Advanced Catalytic Synthesis of Diynes: Technical Breakthroughs and Commercial Scalability
The landscape of organic synthesis is constantly evolving, driven by the need for more efficient and sustainable pathways to complex molecular architectures. A significant advancement in this domain is detailed in patent CN111662147B, which discloses a robust method for preparing various diynes and their analogues. This technology addresses long-standing challenges in constructing conjugated alkyne systems, which are pivotal building blocks in the synthesis of pharmaceuticals, agrochemicals, and advanced electronic materials. By leveraging a monovalent copper catalyst system under remarkably mild conditions, this process offers a streamlined alternative to traditional coupling methods. For R&D directors and procurement specialists alike, understanding the nuances of this patented methodology is crucial for optimizing supply chains and reducing the cost of goods sold for high-value intermediates. The ability to access diverse diyne scaffolds with high purity and yield positions this technology as a cornerstone for modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
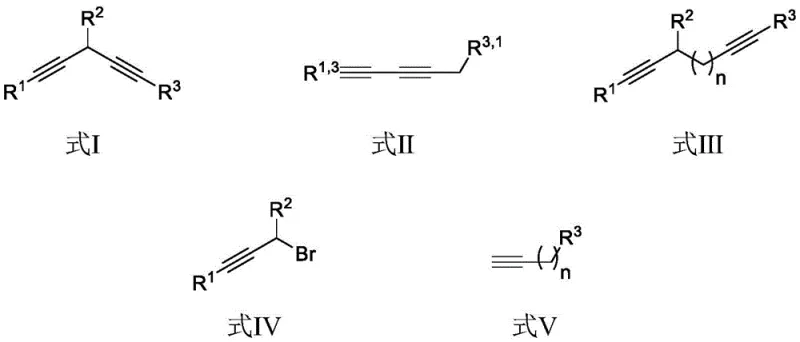
Historically, the synthesis of asymmetric 1,3-diynes and related structures has been plagued by significant inefficiencies inherent to classical coupling strategies. The Glaser-Hay coupling reaction, while foundational, typically necessitates a large excess of alkyne substrates to mitigate the pervasive issue of homocoupling side reactions, leading to poor atom economy and difficult purification processes. Furthermore, the Cadiot-Chodkiewicz coupling, another staple in alkyne chemistry, requires the pre-functionalization of terminal alkynes into unstable alkynyl bromides, introducing additional synthetic steps and safety hazards associated with handling reactive halides. Recent developments involving gold catalysts have offered improved efficiency but at the prohibitive cost of expensive noble metals and specialized ligands, rendering them economically unviable for large-scale industrial applications. These conventional routes often demand harsh reaction conditions, including strong bases or elevated temperatures, which can compromise the integrity of sensitive functional groups present in complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a transformative approach that circumvents these historical bottlenecks through a direct coupling of propargyl bromide compounds and terminal alkynes. This novel route employs a monovalent copper catalyst, specifically copper(I) bromide, in conjunction with potassium phosphate as a mild base, operating effectively in polar aprotic solvents like N,N-dimethylacetamide. The reaction proceeds smoothly at a low temperature of 40°C under a nitrogen atmosphere, drastically reducing energy consumption and thermal stress on the reactants. This温和 condition not only preserves sensitive substituents but also simplifies the operational requirements for reactor setups. By eliminating the need for pre-formed organometallic reagents like lithium or magnesium alkynyls, the process achieves superior atom economy and reduces the generation of hazardous waste streams. The versatility of this system allows for the synthesis of 1,3-diynes, 1,4-diynes, and even extended 1,n-diynes, providing a unified platform for accessing a wide array of conjugated structures essential for drug discovery and material science.
Mechanistic Insights into Cu-Catalyzed Cross-Coupling
The efficacy of this synthesis relies on the precise orchestration of a copper-catalyzed cross-coupling mechanism that facilitates the formation of carbon-carbon bonds between sp-hybridized carbons. The monovalent copper species acts as a soft Lewis acid, coordinating with the terminal alkyne to generate a reactive copper-acetylide intermediate in situ. This activation lowers the pKa barrier for deprotonation by the mild phosphate base, avoiding the need for aggressive alkali metals. Subsequently, the nucleophilic copper-acetylide attacks the electrophilic propargyl bromide substrate, likely proceeding through an SN2-type displacement or a copper-mediated oxidative addition-reductive elimination cycle depending on the specific electronic nature of the substrates. The use of potassium phosphate is particularly strategic, as it provides sufficient basicity to drive the formation of the acetylide without promoting elimination side reactions that could degrade the propargyl bromide. This delicate balance ensures high selectivity for the desired cross-coupled product over potential homocoupling or polymerization byproducts.
