Advanced Palladium-Catalyzed Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates
Introduction to Advanced Biheterocyclic Synthesis
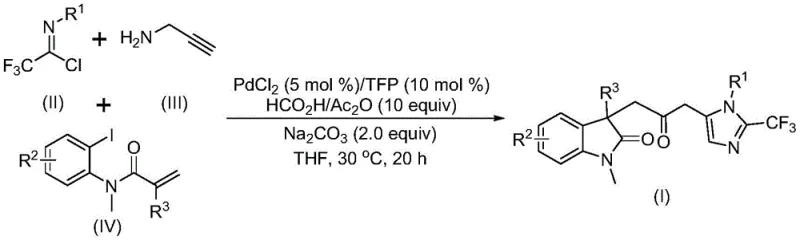
The landscape of modern pharmaceutical development increasingly relies on complex heterocyclic scaffolds that offer unique biological activities and structural diversity. Patent CN115353511A introduces a groundbreaking preparation method for carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs which are prevalent in numerous bioactive natural products and drug candidates. This technology represents a significant leap forward in organic synthesis by addressing the historical challenges associated with constructing carbonyl-bridged systems, traditionally hindered by harsh reaction conditions and poor atom economy. By leveraging a transition metal palladium-catalyzed carbonylation cascade, this invention enables the efficient assembly of these valuable molecular architectures from cheap and readily available starting materials. The strategic elimination of toxic carbon monoxide gas in favor of safer liquid substitutes not only aligns with green chemistry principles but also drastically simplifies the engineering requirements for industrial scale-up, making it an attractive option for reliable pharmaceutical intermediate suppliers seeking to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic frameworks has been plagued by synthetic inefficiencies that limit their widespread application in commercial manufacturing. Traditional approaches often rely on the direct coupling of two pre-formed heterocyclic substrates, a strategy that frequently suffers from low convergence and requires multiple protection-deprotection steps that erode overall yield. Alternatively, oxidative cyclization reactions involving bis-nucleophilic substrates often necessitate the use of stoichiometric amounts of harsh oxidants and elevated temperatures, leading to significant waste generation and safety hazards in a plant environment. Furthermore, when carbonyl groups are required as bridges between heterocycles, conventional carbonylation reactions typically demand the use of high-pressure carbon monoxide gas, which poses severe safety risks and requires specialized, expensive autoclave equipment that many contract manufacturing organizations lack. These limitations create substantial bottlenecks in the supply chain for high-purity pharmaceutical intermediates, driving up costs and extending lead times for critical drug development projects.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a sophisticated multicomponent reaction design that converges three distinct building blocks in a single operational step. By employing trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives as the foundational substrates, the process achieves a high degree of molecular complexity with exceptional atom economy. The reaction operates under remarkably mild conditions, specifically at a temperature of 30°C, which preserves sensitive functional groups and minimizes energy consumption compared to thermal-intensive alternatives. Crucially, the system replaces hazardous gaseous carbon monoxide with a safe and manageable mixture of formic acid and acetic anhydride, effectively generating the necessary carbonyl species in situ. This innovation not only enhances the safety profile of the synthesis but also broadens the scope of compatible substrates, allowing for the introduction of diverse substituents such as halogens, alkyls, and nitro groups without compromising reaction efficiency.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The success of this transformation hinges on a meticulously orchestrated palladium-catalyzed cascade mechanism that seamlessly integrates several bond-forming events. The catalytic cycle is initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This species subsequently undergoes an intramolecular Heck-type insertion into the pendant alkene, constructing the core indolinone ring system and forming a divalent alkyl-palladium intermediate. It is at this critical juncture that the carbonylation event occurs; the alkyl-palladium species reacts with carbon monoxide released from the formic acid/acetic anhydride mixture to form an acyl-palladium intermediate. Concurrently, in a parallel pathway facilitated by the base, the trifluoroethylimidoyl chloride and propargylamine undergo intermolecular carbon-nitrogen bond formation to generate a trifluoroacetamidine species, which then isomerizes to a more reactive form. The final stage involves the activation of this amidine by the acyl-palladium intermediate, triggering an intramolecular cyclization that closes the imidazole ring and releases the final carbonyl-bridged biheterocyclic product while regenerating the active palladium catalyst.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. Because the reaction proceeds through a concerted cascade within a single pot, there are fewer opportunities for the accumulation of stable side products that typically arise from isolating intermediates. The use of tris(2-furyl)phosphine (TFP) as a ligand is particularly noteworthy, as its electron-rich nature stabilizes the palladium center against aggregation and promotes the difficult carbonylation step, thereby suppressing the formation of dehalogenated byproducts. Furthermore, the mild basicity of sodium carbonate ensures that acid-sensitive functionalities on the aromatic rings remain intact, preventing degradation pathways that could complicate downstream purification. This high level of chemoselectivity is essential for producing high-purity OLED material or pharmaceutical intermediates where trace impurities can have detrimental effects on final product performance.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires strict adherence to the optimized stoichiometry and reaction parameters defined in the patent data to ensure maximum yield and reproducibility. The process begins with the preparation of the catalytic solution, where palladium chloride and the TFP ligand are dispersed in an aprotic solvent such as tetrahydrofuran, followed by the addition of the base and the carbon monoxide surrogate mixture. Once the system is equilibrated, the three organic substrates are introduced, and the mixture is stirred at 30°C for a duration of 12 to 20 hours to allow full conversion. Detailed standardized synthesis steps see the guide below.
