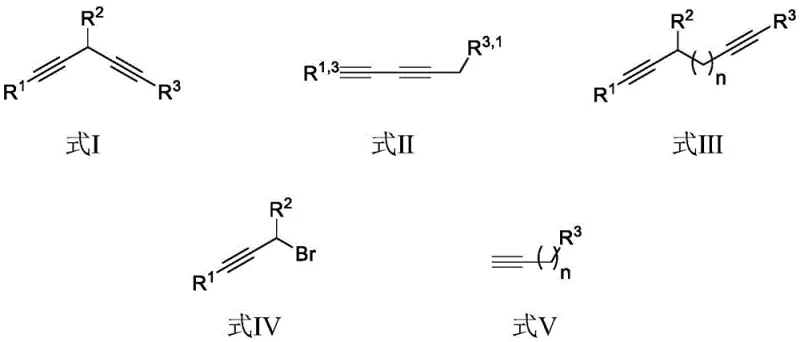
Scalable Copper-Catalyzed Diyne Synthesis for Advanced Pharmaceutical and Material Intermediates
Scalable Copper-Catalyzed Diyne Synthesis for Advanced Pharmaceutical and Material Intermediates
The landscape of organic synthesis for conjugated systems is undergoing a significant transformation, driven by the demand for more sustainable and cost-effective methodologies. Patent CN111662147B introduces a robust and versatile protocol for the preparation of various diynes and their analogues, addressing critical bottlenecks in the manufacturing of pharmaceutical intermediates and electronic materials. This technology leverages a monovalent copper catalytic system to couple propargyl bromides with terminal alkynes, offering a distinct advantage over traditional noble metal-catalyzed routes. For R&D directors and procurement specialists, this represents a pivotal opportunity to optimize supply chains for high-value conjugated building blocks. The method operates under remarkably mild conditions, utilizing potassium phosphate as a base in N,N-dimethylacetamide (DMAc) at just 40°C, which drastically reduces energy consumption and thermal stress on sensitive functional groups. By eliminating the need for expensive gold or palladium catalysts and avoiding the harsh conditions of classical coupling reactions, this process aligns perfectly with modern green chemistry principles while ensuring economic viability for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,3-diyne and 1,4-diyne scaffolds has relied heavily on methods such as Glaser-Hay coupling and Cadiot-Chodkiewicz coupling, which present substantial operational and economic challenges for industrial scale-up. The Glaser-Hay coupling, while effective for symmetrical diynes, suffers from poor selectivity when attempting to synthesize asymmetric variants, often leading to statistical mixtures of homocoupled byproducts that are difficult and costly to separate. Furthermore, the Cadiot-Chodkiewicz reaction requires the pre-functionalization of terminal alkynes into unstable alkynyl bromides, introducing an additional synthetic step that lowers overall atom economy and increases safety risks due to the instability of the intermediates. Alternative approaches utilizing gold catalysts have emerged to improve efficiency, but the prohibitive cost of gold and the necessity for specialized ligands render these methods economically unfeasible for the production of bulk fine chemicals or commodity pharmaceutical intermediates. Additionally, many traditional protocols require strongly basic organometallic reagents or high temperatures, which limit substrate scope and complicate waste treatment processes, thereby inflating the total cost of ownership for manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN111662147B offers a streamlined, direct coupling strategy that bypasses the need for pre-functionalized unstable intermediates or precious metal catalysts. This novel approach utilizes readily available propargyl bromide compounds and terminal alkynes as direct substrates, mediated by an inexpensive monovalent copper catalyst such as CuBr or CuI. The reaction proceeds efficiently in a polar aprotic solvent like DMAc with potassium phosphate serving as a mild, non-corrosive base, creating a reaction environment that is both safer and more environmentally benign. As illustrated in the general reaction scheme below, this method is highly adaptable, capable of producing diverse structural motifs including 1,4-diynes, asymmetric 1,3-diynes, and extended conjugated systems simply by varying the starting materials. The mild thermal requirement of 40°C not only preserves sensitive functional groups but also significantly lowers the energy footprint of the manufacturing process, making it an ideal candidate for sustainable industrial application.
Mechanistic Insights into Cu-Catalyzed Cross-Coupling
The mechanistic elegance of this copper-catalyzed transformation lies in its ability to activate the terminal alkyne C-H bond under mild basic conditions without generating highly reactive and dangerous acetylide salts typically associated with stronger bases. The monovalent copper species coordinates with the terminal alkyne to form a copper-acetylide intermediate in situ, which then undergoes a nucleophilic substitution with the propargyl bromide electrophile. This pathway avoids the radical mechanisms often seen in oxidative homocouplings, thereby inherently suppressing the formation of symmetrical diyne byproducts that plague Glaser-type reactions. The use of potassium phosphate is critical here; it is strong enough to deprotonate the copper-coordinated alkyne but mild enough to prevent side reactions such as elimination or polymerization of the propargyl bromide. This precise balance ensures high chemoselectivity, allowing for the successful coupling of substrates bearing diverse electronic properties, from electron-rich methyl-substituted aromatics to electron-deficient fluoro-derivatives, without compromising yield or purity.
From an impurity control perspective, this mechanism offers significant advantages for pharmaceutical manufacturing where strict limits on heavy metals and organic impurities are enforced. Since the catalyst loading is relatively low (10-20 mol%) and the copper species remains soluble in the polar reaction medium, post-reaction removal is straightforward using standard aqueous workups and chelating agents, unlike heterogeneous palladium catalysts which can leach or become trapped in product matrices. The reaction's tolerance for various substituents is exemplified by the successful synthesis of complex asymmetric diynes, such as the structure shown below, which features multiple aromatic rings and alkyl substitutions. This structural diversity confirms that the catalytic cycle is robust against steric hindrance and electronic variation, providing R&D teams with a reliable platform for generating libraries of conjugated intermediates for drug discovery or material science applications without the need for extensive method re-optimization.
