Advanced Palladium-Catalyzed Multi-Component Synthesis of Carbonyl-Bridged Biheterocyclic Intermediates
Introduction to Patent CN115353511A
The pharmaceutical industry continuously seeks robust methodologies for constructing complex heterocyclic scaffolds, which serve as the fundamental backbone for numerous bioactive molecules. Patent CN115353511A introduces a groundbreaking multi-component synthesis strategy for preparing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technological advancement addresses critical bottlenecks in modern medicinal chemistry by enabling the efficient assembly of these privileged structures through a single-pot palladium-catalyzed carbonylation cascade. The significance of this invention lies not only in its chemical elegance but also in its operational simplicity, utilizing readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives. By eliminating the need for toxic carbon monoxide gas and harsh reaction conditions, this process represents a paradigm shift towards safer and more sustainable manufacturing of high-value pharmaceutical intermediates.
For research and development teams focused on oncology or infectious disease therapeutics, the ability to rapidly access diversified libraries of trifluoromethyl-substituted biheterocycles is invaluable. The patent details a protocol that operates at a remarkably mild temperature of 30°C, ensuring the integrity of sensitive functional groups that might otherwise degrade under traditional thermal stress. Furthermore, the compatibility with a wide range of substituents—including halogens, alkyls, and electron-withdrawing groups—provides medicinal chemists with the flexibility to optimize structure-activity relationships (SAR) without being constrained by synthetic limitations. This report analyzes the technical merits and commercial implications of adopting this novel pathway for the production of reliable pharmaceutical intermediate supplies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems containing both indolinone and imidazole rings has been fraught with synthetic challenges. Traditional approaches often rely on the direct coupling of two pre-formed heterocyclic substrates, a strategy that frequently suffers from low atom economy and poor regioselectivity. Alternatively, oxidative cyclization reactions involving bis-nucleophilic substrates require stoichiometric amounts of oxidants and often necessitate elevated temperatures that can lead to decomposition of thermally labile intermediates. Perhaps the most significant barrier in conventional carbonylation strategies is the reliance on gaseous carbon monoxide, which poses severe safety hazards regarding toxicity and flammability, requiring specialized high-pressure equipment that increases capital expenditure and operational complexity. These legacy methods often result in prolonged reaction times, difficult purification processes due to side-product formation, and limited scope regarding substrate tolerance, thereby hindering the rapid scale-up required for clinical supply chains.
The Novel Approach
In stark contrast, the methodology disclosed in CN115353511A leverages a transition metal palladium-catalyzed cascade reaction that seamlessly integrates three distinct components into a unified molecular architecture. This one-pot protocol utilizes a safe, liquid-based carbon monoxide surrogate generated in situ from formic acid and acetic anhydride, effectively mitigating the risks associated with handling toxic CO gas. The reaction is initiated by the activation of an aryl iodide bond within the acrylamide substrate, followed by an intramolecular Heck-type cyclization to form the indolinone core. Subsequently, the palladium species undergoes carbonylation and reacts with the trifluoroacetamidine intermediate, formed concurrently from the condensation of trifluoroethylimidoyl chloride and propargylamine. This tandem sequence allows for the simultaneous formation of multiple carbon-carbon and carbon-nitrogen bonds with high efficiency.
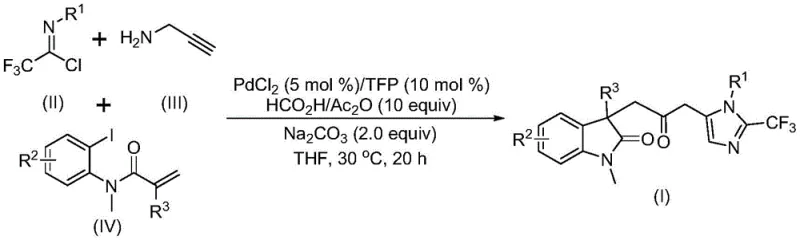
The visual representation of this transformation highlights the convergence of three simple building blocks into a complex carbonyl-bridged biheterocyclic scaffold (Structure I). This convergent synthesis strategy drastically reduces the number of isolation steps compared to linear syntheses, thereby improving overall yield and reducing waste generation. The use of trifluoroethylimidoyl chloride as a precursor introduces the trifluoromethyl group directly, a modification known to enhance metabolic stability and lipophilicity in drug candidates. By operating at 30°C in tetrahydrofuran (THF), the process ensures excellent functional group compatibility, allowing for the incorporation of diverse substituents on the aromatic rings without compromising reaction efficiency. This novel approach not only simplifies the synthetic route but also aligns with green chemistry principles by minimizing hazardous reagents and energy consumption.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
A deep understanding of the catalytic cycle is essential for optimizing this process for commercial scale-up. The mechanism likely initiates with the reduction of the palladium(II) precatalyst, palladium chloride (PdCl2), to the active palladium(0) species in the presence of the electron-rich tris(2-furyl)phosphine (TFP) ligand. This active Pd(0) complex undergoes oxidative addition into the carbon-iodine bond of the N-methyl-N-(2-iodophenyl)acrylamide substrate. Following oxidative addition, an intramolecular migratory insertion of the alkene moiety into the palladium-carbon bond occurs, facilitating the formation of the five-membered indolinone ring and generating a divalent alkyl-palladium intermediate. This step is critical as it establishes the core heterocyclic framework upon which the rest of the molecule is built.
