Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
Scalable Synthesis of 2-Trifluoromethyl Quinazolinones Using Iron Catalysis for Commercial API Production
The pharmaceutical industry continuously seeks efficient, cost-effective pathways to access privileged heterocyclic scaffolds that serve as the backbone for novel therapeutic agents. Among these, quinazolinone derivatives stand out due to their pervasive presence in bioactive molecules exhibiting anti-cancer, anticonvulsant, and anti-inflammatory properties. A significant breakthrough in this domain is detailed in patent CN111675662B, which discloses a robust preparation method for 2-trifluoromethyl substituted quinazolinone compounds. This technology leverages an inexpensive iron-catalyzed cyclization strategy, utilizing readily available isatin and trifluoroethylimidoyl chloride as key building blocks. For R&D directors and procurement specialists alike, this innovation represents a pivotal shift away from costly, condition-intensive legacy syntheses toward a more sustainable and economically viable manufacturing paradigm. The introduction of the trifluoromethyl group is particularly strategic, as it enhances the metabolic stability, lipophilicity, and bioavailability of the final drug candidates, making this synthetic route highly relevant for modern drug discovery pipelines targeting complex diseases.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of quinazolinone rings bearing trifluoromethyl functionalities has been fraught with synthetic challenges that hinder efficient large-scale production. Traditional literature methods predominantly rely on the cyclization of synthons containing the trifluoromethyl group, such as trifluoroacetic anhydride or ethyl trifluoroacetate, with substrates like anthranilamide or isatoic anhydride. While chemically feasible, these approaches suffer from severe limitations including the requirement for harsh reaction conditions, the use of expensive and moisture-sensitive reagents, and often narrow substrate scopes that limit structural diversity. Furthermore, many conventional protocols necessitate the use of precious metal catalysts or stoichiometric amounts of toxic reagents, which not only inflate the cost of goods sold (COGS) but also complicate the downstream purification processes required to meet stringent regulatory limits on heavy metal residues in active pharmaceutical ingredients (APIs). These factors collectively create bottlenecks in the supply chain, extending lead times and increasing the financial risk associated with developing new quinazolinone-based therapeutics.
The Novel Approach
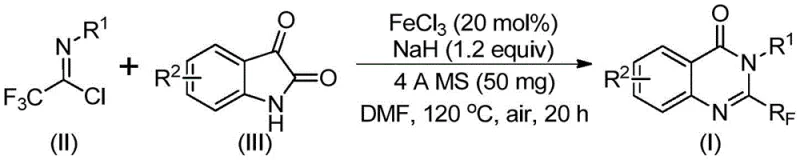
In stark contrast to these legacy issues, the methodology outlined in the referenced patent introduces a streamlined, iron-catalyzed cascade reaction that fundamentally reshapes the economic and operational landscape of quinazolinone synthesis. By employing ferric chloride (FeCl3) as a Lewis acid catalyst in conjunction with sodium hydride, the process facilitates a direct coupling between trifluoroethylimidoyl chloride and isatin derivatives. This novel approach operates under relatively mild conditions, utilizing air as the oxidant rather than requiring strict inert atmospheres, which significantly simplifies reactor engineering and operational safety protocols. The reaction proceeds through a tandem sequence involving initial carbon-nitrogen bond formation followed by decarbonylation and cyclization, delivering the target 2-trifluoromethyl quinazolinones in good to excellent yields. This shift to base-metal catalysis not only drastically reduces raw material costs but also aligns with green chemistry principles by minimizing the environmental footprint of the manufacturing process, thereby offering a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of base-promoted nucleophilic attack and transition metal-mediated cyclization, offering deep insights for process chemists aiming to optimize reaction parameters. Initially, the strong base, sodium hydride, deprotonates the isatin nitrogen, generating a nucleophilic species that attacks the electrophilic carbon of the trifluoroethylimidoyl chloride. This step forms a key trifluoroacetamidine intermediate, establishing the crucial carbon-nitrogen bond that links the two fragments. Subsequently, the ferric chloride catalyst coordinates with the carbonyl oxygen and the imine nitrogen, activating the system for an intramolecular cyclization event. This coordination lowers the activation energy for the ring closure, facilitating the expulsion of the carbonyl group (decarbonylation) and the subsequent aromatization to form the stable quinazolinone core. The ability of the iron catalyst to toggle between oxidation states likely plays a role in managing the electron flow during this redox-neutral or oxidative process, ensuring high turnover numbers and efficient conversion even at low catalyst loadings of 20 mol%.
