Advanced Room-Temperature Synthesis of Quinoxaline Derivatives for Commercial Scale-Up
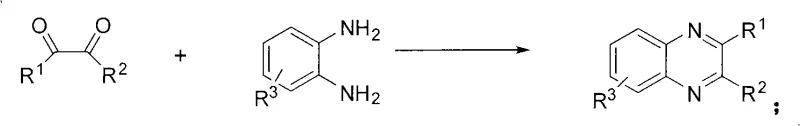
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic pathways, particularly for high-value scaffolds like quinoxaline derivatives. As detailed in patent CN101318938A, a groundbreaking methodology has emerged that challenges the conventional reliance on harsh thermal conditions and toxic reagents. This innovation introduces a robust catalytic system utilizing metal trifluoromethanesulfonates to facilitate the condensation of 1,2-diketones with aryl 1,2-diamines. For R&D directors and procurement strategists in the pharmaceutical and fine chemical sectors, this represents a pivotal shift from energy-intensive batch processes to rapid, ambient-temperature transformations. The patent explicitly highlights the capability to achieve exceptional yields ranging from over 90% up to 100% while operating under remarkably mild conditions, setting a new benchmark for process efficiency in the production of benzopyrazine heterocycles.
Furthermore, the environmental and economic implications of this technology cannot be overstated. Traditional syntheses often suffer from low atom economy and generate substantial waste due to the use of volatile organic compounds and non-recyclable catalysts. In contrast, the disclosed method employs benign solvents like ethanol or water and features a catalyst system that can be reclaimed and reused approximately 10 times. This level of catalyst stability not only drastically reduces the cost of goods sold (COGS) but also simplifies the downstream processing workflow. By eliminating the need for prolonged heating and complex purification steps, this approach offers a compelling value proposition for manufacturers seeking to optimize their supply chains for critical intermediates used in drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoxaline derivatives has been plagued by significant operational inefficiencies that hinder large-scale commercial viability. The classical approach typically involves refluxing 1,2-diketones and aryl 1,2-diamines in solvents such as ethanol or acetic acid for extended periods, often ranging from 2 to 12 hours. Despite this substantial energy investment, the resulting yields are frequently inconsistent and mediocre, typically falling within the range of 34% to 85%. Moreover, many alternative literature methods rely on the use of toxic and expensive noble metal catalysts, such as palladium or ruthenium complexes, which pose severe challenges for residual metal control in pharmaceutical intermediates. The reliance on hazardous organic solvents further exacerbates environmental concerns, necessitating costly waste treatment protocols and increasing the overall carbon footprint of the manufacturing process.
Beyond the immediate reaction parameters, the post-treatment procedures associated with conventional methods are often cumbersome and labor-intensive. Catalysts used in older methodologies are frequently destroyed during workup or lose their activity, rendering them single-use reagents that drive up raw material costs. Additionally, the requirement for high temperatures and inert gas protection adds layers of complexity to reactor operations, increasing the risk of safety incidents and equipment failure. For supply chain managers, these factors translate into longer lead times, higher production costs, and greater vulnerability to regulatory scrutiny regarding solvent emissions and heavy metal contamination. The cumulative effect of these limitations creates a bottleneck for the reliable supply of high-purity quinoxaline derivatives needed for advanced therapeutic and electronic applications.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally redefines the synthesis paradigm by leveraging the potent Lewis acidity of metal trifluoromethanesulfonates. This novel approach enables the reaction to proceed at room temperature, completely eliminating the need for external heating or inert gas protection. The reaction kinetics are remarkably fast, with completion achieved within a mere 5 to 15 minutes of stirring. This dramatic reduction in reaction time not only increases throughput capacity but also minimizes the formation of thermal degradation byproducts, thereby enhancing the purity profile of the crude product. The use of earth-abundant or readily available metal salts, such as gallium, iron, or zinc triflates, replaces the dependency on scarce precious metals, aligning the process with principles of sustainable chemistry and resource conservation.
From a processing standpoint, the simplicity of the workup procedure is a major advantage. Upon completion, the product often crystallizes directly from the reaction mixture upon cooling, allowing for isolation via simple filtration. The catalyst remains dissolved in the mother liquor, facilitating its recovery and subsequent reuse for up to 10 cycles without significant loss of performance. This closed-loop catalyst usage significantly lowers the effective catalyst cost per kilogram of product. Furthermore, the compatibility with green solvents like water and ethanol removes the regulatory burden associated with volatile organic compound (VOC) emissions. For manufacturers, this translates to a streamlined operation that requires less specialized equipment, reduces energy consumption, and delivers a superior quality product with minimal environmental impact, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Metal Triflate-Catalyzed Cyclization
The efficacy of this synthesis relies on the unique electronic properties of the metal trifluoromethanesulfonate catalyst, which acts as a powerful Lewis acid to activate the carbonyl groups of the 1,2-diketone substrate. The metal center coordinates with the oxygen atoms of the diketone, increasing the electrophilicity of the carbonyl carbons and making them more susceptible to nucleophilic attack by the amino groups of the aryl 1,2-diamine. This activation lowers the energy barrier for the initial condensation step, allowing the reaction to proceed rapidly even at ambient temperatures. The triflate anion is non-coordinating and highly stable, ensuring that the metal center remains available for catalysis throughout the cycle without being deactivated by side reactions or solvent interactions. This robust coordination chemistry is key to the observed high turnover numbers and the ability to maintain catalytic activity over multiple reuse cycles.
