Advanced Pd-Catalyzed Multicomponent Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Introduction to Patent CN115353511A
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex heterocyclic scaffolds, which serve as critical cores in numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking multicomponent strategy for the synthesis of carbonyl-bridged biheterocyclic compounds, specifically targeting the efficient assembly of indolinone-imidazole hybrids. This technology represents a significant leap forward in organic synthesis by replacing hazardous gaseous reagents with safer liquid alternatives while maintaining high reaction efficiency. The core innovation lies in a palladium-catalyzed cascade reaction that seamlessly integrates carbonylation, Heck coupling, and cyclization steps into a single operational procedure. For R&D directors and process chemists, this patent offers a compelling solution to the challenges associated with building nitrogen-containing heterocycles, providing a pathway to diverse chemical space with improved safety profiles. The ability to generate these complex architectures from readily available starting materials positions this method as a highly attractive candidate for the development of next-generation pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of carbonyl-bridged biheterocyclic systems has relied heavily on direct coupling reactions between pre-formed heterocyclic units or oxidative cyclization strategies that often suffer from poor atom economy. A major bottleneck in existing literature is the reliance on transition metal-catalyzed carbonylation reactions that necessitate the use of high-pressure carbon monoxide gas. Handling toxic CO gas requires specialized high-pressure equipment, rigorous safety protocols, and extensive engineering controls, which drastically increases capital expenditure and operational complexity for manufacturing facilities. Furthermore, multi-step synthetic routes typically involve isolation and purification of unstable intermediates, leading to cumulative yield losses and increased solvent consumption. These conventional approaches often lack the flexibility to accommodate diverse functional groups, limiting the structural diversity accessible for medicinal chemistry campaigns. Consequently, there is a persistent demand for safer, more efficient, and operationally simpler methodologies that can bypass these inherent limitations of classical heterocycle synthesis.
The Novel Approach
The methodology disclosed in CN115353511A fundamentally transforms the synthetic landscape by employing a one-pot multicomponent reaction that eliminates the need for external carbon monoxide sources. Instead of using dangerous gas cylinders, the process utilizes a mixture of formic acid and acetic anhydride to generate carbon monoxide in situ under mild conditions. This strategic shift not only enhances laboratory and plant safety but also simplifies the reaction setup, allowing it to be performed in standard glassware without high-pressure reactors. The reaction integrates three distinct components—trifluoroethylimidoyl chloride, propargylamine, and an acrylamide derivative—into a unified catalytic cycle driven by a palladium catalyst system. 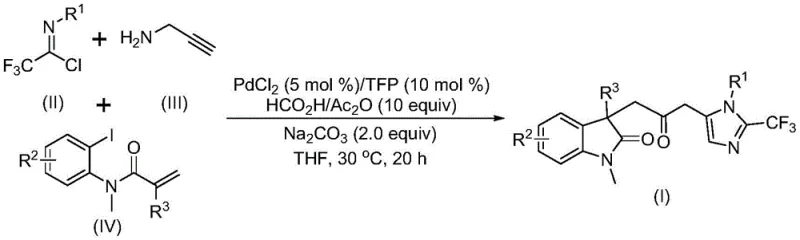 As illustrated in the reaction scheme, this convergent approach allows for the simultaneous formation of multiple chemical bonds, including C-C and C-N bonds, in a single step. The use of cheap and commercially available starting materials further underscores the practical utility of this method, making it an ideal candidate for cost reduction in API manufacturing where raw material availability is a key concern.
As illustrated in the reaction scheme, this convergent approach allows for the simultaneous formation of multiple chemical bonds, including C-C and C-N bonds, in a single step. The use of cheap and commercially available starting materials further underscores the practical utility of this method, making it an ideal candidate for cost reduction in API manufacturing where raw material availability is a key concern.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is crucial for optimizing reaction parameters and ensuring reproducibility at scale. The proposed mechanism initiates with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This is followed by an intramolecular Heck-type reaction that constructs the indolinone core, a privileged scaffold in drug discovery. Subsequently, the in situ generated carbon monoxide inserts into the alkyl-palladium bond to form an acyl-palladium species, effectively introducing the carbonyl bridge. Concurrently, the trifluoroethylimidoyl chloride reacts with propargylamine in a base-promoted manner to form a trifluoroacetamidine intermediate, which undergoes isomerization. The final stage involves the activation of this amidine by the acyl-palladium complex, triggering an intramolecular cyclization that closes the imidazole ring and releases the final carbonyl-bridged biheterocyclic product. This intricate dance of catalytic cycles highlights the sophistication of the design, where multiple reactive pathways are harmonized to proceed sequentially without interference.