From an impurity control perspective, understanding this mechanism is vital for ensuring high purity profiles required for GMP production. The use of 4A molecular sieves in the reaction mixture serves a critical function by sequestering trace water, which could otherwise hydrolyze the sensitive imidoyl chloride starting material or deactivate the sodium hydride base. By maintaining anhydrous conditions, the formation of hydrolysis byproducts such as trifluoroacetamide or isatin hydration products is minimized. Furthermore, the specificity of the iron catalyst for the desired cyclization pathway suppresses competing side reactions, such as polymerization or over-oxidation, which are common pitfalls in heterocycle synthesis. The result is a cleaner crude reaction profile that simplifies the subsequent purification steps, allowing for the isolation of high-purity intermediates with reduced levels of genotoxic impurities or residual solvents, thus streamlining the quality control workflow for the final API.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and scalability, making it an ideal candidate for technology transfer from the laboratory to pilot plant operations. The protocol utilizes a simple one-pot procedure where all reagents are combined in a polar aprotic solvent, typically DMF, which effectively dissolves both the organic substrates and the inorganic base. The reaction profile involves a two-stage heating regimen: an initial period at 40°C to allow for the controlled formation of the intermediate, followed by heating to 120°C to drive the cyclization to completion over a total duration of approximately 20 to 30 hours. This staged approach helps manage the exothermicity of the initial deprotonation and ensures high conversion rates. Detailed standardized synthetic steps, including precise stoichiometry, addition rates, and quenching procedures, are provided in the technical guide below to ensure consistent batch-to-batch quality.
- Combine ferric chloride (20 mol%), sodium hydride (1.2 equiv), 4A molecular sieves, trifluoroethylimidoyl chloride, and isatin derivative in anhydrous DMF solvent within a reaction vessel.
- Initiate the reaction by stirring at 40°C for approximately 10 hours to facilitate initial bond formation, then increase temperature to 120°C under air atmosphere for 20 hours to complete cyclization.
- Upon completion, filter the mixture, adsorb the crude product onto silica gel, and purify via column chromatography to isolate the high-purity 2-trifluoromethyl substituted quinazolinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology translates into tangible strategic benefits that extend far beyond simple yield improvements. The primary driver of value lies in the radical simplification of the bill of materials; by replacing expensive precious metal catalysts or specialized fluorinating agents with commodity chemicals like ferric chloride and isatin, the direct material costs are significantly reduced. Isatin is a bulk chemical produced on a multi-ton scale globally, ensuring a stable and resilient supply chain that is less susceptible to the geopolitical fluctuations often seen with rare earth metals or patented reagents. Additionally, the operational simplicity of running the reaction under air atmosphere removes the capital expenditure burden associated with maintaining extensive inert gas infrastructure, further lowering the barrier to entry for contract manufacturing organizations (CMOs) looking to offer this service.
- Cost Reduction in Manufacturing: The economic impact of switching to this catalytic system is profound, primarily driven by the elimination of high-cost inputs and the reduction of waste disposal fees. Ferric chloride is orders of magnitude cheaper than palladium or rhodium catalysts traditionally used in C-N bond formations, and its low loading requirement means less metal waste to treat at the end of the campaign. Moreover, the high atom economy of the cyclization, which incorporates most of the starting material atoms into the final product, minimizes the generation of organic waste streams. This efficiency leads to substantial cost savings in both raw material procurement and environmental compliance, allowing for more competitive pricing strategies in the global marketplace for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials that do not rely on single-source suppliers or complex logistics chains. Trifluoroethylimidoyl chloride can be synthesized from readily available aromatic amines and trifluoroacetic acid, commodities that are produced by numerous chemical manufacturers worldwide. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in utility quality (such as nitrogen purity), ensuring consistent output even in manufacturing sites with varying infrastructure capabilities, thereby guaranteeing reliable delivery schedules to downstream API producers.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and eventually to metric ton quantities is straightforward due to the absence of hazardous reagents like azides or high-pressure gases. The reaction operates at atmospheric pressure and moderate temperatures, fitting comfortably within the operating envelopes of standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. From an environmental standpoint, the use of iron, a non-toxic metal, simplifies the regulatory burden regarding heavy metal clearance in the final product, reducing the need for expensive scavenging resins or additional purification passes. This alignment with green chemistry metrics not only satisfies internal sustainability goals but also appeals to increasingly eco-conscious partners in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of this route for your specific project needs. Whether you are concerned about substrate compatibility, catalyst recovery, or regulatory acceptance, understanding these details is crucial for making informed sourcing and development decisions. We encourage technical teams to review these points closely when assessing the fit of this chemistry for their pipeline.