Impurity control is inherently managed through the mildness of the reaction conditions. In traditional thermal methods, high temperatures can promote side reactions such as polymerization of the diketone or oxidation of the amine, leading to complex impurity profiles that are difficult to separate. By conducting the reaction at room temperature, these thermal degradation pathways are effectively suppressed. The rapid kinetics ensure that the desired cyclization to form the quinoxaline ring occurs preferentially before any competing reactions can take place. Additionally, the precipitation of the product upon cooling serves as a built-in purification step, as many potential impurities remain soluble in the ethanol or water solvent system. This mechanistic advantage ensures that the final product meets stringent purity specifications required for sensitive applications in medicinal chemistry and organic electronics.
How to Synthesize Quinoxaline Derivatives Efficiently
The practical implementation of this technology is straightforward and adaptable to existing manufacturing infrastructure. The process begins by dissolving the 1,2-diketone and aryl 1,2-diamine reactants in a chosen solvent, typically ethanol or water, maintaining a stoichiometric ratio between 1:1 and 1:1.01 to ensure complete conversion of the limiting reagent. A catalytic amount of the metal triflate, usually around 1 mol% relative to the diketone, is then introduced to the mixture. The reaction vessel is stirred at room temperature, and monitoring indicates that conversion is typically complete within 5 to 15 minutes. Following the reaction, the mixture is cooled, often in an ice-salt bath, to induce crystallization of the product. The solid is collected by filtration, washed, and if necessary, recrystallized to achieve the desired purity levels. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve 1,2-diketone and aryl 1,2-diamine in ethanol or water with a molar ratio of 1: 1 to 1:1.01.
- Add a catalytic amount of metal trifluoromethanesulfonate (e.g., Gallium triflate) to the mixture.
- Stir at room temperature for 5-15 minutes, then cool, filter, and recrystallize to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers transformative benefits that extend far beyond simple yield improvements. The primary driver of value is the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By shifting from a multi-hour reflux process to a 15-minute room-temperature stir, facility throughput can be increased significantly without the need for capital investment in new reactors. The elimination of heating requirements leads to substantial energy savings, while the use of water or ethanol as solvents reduces the costs associated with solvent purchase, recovery, and hazardous waste disposal. These factors combine to create a leaner, more cost-effective production model that enhances competitiveness in the global market for fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this method is profound, primarily driven by the recyclability of the catalyst and the efficiency of the reaction. Since the metal triflate catalyst can be recovered and reused approximately 10 times, the amortized cost of the catalyst per batch becomes negligible compared to traditional single-use systems. Furthermore, the high yields, often reaching 100%, mean that raw material waste is minimized, maximizing the return on investment for expensive starting materials like substituted diketones. The simplified workup, which often requires only filtration and recrystallization, reduces labor hours and consumable costs associated with chromatography or extensive extraction procedures. This holistic reduction in variable costs allows for more aggressive pricing strategies while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and speed of this synthetic route. The short reaction time of 5 to 15 minutes allows for rapid response to fluctuating market demands, enabling manufacturers to produce batches on a just-in-time basis. The use of commodity solvents like ethanol and water eliminates supply risks associated with specialized or regulated organic solvents. Additionally, the stability of the catalyst ensures consistent batch-to-batch quality, reducing the incidence of failed runs and production delays. This reliability is crucial for maintaining continuous supply lines to downstream customers in the pharmaceutical industry, where interruptions can have cascading effects on drug development timelines and regulatory filings.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is seamless due to the absence of exothermic hazards and the use of non-flammable or low-flammability solvents. The mild conditions reduce the engineering controls required for heat management, making it easier to transition from pilot plants to multi-ton production facilities. From an environmental perspective, the process aligns perfectly with increasingly stringent global regulations on industrial emissions and waste. The use of green solvents and the minimization of heavy metal waste through catalyst recycling simplify the permitting process and reduce the liability associated with environmental compliance. This sustainability profile enhances the brand reputation of the manufacturer and meets the growing demand for eco-friendly supply chains from multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoxaline synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing production workflows or for procurement specialists assessing the quality and consistency of the supplied intermediates.
Q: What are the advantages of using metal trifluoromethanesulfonate catalysts over traditional methods?
A: Unlike traditional methods requiring reflux for 2-12 hours with yields of 34-85%, this catalytic method operates at room temperature for 5-15 minutes with yields exceeding 90%, often reaching 100%.
Q: Is the catalyst reusable in this quinoxaline synthesis process?
A: Yes, the metal trifluoromethanesulfonate catalyst is stable and can be recovered and reused approximately 10 times without significant loss of catalytic activity, significantly reducing material costs.
Q: What solvents are compatible with this green synthesis route?
A: The process utilizes environmentally benign solvents such as ethanol or water, eliminating the need for toxic organic solvents and simplifying waste treatment and environmental compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoxaline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern chemical industry. Our team of expert chemists has thoroughly analyzed the potential of the metal triflate-catalyzed method described in CN101318938A and is fully equipped to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is smooth and predictable. Our state-of-the-art rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of quinoxaline derivatives we deliver meets the highest international standards for pharmaceutical and electronic grade materials.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthesis method. Whether you require custom synthesis of specific substituted quinoxalines or support in optimizing your current supply chain for cost and efficiency, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can drive value and reliability in your supply chain.
