Scalable Palladium-Catalyzed Synthesis of N-Acyl Indoles for Advanced Pharmaceutical Manufacturing
Scalable Palladium-Catalyzed Synthesis of N-Acyl Indoles for Advanced Pharmaceutical Manufacturing
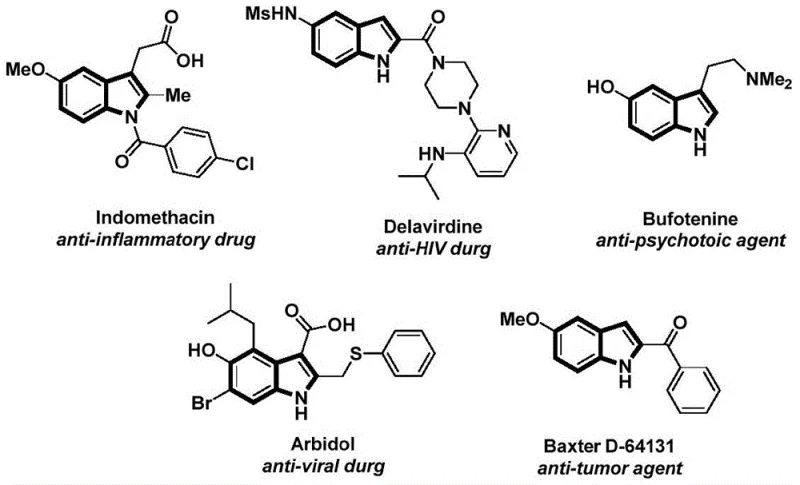
The structural motif of indole is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to anti-HIV drugs such as Delavirdine. As depicted in the reference structures, the versatility of the indole nucleus makes it a critical target for efficient synthetic methodologies. Patent CN112898192B introduces a significant advancement in this domain by disclosing a robust preparation method for N-acyl indole compounds. This innovation addresses long-standing challenges in heterocyclic synthesis by utilizing a palladium-catalyzed carbonylation cyclization strategy that operates under remarkably mild conditions. For R&D directors and process chemists seeking reliable pharmaceutical intermediate supplier partnerships, this technology represents a paradigm shift towards safer, more efficient manufacturing protocols that bypass the limitations of traditional high-pressure carbonylation techniques.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-acyl indoles via carbonylation has been fraught with operational hazards and inefficiencies. Traditional methods often rely on the direct use of carbon monoxide gas, which poses severe safety risks due to its high toxicity and flammability, requiring specialized high-pressure equipment and rigorous safety protocols that inflate capital expenditure. Furthermore, conventional catalytic systems frequently suffer from poor substrate tolerance, leading to low yields when complex functional groups are present on the aromatic rings. Many existing routes also require harsh reaction conditions, such as elevated temperatures exceeding 100°C or the use of strong bases that can degrade sensitive intermediates. These factors collectively result in inconsistent product quality, difficult purification processes, and substantial waste generation, making scale-up for commercial production economically unviable for many fine chemical manufacturers.
The Novel Approach
The methodology outlined in the patent data offers a transformative solution by employing 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide surrogate. This substitution eliminates the need for gaseous CO, drastically simplifying the reactor setup and enhancing workplace safety. The process utilizes a dual-stage thermal protocol where the reaction initiates at a moderate 60°C for 24 hours, followed by the addition of silver oxide and a subsequent 24-hour hold at the same temperature. This mild thermal profile ensures high conversion rates while preserving the integrity of sensitive functional groups. By integrating the carbonylation and cyclization steps into a streamlined sequence, this approach achieves high reaction efficiency and broad substrate compatibility, enabling the rapid synthesis of diverse N-acyl indole derivatives with minimal operational complexity.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The core of this synthetic breakthrough lies in the intricate interplay between the palladium catalyst and the specific reagents chosen. The reaction mechanism initiates with the oxidative addition of the aryl iodide to the palladium(0) species, generating a reactive aryl-palladium intermediate. Subsequently, carbon monoxide, which is slowly released in situ from the decomposition of TFBen, inserts into the palladium-carbon bond to form an acyl-palladium complex. This acyl species then undergoes nucleophilic attack by the amine group of the 2-alkynyl aniline, followed by reductive elimination to yield an amide intermediate. The presence of potassium carbonate acts as a base to neutralize acidic byproducts and facilitate the transmetallation steps. This precise orchestration of organometallic steps ensures that the carbonyl group is installed regioselectively at the nitrogen position, setting the stage for the final ring closure.