Q: What are the primary advantages of using FeCl3 over traditional catalysts for quinazolinone synthesis?
A: The use of ferric chloride (FeCl3) offers significant economic and operational advantages compared to traditional methods that often rely on expensive precious metals or harsh reagents like trifluoroacetic anhydride. Iron is abundant, non-toxic, and extremely cost-effective, which drastically reduces the raw material costs associated with catalyst loading. Furthermore, this catalytic system operates under aerobic conditions (air atmosphere), eliminating the need for complex inert gas setups and simplifying the engineering requirements for large-scale manufacturing.
Q: Does this synthetic method tolerate diverse functional groups on the substrate?
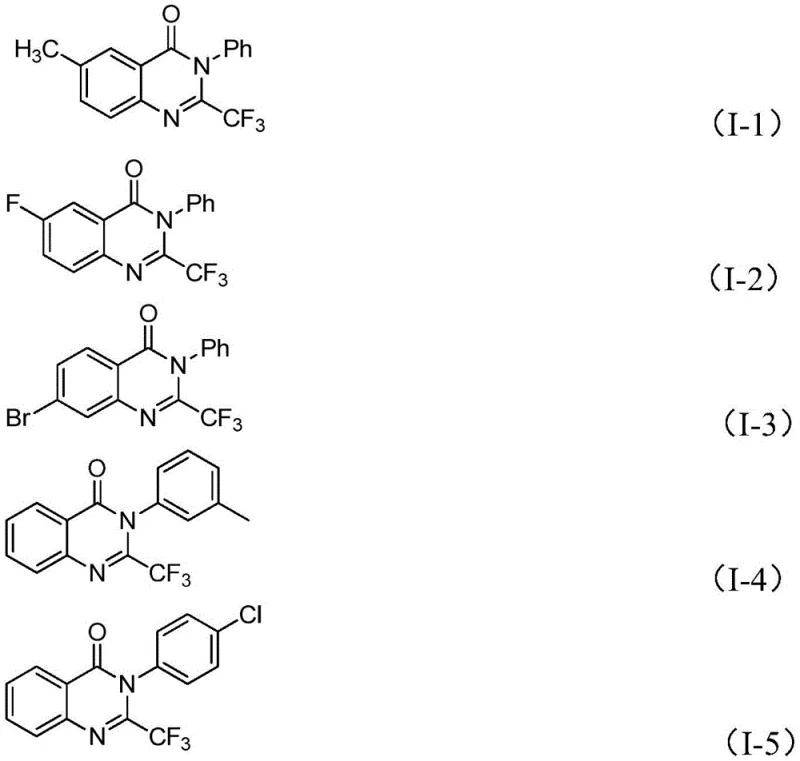
A: Yes, the methodology described in patent CN111675662B demonstrates excellent functional group tolerance, accommodating various substituents such as halogens (fluorine, chlorine, bromine), alkyl groups (methyl), and electron-withdrawing groups (nitro) on both the isatin and the imidoyl chloride components. This broad substrate scope is critical for medicinal chemists who need to generate diverse libraries of analogs for structure-activity relationship (SAR) studies without needing to redesign the synthetic route for each new derivative.
Q: Is this process suitable for industrial scale-up and GMP production?
A: The process is highly amenable to scale-up due to its use of commercially available starting materials like isatin and simple workup procedures involving filtration and standard column chromatography. The reaction utilizes DMF as a solvent, which is common in industrial settings, and the two-stage temperature profile (40°C followed by 120°C) is easily manageable in standard jacketed reactors. The elimination of sensitive reagents and the robustness of the iron catalyst make it a viable candidate for multi-kilogram production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate innovative academic chemistry into robust commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial supply is seamless and risk-mitigated. We are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone intermediate meets the exacting standards required for clinical and commercial API manufacturing. Our commitment to quality assurance means that we proactively manage impurity profiles and validate our analytical methods to support your regulatory filings with confidence.
We invite you to engage with our technical procurement team to discuss how this cost-effective synthesis route can be integrated into your supply chain. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage potential partners to contact us directly to request specific COA data for our catalog compounds or to initiate a dialogue regarding route feasibility assessments for custom derivatives. Let us collaborate to accelerate your drug development programs with high-quality, sustainably produced chemical intermediates that drive your success in the competitive pharmaceutical market.
