Advanced Nickel-Catalyzed Thioester Synthesis for Commercial Scale-up of Complex Pharmaceutical Intermediates
Advanced Nickel-Catalyzed Thioester Synthesis for Commercial Scale-up of Complex Pharmaceutical Intermediates
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical and agrochemical manufacturing, where efficiency, safety, and cost-effectiveness are paramount. A significant breakthrough in this domain is detailed in patent CN112239384B, which discloses a novel preparation method for thioester compounds utilizing a nickel-catalyzed carbonylation strategy. Thioesters are not merely academic curiosities; they are critical structural motifs found in a vast array of bioactive molecules, including notable pharmaceutical agents such as Timobesone, Pivalopril, and Spironolactone, as illustrated in the structural diversity shown below. This patent introduces a transformative approach that replaces traditional, hazardous sulfur sources with benign sulfonyl chlorides, thereby addressing long-standing challenges in the production of high-purity pharmaceutical intermediates.
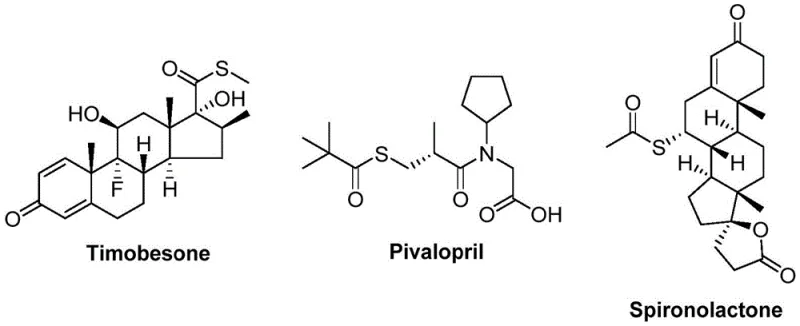
The strategic importance of this technology cannot be overstated for R&D directors and supply chain managers alike. By leveraging cheap and readily available starting materials like arylboronic acids and sulfonyl chlorides, the method offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. The process is characterized by its operational simplicity, wide functional group tolerance, and high reaction efficiency, eliminating the need for additional oxidants or reductants. This represents a substantial leap forward in green chemistry principles applied to industrial synthesis, ensuring that the production of these vital building blocks is both economically viable and environmentally sustainable for reliable pharmaceutical intermediate suppliers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has relied heavily on the direct acylation of thiols with carboxylic acids or their derivatives. While chemically straightforward in a laboratory setting, this conventional approach presents severe limitations when translated to an industrial manufacturing environment. The primary drawback is the inherent nature of thiols, which are infamous for their extremely unpleasant, pervasive odors that pose significant occupational health and safety hazards. Furthermore, thiols are potent catalyst poisons; their strong affinity for transition metals often leads to catalyst deactivation, resulting in inconsistent reaction rates, lower yields, and the necessity for excessive catalyst loading. These factors collectively drive up the cost of goods sold and complicate waste management protocols, making traditional thiol-based routes less attractive for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN112239384B utilizes sulfonyl chlorides as the sulfur source, effectively circumventing the drawbacks associated with thiols. Sulfonyl chlorides are stable, odorless, and widely available commodity chemicals, which drastically simplifies handling and storage requirements. The new protocol employs a sophisticated nickel-catalyzed carbonylation system that activates the sulfonyl chloride for coupling with arylboronic acids. This shift in strategy not only enhances the safety profile of the synthesis but also broadens the substrate scope significantly. The method demonstrates excellent compatibility with various functional groups, allowing for the synthesis of diverse thioester derivatives without the need for extensive protecting group strategies. This flexibility is crucial for reducing lead time for high-purity pharmaceutical intermediates during the drug development lifecycle.
Mechanistic Insights into Nickel-Catalyzed Carbonylation
The core of this innovative synthesis lies in the intricate catalytic cycle driven by nickel triflate in conjunction with a bipyridine ligand and molybdenum hexacarbonyl as the carbon monoxide source. The reaction initiates with the oxidative addition of the sulfonyl chloride to the low-valent nickel center, generating a reactive nickel-sulfur species. Subsequently, carbon monoxide, released in situ from the molybdenum carbonyl precursor, inserts into the nickel-carbon or nickel-sulfur bond, forming an acyl-nickel intermediate. This step is critical as it constructs the carbonyl functionality of the thioester backbone. The presence of zinc iodide acts as a Lewis acid promoter, facilitating the activation of the sulfonyl chloride and stabilizing key intermediates within the catalytic cycle. The final step involves transmetallation with the arylboronic acid followed by reductive elimination to release the desired thioester product and regenerate the active nickel catalyst.