From an impurity control perspective, the choice of reagents and conditions plays a pivotal role in ensuring the high purity required for pharmaceutical applications. The mild reaction temperature of 40°C minimizes thermal degradation pathways that often lead to complex impurity profiles in high-temperature couplings. Furthermore, the use of DMAc as a solvent ensures excellent solubility for both organic substrates and inorganic salts, promoting homogeneous reaction kinetics and preventing localized hot spots that could trigger side reactions. The workup procedure described involves standard aqueous extraction and silica gel chromatography, which effectively removes residual copper salts and inorganic bases. The patent data indicates that recrystallization can be employed for solid products to achieve single-crystal purity, demonstrating the robustness of the purification protocol. This level of control over the impurity profile is critical for regulatory compliance in API manufacturing, where strict limits on heavy metals and organic impurities must be maintained.
How to Synthesize Diynes Efficiently
The practical implementation of this synthesis route is designed for ease of operation while maintaining rigorous control over reaction parameters to ensure reproducibility and high yield. The process begins with the precise weighing of propargyl bromide and terminal alkyne substrates, followed by the addition of the copper catalyst and phosphate base in the chosen solvent. Maintaining an inert nitrogen atmosphere is essential to prevent oxidation of the copper catalyst and the alkyne substrates, which could otherwise lead to catalyst deactivation or oxidative coupling byproducts. The reaction mixture is then stirred at the optimized temperature, allowing sufficient time for the conversion to reach completion as monitored by thin-layer chromatography. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Mix propargyl bromide compound IV and terminal alkyne compound V with potassium phosphate base and CuBr catalyst in DMAc solvent.
- Maintain the reaction mixture under a nitrogen atmosphere at a controlled temperature of 40°C for approximately 16 hours.
- Perform workup by extracting with ethyl acetate, drying over magnesium sulfate, and purifying via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology translates into tangible strategic benefits that extend beyond mere chemical yield. The shift away from expensive noble metal catalysts like gold and the elimination of unstable organometallic reagents significantly lowers the raw material costs associated with diyne production. This cost structure improvement is compounded by the reduced need for specialized handling equipment, as the mild reaction conditions do not require cryogenic cooling or high-pressure reactors. Consequently, the overall cost of manufacturing is substantially reduced, enhancing the competitiveness of the final product in the global market. Additionally, the simplified workflow reduces the operational burden on production teams, allowing for faster turnaround times and more flexible scheduling.
- Cost Reduction in Manufacturing: The replacement of costly gold catalysts with inexpensive copper salts represents a direct and significant reduction in catalyst expenditure. Furthermore, the avoidance of pre-functionalization steps, such as the synthesis of alkynyl bromides or organolithium reagents, eliminates entire unit operations from the process flow. This streamlining reduces labor costs, solvent consumption, and waste disposal fees, leading to a leaner and more cost-effective manufacturing process. The high atom economy of the reaction ensures that a greater proportion of raw materials are converted into the desired product, minimizing waste and maximizing resource utilization.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as propargyl bromides and terminal alkynes, mitigates the risk of supply chain disruptions often associated with specialized or hazardous reagents. The robustness of the reaction conditions allows for sourcing from a broader range of suppliers, increasing negotiation leverage and ensuring continuity of supply. Moreover, the mild nature of the process reduces the safety risks associated with transportation and storage of reactive intermediates, simplifying logistics and compliance with hazardous material regulations. This stability is crucial for maintaining consistent production schedules and meeting delivery commitments to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently with various substrates without the need for extensive re-optimization. The use of mild bases and the absence of toxic heavy metals simplify the wastewater treatment process, aligning with increasingly stringent environmental regulations. The reduced energy footprint, owing to the low reaction temperature, contributes to a lower carbon footprint for the manufacturing facility. These factors collectively enhance the sustainability profile of the production process, making it attractive for companies aiming to meet corporate social responsibility goals and regulatory standards for green chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diyne synthesis technology. These insights are derived directly from the patent specifications and experimental data, providing a reliable foundation for decision-making. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows. The answers cover critical areas such as substrate scope, catalyst loading, and purification strategies.
Q: What are the advantages of this copper-catalyzed method over traditional Glaser-Hay coupling?
A: Unlike Glaser-Hay coupling which suffers from alkyne self-coupling side reactions requiring excess reagents, this method utilizes propargyl bromides and terminal alkynes directly, offering better atom economy and selectivity for asymmetric diynes.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process operates at mild temperatures (40°C) and uses common reagents like CuBr and K3PO4, eliminating the need for cryogenic conditions or expensive gold catalysts, which significantly enhances scalability and safety.
Q: What types of diyne structures can be synthesized using this protocol?
A: The method is versatile, capable of producing 1,3-diynes, 1,4-diynes, and extended 1,n-diynes with various aryl, heteroaryl, and alkyl substituents, making it ideal for diverse organic synthesis applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diynes Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis route for the production of high-value diyne intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of diynes delivered meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this process further, tailoring it to your specific volume and quality requirements.
We invite you to collaborate with us to unlock the full commercial potential of your diyne-based projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this greener, more efficient synthesis method. Please contact us to request specific COA data for our reference standards and comprehensive route feasibility assessments. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and driving your supply chain towards greater efficiency and sustainability.
