Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrol-2-One Scaffolds for Commercial Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of 1,5-Dihydro-2H-Pyrrol-2-One Scaffolds for Commercial Pharmaceutical Applications
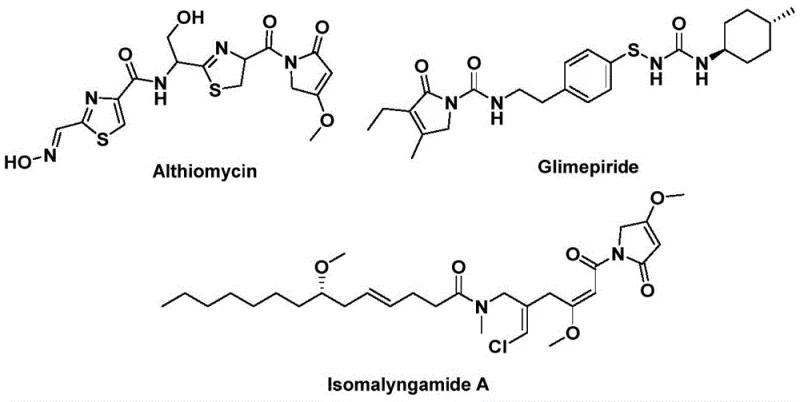
The landscape of organic synthesis for bioactive heterocycles is constantly evolving, driven by the need for safer, more efficient, and scalable methodologies. A significant breakthrough in this domain is detailed in patent CN112694430B, which discloses a robust preparation method for 1,5-dihydro-2H-pyrrole-2-ketone compounds. This structural motif is not merely an academic curiosity but a critical pharmacophore found in numerous high-value natural products and therapeutic agents. For instance, the antibiotic althiomycin, the hypoglycemic agent glimepiride, and the anticancer candidate isomalyngamide A all share this core architecture, highlighting its immense versatility in drug discovery. The ability to construct this five-membered lactam ring efficiently is therefore of paramount importance to the global pharmaceutical supply chain.
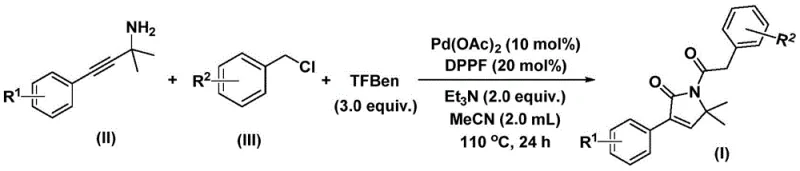
The patented methodology leverages a palladium-catalyzed bis-carbonylation strategy that transforms readily available propargyl amines and benzyl chlorides into the desired pyrrolone derivatives in a single operational step. Unlike traditional approaches that often rely on toxic gaseous carbon monoxide under high pressure, this innovation utilizes phenyl 1,3,5-tricarboxylate as a safe and manageable solid carbon monoxide surrogate. This shift from gas to solid reagents represents a paradigm shift in process safety, making the technology particularly attractive for large-scale manufacturing environments where regulatory compliance and operator safety are non-negotiable priorities for any reliable pharmaceutical intermediate supplier.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 1,5-dihydro-2H-pyrrol-2-ones has been fraught with significant technical and logistical challenges. Conventional carbonylation reactions typically necessitate the use of carbon monoxide gas, which poses severe safety hazards due to its toxicity and flammability. Handling CO gas requires specialized high-pressure autoclaves and rigorous safety protocols, which inherently increases the capital expenditure and operational complexity for chemical manufacturers. Furthermore, many existing synthetic routes involve multi-step sequences to install the necessary functional groups prior to cyclization, leading to cumulative yield losses and increased waste generation. These inefficiencies create bottlenecks in the supply chain, resulting in longer lead times and higher costs for downstream drug developers seeking high-purity intermediates.
The Novel Approach
In stark contrast, the novel approach described in the patent offers a streamlined, one-pot solution that circumvents these traditional hurdles. By employing a palladium catalyst system in conjunction with a solid CO surrogate, the reaction proceeds under relatively mild thermal conditions without the need for pressurized gas infrastructure. The process exhibits remarkable substrate compatibility, tolerating a wide array of electronic and steric variations on both the amine and chloride coupling partners. This versatility ensures that a diverse library of analogs can be generated rapidly, accelerating the structure-activity relationship (SAR) studies crucial for modern drug discovery. The simplicity of the workup procedure, involving basic filtration and chromatography, further underscores the practical utility of this method for industrial applications.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
To fully appreciate the elegance of this transformation, one must delve into the catalytic cycle that drives the formation of the pyrrolone ring. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. This key organometallic species then undergoes the first migratory insertion of carbon monoxide, which is released in situ from the decomposition of the phenyl 1,3,5-tricarboxylate additive. This step forms an acyl-palladium complex, setting the stage for the subsequent nucleophilic attack by the propargyl amine.
