Advanced Palladium-Catalyzed Bis-Carbonylation for Scalable Pharmaceutical Intermediate Production
Advanced Palladium-Catalyzed Bis-Carbonylation for Scalable Pharmaceutical Intermediate Production
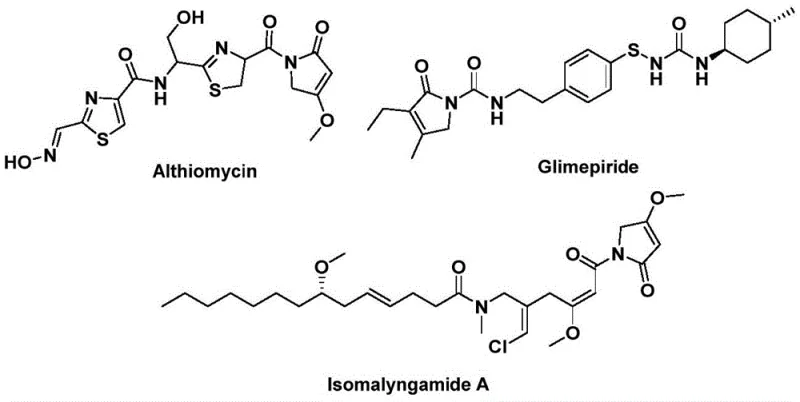
The development of efficient synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry, particularly for scaffolds found in potent bioactive natural products. Patent CN112694430B introduces a robust and versatile methodology for the preparation of 1,5-dihydro-2H-pyrrol-2-one compounds, a structural motif prevalent in significant therapeutic agents. As illustrated in the provided structural examples, this core skeleton is integral to molecules such as Althiomycin, known for its antibacterial properties, Glimepiride, a widely used hypoglycemic agent, and Isomalyngamide A, which exhibits promising anticancer activity. The ability to construct this five-membered lactam ring efficiently is therefore of paramount importance to research and development teams aiming to access novel drug candidates or optimize existing supply chains for key intermediates.
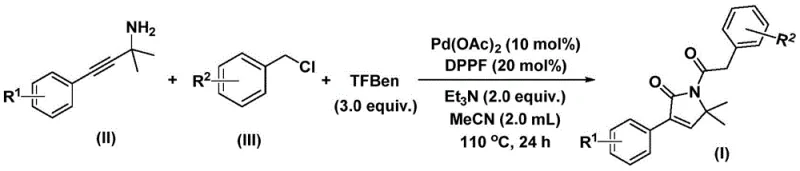
This patented technology addresses the longstanding challenges associated with carbonylation reactions by replacing hazardous gaseous carbon monoxide with a solid surrogate, thereby streamlining the operational workflow. The process employs a palladium-catalyzed bis-carbonylation strategy that couples readily available propargyl amines with benzyl chlorides in a single pot. For procurement managers and supply chain directors, this represents a significant shift towards safer, more manageable chemistry that does not compromise on yield or purity. The method operates under relatively mild thermal conditions using standard organic solvents, facilitating easier scale-up from gram-scale discovery to multi-kilogram commercial production without the need for specialized high-pressure infrastructure.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches to 1,5-dihydro-2H-pyrrol-2-one derivatives often rely on multi-step sequences involving harsh reagents or the direct use of carbon monoxide gas, which poses severe safety and logistical hurdles. Handling pressurized CO gas requires dedicated autoclaves, rigorous safety protocols, and specialized training, creating bottlenecks in both R&D and manufacturing environments. Furthermore, conventional carbonylation processes frequently suffer from poor atom economy or require expensive, air-sensitive catalysts that degrade rapidly, leading to inconsistent batch quality and increased waste generation. The reliance on gaseous reagents also complicates the stoichiometry control, often resulting in incomplete conversions or the formation of difficult-to-remove side products that necessitate complex purification procedures, ultimately driving up the cost of goods sold.
The Novel Approach
In stark contrast, the methodology disclosed in CN112694430B utilizes TFBen (a solid carbon monoxide substitute) to generate CO in situ, effectively mitigating the risks associated with gas handling while maintaining high reaction efficiency. This one-pot transformation seamlessly integrates the carbonylation and cyclization steps, directly converting simple starting materials into the complex heterocyclic core with impressive yields ranging from 70% to over 90% across various substrates. By employing a palladium acetate and DPPF ligand system, the reaction achieves excellent functional group tolerance, accommodating electron-rich and electron-deficient substituents on the aromatic rings without significant loss in performance. This operational simplicity not only accelerates the timeline for process development but also ensures a more consistent and reliable supply of high-purity intermediates for downstream applications.
Mechanistic Insights into Pd-Catalyzed Bis-Carbonylation Cyclization
The catalytic cycle begins with the oxidative addition of the palladium(0) species into the carbon-chlorine bond of the benzyl chloride substrate, generating a reactive benzyl-palladium intermediate. Subsequently, carbon monoxide, which is liberated in situ from the decomposition of the phenol 1,3,5-tricarboxylate surrogate, inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate then undergoes nucleophilic attack by the propargyl amine, initiating the ring-closing sequence to form a five-membered palladacycle. A second insertion of carbon monoxide follows, expanding the coordination sphere before a final reductive elimination step releases the desired 1,5-dihydro-2H-pyrrol-2-one product and regenerates the active palladium catalyst for the next turnover.