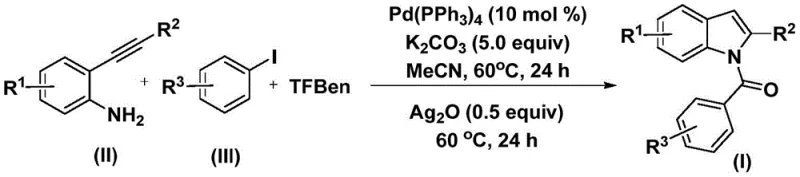
The second phase of the mechanism involves the crucial role of silver oxide (Ag2O). Upon addition to the reaction mixture, silver oxide promotes the intramolecular cyclization of the previously formed amide intermediate. It likely functions by activating the alkyne moiety or facilitating the deprotonation necessary for the nucleophilic attack of the nitrogen onto the triple bond, ultimately forming the five-membered pyrrole ring of the indole system. This step is critical for achieving the final N-acyl indole structure with high purity. The use of acetonitrile as the solvent further optimizes this process by effectively dissolving both the organic substrates and the inorganic salts, ensuring homogeneous reaction conditions that minimize side reactions and impurity formation. The result is a clean transformation that tolerates a wide range of substituents, including halogens and electron-donating groups, as evidenced by the successful synthesis of compounds I-1 through I-5.
How to Synthesize N-Acyl Indole Compounds Efficiently
To implement this high-efficiency synthesis in a laboratory or pilot plant setting, operators must adhere to the specific stoichiometric ratios and thermal profiles defined in the patent. The process begins by charging a reactor with the palladium catalyst, specifically tetrakis(triphenylphosphine)palladium, along with potassium carbonate and the solid CO source TFBen. The substrates, 2-alkynyl aniline and aryl iodide, are then introduced into the acetonitrile solvent system. The detailed standardized synthesis steps below outline the precise addition sequences and workup procedures required to maximize yield and purity.
- Combine palladium catalyst, potassium carbonate, TFBen (CO source), 2-alkynyl aniline, and aryl iodide in acetonitrile.
- Heat the mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide to the reaction mixture and continue heating at 60°C for another 24 hours to induce cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers distinct strategic advantages over legacy synthesis routes. The primary benefit stems from the substitution of hazardous gaseous carbon monoxide with a stable solid reagent, which significantly reduces logistics costs and regulatory burdens associated with transporting and storing toxic gases. This shift not only lowers the barrier to entry for manufacturing facilities lacking high-pressure infrastructure but also enhances overall supply chain reliability by mitigating the risk of production stoppages due to safety incidents. Furthermore, the use of commercially available starting materials like aryl iodides and 2-alkynyl anilines ensures a steady supply of raw inputs, reducing lead time for high-purity pharmaceutical intermediates and stabilizing production schedules.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment requirements translates directly into lower capital expenditure and reduced maintenance costs for production facilities. Additionally, the mild reaction temperature of 60°C consumes significantly less energy compared to traditional high-temperature reflux methods, leading to substantial operational savings. The high atom economy of the carbonylation cyclization minimizes waste disposal costs, while the simplified one-pot procedure reduces labor hours and solvent usage, collectively driving down the cost per kilogram of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: By utilizing robust and widely available reagents such as potassium carbonate and silver oxide, manufacturers can avoid bottlenecks associated with specialty catalysts or unstable precursors. The broad substrate scope allows for the flexible sourcing of various substituted aryl iodides, providing procurement teams with multiple vendor options to mitigate supply risks. This flexibility ensures continuous production flow even when specific raw material markets experience volatility, thereby securing the continuity of supply for downstream drug formulation partners.
- Scalability and Environmental Compliance: The protocol's reliance on mild conditions and non-toxic CO surrogates aligns perfectly with modern green chemistry principles and stringent environmental regulations. Scaling this process from gram-scale R&D to multi-ton commercial production is straightforward, as it does not require complex engineering controls for gas handling. The reduced generation of hazardous waste and the use of common solvents like acetonitrile simplify effluent treatment processes, ensuring that large-scale manufacturing remains compliant with global environmental standards while maintaining high throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this N-acyl indole synthesis route. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, providing clarity on reaction parameters and scope limitations for potential licensees and manufacturing partners.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a solid, safe, and easy-to-handle carbon monoxide substitute, eliminating the need for hazardous high-pressure CO gas cylinders while maintaining high reaction efficiency.
Q: What is the role of Silver Oxide (Ag2O) in the reaction mechanism?
A: Silver oxide acts as a crucial promoter in the second stage of the reaction, facilitating the intramolecular cyclization of the intermediate amide to form the final N-acyl indole structure under mild thermal conditions.
Q: Does this method support diverse functional groups on the substrates?
A: Yes, the protocol demonstrates excellent substrate compatibility, tolerating various substituents such as halogens (F, Cl, Br), alkyl groups (Me, tBu), and alkoxy groups (OMe) on both the aniline and aryl iodide components.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering high-purity N-acyl indole intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced catalytic technologies like the palladium-mediated carbonylation described here, we empower our clients to accelerate their drug development timelines with confidence.
We invite global pharmaceutical partners to collaborate with us to optimize their supply chains and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can enhance your production efficiency and product quality.