From an impurity control perspective, the mild reaction temperature of 30°C plays a pivotal role in suppressing side reactions such as polymerization of the acrylamide or decomposition of sensitive intermediates. The choice of tetrahydrofuran (THF) as the solvent is also critical, as it effectively solubilizes all organic components while stabilizing the palladium catalytic species. The use of sodium carbonate as a base ensures neutralization of acidic byproducts without promoting hydrolysis of the sensitive imidoyl chloride moiety. For quality assurance teams, this mechanistic clarity suggests a clean impurity profile, primarily consisting of unreacted starting materials which are easily removed during workup. The high functional group tolerance observed across various substrates indicates that the catalytic cycle is robust against electronic variations, ensuring consistent product quality regardless of the specific substituents on the aromatic rings. This level of predictability is essential for regulatory filings where defining the impurity landscape is a mandatory requirement.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow a standardized protocol that emphasizes precise stoichiometry and atmospheric control. The process begins with the preparation of the catalyst system, where palladium chloride and trifurylphosphine are mixed with the CO-generating reagents in the chosen solvent. It is imperative to maintain an inert atmosphere to prevent oxidation of the low-valent palladium species, which could deactivate the catalyst. Once the reaction mixture is equilibrated, the three key substrates are introduced, and the system is allowed to stir at the specified mild temperature for a duration of 12 to 20 hours.
- Combine palladium chloride, trifurylphosphine ligand, sodium carbonate, and the formic acid/acetic anhydride CO source in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the acrylamide substrate to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12-20 hours, followed by filtration and column chromatography purification to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented technology offers tangible benefits that extend beyond mere chemical novelty. The elimination of high-pressure carbon monoxide gas removes a significant logistical and safety hurdle, thereby reducing the need for specialized storage infrastructure and hazardous material handling certifications. This simplification directly translates to lower overhead costs and reduced insurance premiums for manufacturing sites. Furthermore, the use of commodity chemicals like formic acid and acetic anhydride as CO surrogates ensures a stable and predictable supply chain, mitigating risks associated with the sourcing of specialized gaseous reagents. The one-pot nature of the reaction significantly reduces solvent usage and processing time compared to multi-step linear syntheses, leading to substantial cost savings in waste disposal and energy consumption. These factors collectively enhance the overall economic viability of producing these complex intermediates, making them more accessible for large-scale drug development programs.
- Cost Reduction in Manufacturing: The streamlined one-pot procedure minimizes unit operations, effectively lowering labor costs and equipment occupancy time. By avoiding the isolation of intermediates, the process reduces material loss and solvent requirements, driving down the cost of goods sold (COGS). The use of inexpensive palladium chloride instead of more exotic catalysts further optimizes the catalyst cost contribution. Additionally, the mild reaction conditions reduce energy demands for heating or cooling, contributing to a smaller carbon footprint and lower utility bills. These cumulative efficiencies make the process highly competitive for commercial production of high-value pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures that production schedules are not disrupted by shortages of niche reagents. The robustness of the reaction across a wide range of substrates means that a single manufacturing line can be adapted to produce a diverse library of analogues without significant retooling. This flexibility allows suppliers to respond rapidly to changing market demands or clinical trial requirements. The scalability demonstrated in the patent, extending from milligram to gram scales, provides confidence that the process can be transferred to multi-kilogram production with minimal technical risk, ensuring continuity of supply for downstream customers.
- Scalability and Environmental Compliance: Operating at ambient pressure and low temperature significantly lowers the safety risks associated with scale-up, facilitating easier regulatory approval for new manufacturing processes. The reduced generation of hazardous waste, particularly the avoidance of heavy metal contamination from complex catalyst systems, simplifies effluent treatment and disposal. This aligns with modern green chemistry principles and helps manufacturers meet increasingly stringent environmental regulations. The high atom economy of the multicomponent reaction ensures that a greater proportion of raw materials end up in the final product, minimizing waste generation and maximizing resource efficiency throughout the production lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis method. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to provide accurate guidance for potential adopters. Understanding these nuances is vital for assessing the feasibility of integrating this technology into existing production workflows.
Q: How does this method address the safety concerns of traditional carbonylation?
A: Unlike conventional methods requiring toxic carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate CO in situ, significantly enhancing operational safety and reducing regulatory compliance burdens.
Q: What is the substrate compatibility for this Pd-catalyzed reaction?
A: The protocol demonstrates excellent functional group tolerance, accommodating various substituents such as halogens, alkyl groups, alkoxy, nitro, and trifluoromethyl groups on the aromatic rings without compromising yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates at mild temperatures (30°C) and has been successfully expanded to gram-scale synthesis, indicating strong potential for commercial scale-up with simplified thermal management requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115353511A for accelerating drug discovery and development. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to marketplace. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with stringent purity specifications, supported by rigorous QC labs that guarantee the highest quality standards for every batch. We are committed to leveraging our technical expertise to optimize this specific carbonylation route for your unique needs, delivering reliable Carbonyl-Bridged Biheterocyclic Compounds that meet the demanding requirements of the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can benefit your specific pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to bring your next-generation therapeutics to life with speed, safety, and scientific excellence.