Subsequently, the alkyl-palladium species interacts with the carbon monoxide released from the formic acid/acetic anhydride mixture, undergoing carbonylation to form an acyl-palladium intermediate. Concurrently, in a separate parallel pathway facilitated by the sodium carbonate base, trifluoroethylimidoyl chloride reacts with propargylamine to form a trifluoroacetamidine species, which may undergo isomerization to a more reactive tautomer. The acyl-palladium intermediate then activates this trifluoroacetamidine compound, promoting an intramolecular cyclization that closes the imidazole ring and releases the final carbonyl-bridged biheterocyclic product while regenerating the palladium catalyst. This intricate interplay between the carbonylation cycle and the amidine formation ensures high atom economy. The choice of TFP as a ligand is particularly strategic, as its electron-donating properties stabilize the palladium center during the oxidative addition step while remaining labile enough to allow for substrate coordination, thus driving the reaction forward with high turnover numbers.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction parameters to maximize yield and purity. The patent outlines a standardized procedure where the catalyst system is prepared first, followed by the sequential or simultaneous addition of the three organic substrates. Maintaining an inert atmosphere is crucial to prevent oxidation of the phosphine ligand and the palladium center. The reaction progress should be monitored via TLC or HPLC to determine the optimal endpoint, typically falling within the 12 to 20-hour window specified in the patent data. Post-reaction workup involves simple filtration to remove inorganic salts, followed by adsorption onto silica gel and purification via flash column chromatography. For detailed operational parameters and specific molar ratios tailored to different substrates, please refer to the structured guide below.
- Prepare the catalytic system by mixing palladium chloride (PdCl2), tris(2-furyl)phosphine (TFP) ligand, sodium carbonate base, and a carbon monoxide substitute mixture of formic acid and acetic anhydride in an organic solvent such as THF.
- Introduce the three key substrates: trifluoroethylimidoyl chloride (II), propargylamine (III), and the substituted acrylamide (IV) into the reaction vessel under inert atmosphere.
- Maintain the reaction mixture at a mild temperature of 30°C for a duration of 12 to 20 hours to ensure complete conversion via the carbonylation cascade.
- Upon completion, perform standard post-treatment including filtration, silica gel adsorption, and column chromatography purification to isolate the high-purity carbonyl-bridged biheterocyclic product (I).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this multi-component synthesis offers substantial strategic advantages over legacy manufacturing routes. The primary driver for cost optimization is the utilization of commodity chemicals as starting materials. Trifluoroethylimidoyl chloride, propargylamine, and substituted acrylamides are widely available in the global chemical market, ensuring a stable and competitive supply base. Unlike processes that require custom-synthesized complex intermediates, this route builds complexity from simple, low-cost precursors, significantly reducing the raw material cost per kilogram of the final API intermediate. Furthermore, the elimination of high-pressure carbon monoxide infrastructure removes a major capital expenditure barrier, allowing production to occur in standard glass-lined or stainless steel reactors without the need for specialized autoclaves.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the high atom efficiency of the multi-component reaction, which minimizes the generation of waste byproducts and reduces the burden on downstream purification systems. By avoiding the use of expensive noble metal catalysts in favor of relatively inexpensive palladium chloride and simple phosphine ligands, the direct material costs are kept low. Additionally, the mild reaction temperature of 30°C translates to significant energy savings compared to processes requiring reflux conditions or cryogenic cooling, contributing to a lower overall cost of goods sold (COGS). The simplified workup procedure, involving basic filtration and chromatography, further reduces labor and solvent consumption costs associated with complex extraction protocols.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures consistent batch-to-batch quality, which is critical for maintaining regulatory compliance in pharmaceutical manufacturing. The broad substrate scope means that supply chain disruptions for specific substituted precursors can often be mitigated by switching to alternative analogs without redesigning the entire synthetic pathway. Since the reaction tolerates a wide variety of functional groups, suppliers can source generic grades of starting materials rather than ultra-high-purity custom reagents, shortening lead times and increasing procurement flexibility. This resilience makes the supply chain less vulnerable to single-source dependencies and market volatility.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the feasibility of scaling this reaction to the gram level with maintained efficiency, indicating a clear path toward kilogram and ton-scale production. The absence of toxic gas handling simplifies environmental health and safety (EHS) permitting and reduces the risk of regulatory shutdowns. Moreover, the use of THF as a solvent, which is easily recoverable and recyclable, aligns with modern sustainability goals. The reduced waste profile and lower energy footprint position this technology as a preferred choice for manufacturers aiming to meet stringent environmental standards while expanding production capacity to meet growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and mechanistic proposals within the patent documentation to assist technical teams in evaluating feasibility. Understanding these nuances is vital for successful technology transfer and process validation.
Q: What is the carbon monoxide source in this synthesis?
A: Unlike traditional carbonylations that require hazardous high-pressure CO gas, this protocol utilizes a safe liquid surrogate generated in situ from a mixture of formic acid and acetic anhydride.
Q: What are the optimal reaction conditions described in the patent?
A: The reaction proceeds efficiently at a mild temperature of 30°C in tetrahydrofuran (THF) solvent, typically requiring 12 to 20 hours for completion with high substrate compatibility.
Q: Does this method support structural diversity for drug discovery?
A: Yes, the method exhibits broad functional group tolerance, allowing for various substituents on the aryl rings (R1, R2, R3) including halogens, alkyl groups, and trifluoromethyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN115353511A for accelerating drug discovery programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale research to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting requirements of global pharmaceutical regulators. We are committed to delivering high-purity pharmaceutical intermediates that empower your R&D teams to focus on innovation rather than supply constraints.
We invite you to engage with our technical procurement team to discuss how this advanced carbonylation technology can be integrated into your specific project pipeline. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this multi-component route for your specific target molecules. Contact us today to request specific COA data for our catalog of biheterocyclic intermediates and to receive comprehensive route feasibility assessments tailored to your development timelines. Let us be your partner in turning complex chemical challenges into commercial successes.