Following the initial coupling, the mechanism proceeds through a second carbonylation event. The intermediate formed after amine addition undergoes another CO insertion, effectively building the carbon backbone required for the five-membered ring closure. This bis-carbonylation sequence is critical, as it installs the two carbonyl groups characteristic of the final 1,5-dihydro-2H-pyrrol-2-one structure. The cycle concludes with a reductive elimination step that releases the final product and regenerates the active palladium catalyst, allowing the turnover to continue. Understanding this mechanistic pathway is vital for process chemists aiming to optimize reaction parameters and minimize impurity formation during scale-up.
Impurity control is inherently managed by the specificity of the palladium catalyst and the controlled release of carbon monoxide. The use of the solid CO surrogate prevents the local excess of CO that can sometimes lead to over-carbonylation or polymerization side reactions. Additionally, the choice of ligand, specifically 1,1'-bis(diphenylphosphino)ferrocene (DPPF), plays a crucial role in stabilizing the palladium center and directing the regioselectivity of the insertion steps. This precise control over the reaction trajectory ensures that the crude reaction mixture is clean, thereby reducing the burden on downstream purification processes and enhancing the overall yield of the target API intermediate.
How to Synthesize 1,5-Dihydro-2H-Pyrrol-2-One Efficiently
The execution of this synthesis is designed for reproducibility and ease of handling, making it accessible for both laboratory research and pilot plant operations. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and the two primary substrates in an organic solvent such as acetonitrile. The mixture is then heated to promote the cascade reaction. Detailed standard operating procedures regarding stoichiometry, temperature profiles, and purification techniques are essential for consistent results. For a comprehensive guide on the specific molar ratios and workup details, please refer to the standardized synthesis steps outlined below.
- Combine palladium acetate catalyst, DPPP ligand, triethylamine base, and phenyl 1,3,5-tricarboxylate (CO source) with propargyl amine and benzyl chloride substrates in acetonitrile solvent.
- Heat the reaction mixture to 110°C and maintain stirring for 24 hours to facilitate the bis-carbonylation and cyclization sequence.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 1,5-dihydro-2H-pyrrol-2-one product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling value propositions for procurement managers and supply chain directors tasked with securing reliable sources of complex intermediates. The shift away from hazardous gaseous reagents to stable solid surrogates fundamentally alters the risk profile of the manufacturing process. This reduction in hazard translates directly into lower insurance premiums and reduced regulatory overhead, contributing to a more resilient supply chain. Furthermore, the use of commodity chemicals like benzyl chlorides and propargyl amines ensures that raw material availability is not a bottleneck, safeguarding against market volatility.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily driven by the elimination of expensive high-pressure equipment and the simplification of the synthetic route. By condensing what might traditionally be a multi-step sequence into a single pot, manufacturers save significantly on labor, energy, and solvent consumption. The high atom economy of the carbonylation reaction also means less waste disposal cost. These factors combine to offer a highly competitive cost structure for the production of these valuable scaffolds, enabling significant cost reduction in pharmaceutical intermediate manufacturing without compromising quality.
- Enhanced Supply Chain Reliability: Supply continuity is critical for pharmaceutical companies, and this method enhances reliability by relying on widely available starting materials. Benzyl chlorides and substituted propargyl amines are commodity chemicals produced by numerous global suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate various functional groups, means that minor variations in raw material quality are less likely to cause batch failures. This resilience ensures a steady flow of materials, reducing lead time for high-purity pharmaceutical intermediates and supporting just-in-time manufacturing strategies.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently scalable due to its homogeneous nature and lack of gas handling requirements. The absence of high-pressure CO simplifies the engineering controls needed for ton-scale production. Moreover, the environmental footprint is minimized through the use of efficient catalysis and the generation of benign byproducts. This aligns with the increasing industry demand for green chemistry solutions, facilitating easier permitting and compliance with stringent environmental regulations in major manufacturing hubs.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding this synthesis platform, we have compiled answers based on the specific data and findings within the patent literature. These questions cover the scope of the reaction, the nature of the reagents, and the practical implications for process development. Understanding these details helps stakeholders make informed decisions about integrating this technology into their own pipelines.
Q: What are the primary advantages of this palladium-catalyzed method over traditional carbonylation?
A: This method eliminates the need for hazardous high-pressure carbon monoxide gas by utilizing a solid phenol ester surrogate, significantly enhancing operational safety and simplifying reactor requirements for scale-up.
Q: What is the substrate scope for the benzyl chloride and propargyl amine components?
A: The process demonstrates excellent functional group tolerance, accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing substituents such as halogens and trifluoromethyl groups on both aromatic rings.
Q: How does this synthesis impact the cost structure for pharmaceutical intermediates?
A: By employing commercially available starting materials and avoiding complex multi-step sequences or specialized high-pressure equipment, the method drastically reduces capital expenditure and raw material costs associated with producing pyrrolone scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-Pyrrol-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like this palladium-catalyzed bis-carbonylation in driving drug innovation. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned to industrial reality. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch.
We invite potential partners to engage with us to explore how this efficient synthesis route can optimize your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a partnership built on transparency and technical excellence.