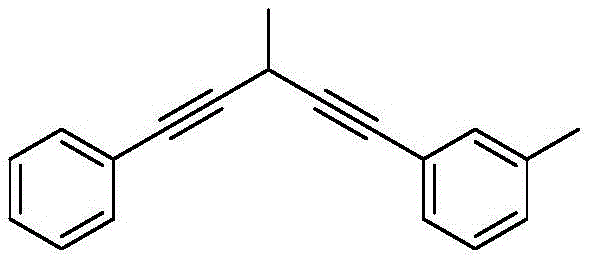
How to Synthesize 1,5-Diphenyl-1,4-pentadiyne Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard laboratory equipment and commercially available reagents that facilitate rapid technology transfer from bench to pilot plant. The protocol involves a simple one-pot procedure where the propargyl bromide, terminal alkyne, copper catalyst, and base are combined in DMAc under an inert nitrogen atmosphere to prevent oxidative degradation of the catalyst or substrates. Detailed standardized operating procedures regarding exact stoichiometric ratios, addition rates, and specific workup parameters are critical for ensuring consistent batch-to-batch reproducibility and maximizing yield. For a comprehensive understanding of the operational parameters and safety considerations required for scaling this reaction, please refer to the detailed synthesis guide provided below.
- Charge a reaction vessel with propargyl bromide derivative, terminal alkyne, CuBr catalyst (10-20 mol%), and potassium phosphate base in DMAc solvent.
- Maintain the reaction mixture under a nitrogen atmosphere at a mild temperature of 40°C for approximately 16 hours with continuous stirring.
- Quench with saturated ammonium chloride, extract with ethyl acetate, dry over magnesium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed methodology translates directly into tangible improvements in cost structure and supply reliability. The shift from precious metal catalysts like gold or palladium to abundant copper salts results in a drastic reduction in raw material costs, removing the volatility associated with noble metal markets from the production budget. Furthermore, the elimination of pre-functionalization steps, such as the separate synthesis of unstable alkynyl bromides or organometallic reagents, simplifies the supply chain by reducing the number of discrete starting materials that need to be sourced, qualified, and stored. This consolidation of the synthetic route not only lowers the direct cost of goods sold (COGS) but also reduces the logistical complexity and lead times associated with multi-step sequences, enabling faster response to market demands for key intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive catalytic systems with low-cost copper salts and the use of inexpensive inorganic bases like potassium phosphate. By avoiding the need for specialized ligands or cryogenic conditions often required for organolithium or Grignard reagents, the process significantly lowers utility costs related to cooling and energy consumption. Additionally, the high atom economy of the direct coupling reaction minimizes waste generation, which in turn reduces the costs associated with solvent disposal and environmental compliance, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials such as propargyl bromides and terminal alkynes ensures a robust supply chain that is less susceptible to disruptions compared to routes requiring custom-synthesized or unstable intermediates. The mild reaction conditions (40°C) allow for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure or high-temperature equipment, increasing the number of potential contract manufacturing organizations (CMOs) capable of executing the process. This flexibility enhances supply security by diversifying the pool of qualified suppliers and reducing the risk of production bottlenecks caused by equipment limitations or safety constraints.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic profile being manageable at low temperatures, which mitigates safety risks during large-batch production and simplifies heat transfer requirements. The use of DMAc as a solvent, while requiring careful handling, is well-established in the industry with known recovery and recycling protocols, supporting sustainable manufacturing practices. Moreover, the absence of toxic tin or zinc byproducts, common in traditional Stille or Negishi couplings, simplifies the purification process and ensures that the final product meets stringent regulatory standards for residual metals, facilitating smoother registration and approval processes for downstream pharmaceutical applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diyne synthesis technology, based on the specific data and embodiments disclosed in the patent literature. These insights are intended to clarify the operational feasibility and strategic value of adopting this method for your specific production needs. For further technical discussions or custom feasibility studies, our team is available to provide detailed analysis tailored to your project requirements.
Q: What are the advantages of using CuBr over Gold catalysts for diyne synthesis?
A: The patented process utilizes inexpensive monovalent copper (CuBr) instead of costly gold catalysts, significantly reducing raw material costs while maintaining high yields and selectivity under mild conditions (40°C).
Q: Can this method tolerate diverse functional groups on the aromatic rings?
A: Yes, the method demonstrates excellent substrate scope, successfully accommodating electron-donating groups like methyl and electron-withdrawing groups like fluoro on both the propargyl bromide and terminal alkyne components.
Q: How is the reaction workup handled to ensure high purity?
A: The process employs a standard aqueous workup followed by silica gel column chromatography. For solid products, recrystallization using dichloromethane and n-hexane is recommended to achieve stringent purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diynes Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating the development of next-generation therapeutics and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and compliant. We are equipped with rigorous QC labs and state-of-the-art analytical capabilities to guarantee stringent purity specifications for all our conjugated intermediates, adhering to the highest international standards for pharmaceutical and electronic grade chemicals. Our commitment to process innovation allows us to offer competitive pricing without compromising on quality or delivery performance.
We invite you to collaborate with us to leverage this advanced copper-catalyzed technology for your upcoming projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient route for your specific target molecules. Please contact us today to obtain specific COA data for our existing diyne inventory or to discuss route feasibility assessments for your custom synthesis needs, and let us help you optimize your supply chain for the future.