- Prepare the catalytic system by mixing palladium chloride (PdCl2) and tris(2-furyl)phosphine (TFP) ligand with sodium carbonate base in an organic solvent like THF.
- Introduce the carbon monoxide substitute mixture (formic acid and acetic anhydride) along with the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the acrylamide derivative.
- Maintain the reaction mixture at a mild temperature of 30°C for 12 to 20 hours to allow the cascade cyclization to complete, followed by standard filtration and chromatographic purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers transformative benefits that extend far beyond simple chemical yield improvements. The primary economic driver is the utilization of commodity chemicals as starting materials; trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives are widely available from global chemical suppliers, ensuring a robust and competitive supply base that mitigates the risk of raw material shortages. By eliminating the need for high-pressure carbon monoxide infrastructure, manufacturers can significantly reduce capital expenditure on specialized reactor vessels and safety systems, translating directly into lower overhead costs per kilogram of product. Additionally, the mild reaction temperature of 30°C drastically reduces energy consumption compared to traditional high-temperature cyclizations, contributing to a smaller carbon footprint and lower utility bills. These factors combine to create a highly cost-effective manufacturing route that enhances the overall margin profile for complex heterocyclic intermediates.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the substitution of expensive and hazardous reagents with inexpensive, stable alternatives. The avoidance of toxic carbon monoxide gas removes the necessity for costly gas handling systems, leak detection monitors, and high-pressure autoclaves, which are significant capital sinks in traditional carbonylation plants. Furthermore, the high atom economy of the multicomponent reaction means that a larger proportion of the input mass is converted into the desired product, reducing the volume of waste solvent and byproducts that require disposal. This efficiency gain leads to substantial cost savings in both raw material procurement and waste management, allowing for more competitive pricing strategies in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the reliance on commercially available, off-the-shelf reagents rather than custom-synthesized precursors that may have long lead times. Propargylamine and acrylamide derivatives are produced at scale by multiple vendors worldwide, ensuring that production schedules are not held hostage by single-source supplier bottlenecks. The robustness of the reaction conditions, which tolerate a wide range of functional groups including halogens and nitro groups, means that the process is less sensitive to minor variations in raw material quality, further stabilizing the supply output. This reliability is critical for maintaining continuous production flows for critical API intermediates, ensuring that downstream drug manufacturing timelines are met without interruption.
- Scalability and Environmental Compliance: The transition from laboratory benchtop to commercial scale is facilitated by the inherent safety and simplicity of the reaction design. Operating at ambient pressure and low temperature removes the thermodynamic barriers that often complicate scale-up, allowing for straightforward expansion from gram-scale experiments to multi-ton annual production capacities. The use of tetrahydrofuran as a solvent, while requiring standard recovery protocols, is well-understood in the industry, and the absence of heavy metal waste streams (beyond the catalytic amount of palladium which can be recovered) simplifies environmental compliance reporting. This ease of scale-up ensures that the technology can rapidly meet surging market demand for advanced biheterocyclic compounds without the need for prolonged process re-engineering phases.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology, derived directly from the experimental data and claims within the patent documentation. Understanding these nuances is vital for R&D teams evaluating the feasibility of integrating this route into their existing process portfolios. The answers reflect the specific conditions and outcomes observed during the validation of the method, providing a realistic expectation of performance.
Q: What is the carbon monoxide source in this synthesis?
A: Instead of using toxic high-pressure carbon monoxide gas, this protocol utilizes an in-situ generation system comprising a mixture of formic acid and acetic anhydride, which significantly enhances operational safety.
Q: What are the optimal reaction conditions described in the patent?
A: The reaction proceeds efficiently at a mild temperature of 30°C in tetrahydrofuran (THF) solvent, typically requiring 12 to 20 hours to reach completion with high substrate compatibility.
Q: Does this method support diverse functional groups?
A: Yes, the methodology demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, alkoxy groups, and nitro groups on the aromatic rings without compromising yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
As the pharmaceutical and fine chemical industries continue to demand increasingly complex molecular scaffolds, the ability to execute sophisticated multicomponent reactions at scale becomes a key differentiator for manufacturing partners. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications. Our rigorous QC labs are equipped to handle the analytical challenges posed by fluorinated heterocycles, ensuring that every batch of carbonyl-bridged biheterocyclic compounds meets the exacting standards required for clinical and commercial applications. We understand that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands a deep commitment to process safety, regulatory compliance, and cost optimization.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain visibility into the potential economic benefits of switching to this greener, more efficient methodology. We encourage you to contact us today to obtain specific COA data for related intermediates and to receive comprehensive route feasibility assessments that will accelerate your development timelines. Let us collaborate to bring these high-value biheterocyclic structures from the laboratory to the marketplace with speed, precision, and economic efficiency.