Understanding the impurity profile is critical for R&D directors focused on regulatory compliance, and this mechanism offers inherent advantages in that regard. The use of a solid CO source allows for a controlled, steady release of carbon monoxide, preventing the local excesses that often lead to double-carbonylation side products or polymerization in gas-phase reactions. Additionally, the specific choice of the DPPF ligand stabilizes the palladium center throughout the multiple insertion steps, minimizing catalyst decomposition and the formation of palladium black, which can be a source of metal contamination in the final API. The reaction conditions, specifically the use of acetonitrile as a solvent and triethylamine as a base, are optimized to solubilize the ionic intermediates while suppressing competing hydrolysis pathways, ensuring a clean reaction mixture that simplifies downstream purification.
How to Synthesize 1,5-Dihydro-2H-pyrrol-2-one Efficiently
The synthesis protocol outlined in the patent provides a straightforward recipe for accessing these valuable heterocycles with minimal equipment requirements. The process involves charging a reaction vessel with the palladium catalyst, ligand, base, CO surrogate, and the two primary coupling partners in acetonitrile, followed by heating to 110°C for 24 hours. This standardized approach eliminates the need for custom reactor setups and allows for parallel synthesis of diverse analogues by simply varying the substitution patterns on the starting benzyl chloride or propargyl amine. For detailed operational parameters and specific molar ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Combine palladium acetate, DPPF ligand, triethylamine, TFBen (CO source), propargyl amine, and benzyl chloride in acetonitrile solvent within a reaction vessel.
- Heat the reaction mixture to a temperature range of 100-120°C and maintain stirring for a duration of 24 to 48 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 1,5-dihydro-2H-pyrrol-2-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers substantial benefits that directly address the pain points of cost and supply continuity in fine chemical manufacturing. The shift from gaseous reagents to solid surrogates fundamentally changes the capital expenditure requirements for production facilities, allowing manufacturers to utilize standard glass-lined reactors rather than investing in expensive high-pressure equipment. This reduction in infrastructure complexity translates directly into lower overhead costs and faster turnaround times for campaign manufacturing. Furthermore, the high atom economy and single-step nature of the transformation reduce the consumption of solvents and auxiliary materials, aligning with modern green chemistry initiatives and reducing the environmental footprint of the supply chain.
- Cost Reduction in Manufacturing: The elimination of high-pressure carbon monoxide gas removes the need for specialized safety containment and monitoring systems, drastically lowering the operational expenditure associated with the synthesis. Additionally, the starting materials, specifically benzyl chlorides and propargyl amines, are commodity chemicals available from multiple global suppliers, ensuring competitive pricing and preventing single-source dependency. The high yields reported across a broad substrate scope mean that less raw material is wasted per kilogram of product, further optimizing the cost basis for large-scale production runs.
- Enhanced Supply Chain Reliability: By utilizing stable, solid reagents like TFBen and commercially available catalysts, the risk of supply disruption due to hazardous material transport restrictions is significantly minimized. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that supply chain managers can source alternative grades of starting materials without needing to requalify the entire process. This flexibility ensures continuous production capability even when specific raw material lots vary slightly in quality, providing a buffer against market volatility.
- Scalability and Environmental Compliance: The simplified workup procedure, involving filtration and standard column chromatography or crystallization, is easily adaptable to industrial scales without generating excessive hazardous waste streams. The absence of toxic gas emissions simplifies the permitting process for new manufacturing sites and reduces the burden on waste treatment facilities. This environmental compatibility is increasingly important for multinational corporations seeking to meet stringent sustainability goals and regulatory standards in regions with tight environmental controls.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is essential for successful technology transfer and process optimization.
Q: What are the safety advantages of this carbonylation method compared to traditional CO gas methods?
A: This method utilizes TFBen (a solid carbon monoxide surrogate) instead of toxic, high-pressure carbon monoxide gas. This significantly reduces safety risks associated with gas handling and eliminates the need for specialized high-pressure autoclaves, making the process safer and more accessible for standard laboratory and pilot plant setups.
Q: What is the substrate scope for the benzyl chloride component in this reaction?
A: The reaction demonstrates excellent functional group tolerance. Benzyl chlorides substituted with electron-donating groups (like methyl, methoxy) and electron-withdrawing groups (like fluoro, chloro, bromo, trifluoromethyl, cyano) at various positions (ortho, meta, para) are all compatible, yielding products with high efficiency.
Q: How does the use of DPPF ligand impact the reaction efficiency?
A: The use of 1,1'-bis(diphenylphosphino)ferrocene (DPPF) as a bidentate ligand in conjunction with palladium acetate creates a highly active catalytic species. This specific ligand system facilitates the critical bis-carbonylation and cyclization steps, leading to higher yields and better compatibility with diverse substrates compared to monodentate phosphines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,5-Dihydro-2H-pyrrol-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless and compliant. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,5-dihydro-2H-pyrrol-2-one intermediate meets the exacting standards required for pharmaceutical applications, providing our partners with the confidence they need to move forward.
We invite you to leverage our technical expertise to evaluate the feasibility of this palladium-catalyzed route for your specific project needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, where we can model the potential economic benefits of switching to this safer, more efficient process. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process and help you secure a reliable supply of high-quality intermediates.