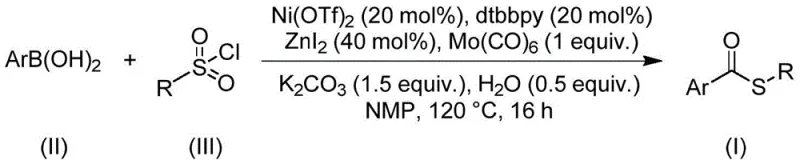
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or thiol-oxidation pathways. The controlled nature of the transition metal catalysis ensures high chemoselectivity, minimizing the formation of side products such as symmetrical disulfides or over-oxidized sulfones. The use of water as a mild additive in the reaction mixture further aids in maintaining the stability of the boronic acid species while participating in the proton transfer steps necessary for catalyst turnover. For R&D teams, understanding this mechanistic nuance is vital for troubleshooting and optimizing the process for specific substrates. The robustness of the nickel cycle allows for the tolerance of electron-withdrawing and electron-donating groups on both the arylboronic acid and the sulfonyl chloride components, ensuring consistent quality and purity profiles essential for regulatory compliance in pharmaceutical applications.
How to Synthesize Thioester Compound Efficiently
The practical implementation of this synthesis is designed for scalability and ease of operation, making it highly suitable for contract development and manufacturing organizations. The standard protocol involves charging a reaction vessel with the nickel catalyst system, including nickel triflate and the dtbbpy ligand, along with the carbonyl source and promoters. The reactants, arylboronic acid and sulfonyl chloride, are then introduced into an aprotic organic solvent such as N-methylpyrrolidone (NMP), which has been identified as the optimal medium for solubilizing all components and promoting high conversion rates. The mixture is heated to a moderate temperature of 120°C and maintained for approximately 16 hours. Detailed standardized synthesis steps for specific derivatives are provided in the guide below.
- Combine nickel triflate, dtbbpy ligand, molybdenum carbonyl, zinc iodide, potassium carbonate, water, arylboronic acid, and sulfonyl chloride in NMP solvent.
- Heat the reaction mixture to 120°C and maintain stirring for 16 hours to ensure complete conversion.
- Perform post-treatment involving filtration and silica gel mixing, followed by column chromatography purification to isolate the pure thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nickel-catalyzed thioester synthesis offers compelling strategic advantages that extend beyond mere chemical novelty. The shift from specialized, hazardous thiols to commodity sulfonyl chlorides fundamentally alters the cost structure and risk profile of the supply chain. By utilizing raw materials that are produced on a multi-ton scale for other industries, manufacturers can secure more stable pricing and avoid the supply bottlenecks often associated with fine chemical sulfur sources. Furthermore, the elimination of malodorous reagents reduces the burden on facility ventilation and waste treatment systems, leading to significant operational expenditure savings. This process aligns perfectly with the goals of enhancing supply chain reliability and achieving cost reduction in pharmaceutical intermediate manufacturing through intelligent process design.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and difficult-to-handle thiols with inexpensive sulfonyl chlorides. Since sulfonyl chlorides are mass-produced industrial chemicals, their unit cost is significantly lower than that of specialized thiols, directly impacting the bill of materials. Additionally, the high catalytic efficiency means that precious metal loading can be optimized, and the absence of external oxidants or reductants reduces the consumption of auxiliary reagents. The simplified workup procedure, which avoids complex quenching steps required for reactive sulfur species, further lowers labor and utility costs, contributing to substantial overall cost savings in the production of thioester intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of the starting materials. Arylboronic acids and sulfonyl chlorides are stocked by numerous global chemical suppliers, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of functional groups, means that the process is less sensitive to minor variations in raw material quality, ensuring consistent batch-to-batch performance. This reliability is critical for maintaining continuous production schedules and meeting tight delivery windows for downstream API synthesis. By mitigating the risks associated with hazardous material transport and storage, companies can also streamline their logistics operations and reduce insurance premiums related to chemical handling.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and eventually to tonnage is facilitated by the use of standard reactor equipment and common solvents like NMP. The reaction does not generate gaseous byproducts or highly toxic waste streams, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The atom economy of the carbonylation reaction is favorable, as the carbonyl group is efficiently incorporated into the product structure. For sustainability-focused organizations, this method represents a greener alternative to traditional thioester synthesis, supporting corporate social responsibility goals while maintaining high production throughput and minimizing the environmental footprint of chemical manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thioester synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is sulfonyl chloride preferred over thiols for thioester synthesis?
A: Sulfonyl chlorides are odorless, stable, and commercially available commodities, whereas thiols are notoriously malodorous and prone to poisoning transition metal catalysts, making sulfonyl chlorides superior for large-scale manufacturing.
Q: What is the typical yield range for this nickel-catalyzed process?
A: The patented method demonstrates high reaction efficiency, with isolated yields typically ranging from 60% to 90% across a diverse array of substrates including phenyl, thienyl, and naphthyl derivatives.
Q: Does this method require expensive oxidants or reductants?
A: No, a key advantage of this protocol is that it operates without the need for additional external oxidants or reductants, simplifying the reaction profile and reducing waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into viable industrial processes. We are committed to delivering high-purity thioester compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to implement the nickel-catalyzed carbonylation process described in CN112239384B allows us to offer competitive pricing and reliable supply for this important class of pharmaceutical intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this sulfonyl chloride-based protocol. We encourage you to contact us today to obtain specific COA data for our catalog items and to receive comprehensive route feasibility assessments tailored to your target molecules, ensuring a seamless path from concept to commercial reality.
