removal steps, further decreases the consumption of solvents and purification media, leading to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: Reliance on scarce transition metals often introduces supply chain vulnerabilities, whereas the reagents required for this organocatalytic method are chemically stable and widely sourced. The robustness of the reaction across a broad substrate scope means that a single catalytic system can be used to produce a variety of derivatives, reducing the need for multiple specialized catalyst inventories. This flexibility allows supply chain planners to respond more agilely to changing demand patterns for different API intermediates without requalifying entirely new synthetic routes, thereby ensuring greater continuity of supply for critical pharmaceutical projects.
- Scalability and Environmental Compliance: The operational simplicity of this resolution method makes it highly amenable to scale-up from laboratory to commercial production. The use of common solvents like dichloromethane and the absence of air-sensitive or pyrophoric reagents facilitate safer handling in large-scale reactors. From an environmental perspective, the metal-free nature of the process significantly reduces the toxicity of the waste stream, easing the burden on wastewater treatment facilities and ensuring compliance with global environmental standards. This combination of scalability and eco-friendliness positions the technology as a sustainable choice for long-term manufacturing partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial applications. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the primary advantages of this organocatalytic resolution over traditional metal-catalyzed methods?
A: This method eliminates the need for expensive and toxic transition metal catalysts like palladium or titanium, utilizing a metal-free chiral phosphoric acid instead. It operates under significantly milder conditions (0-10°C) compared to harsh coupling reactions, reducing energy consumption and simplifying downstream purification by avoiding heavy metal removal steps.
Q: What level of enantioselectivity can be achieved with this resolution process?
A: The patented process demonstrates exceptional stereocontrol, achieving enantiomeric excess (ee) values up to 96% across a broad range of substrates. This high optical purity is critical for downstream applications in asymmetric catalysis and pharmaceutical synthesis where trace impurities can compromise efficacy.
Q: Can the resolved styrene compounds be further functionalized into active catalysts?
A: Yes, the resolved axially chiral styrenes serve as versatile precursors. They can be efficiently derivatized into chiral tertiary amine catalysts (Formula 5), which have been proven to effectively catalyze asymmetric [4+2] cyclization reactions with yields up to 90% and high stereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-purity chiral intermediates for the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial supply is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation to verify enantiomeric excess and chemical identity. Our capability to handle complex organocatalytic resolutions allows us to provide reliable solutions for clients seeking to optimize their supply chains for axially chiral building blocks.
We invite potential partners to engage with our technical procurement team to discuss how this innovative resolution technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this metal-free process. We encourage you to contact us to obtain specific COA data and route feasibility assessments, ensuring that your supply of high-purity pharmaceutical intermediates is secure, compliant, and cost-optimized for the future.
