Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Applications
Scalable Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Applications
The pharmaceutical industry continuously seeks efficient, scalable, and environmentally benign pathways for constructing complex heterocyclic scaffolds that serve as the backbone of modern therapeutics. Patent CN113105402B introduces a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic methodologies. This innovation leverages a non-metallic iodine-promoted strategy to construct the triazole ring, specifically incorporating trifluoromethyl and acyl functionalities which are paramount for enhancing metabolic stability and bioavailability in drug candidates. By utilizing cheap and readily available starting materials such as aryl ethyl ketones and trifluoroethylimide hydrazides, this technology offers a compelling alternative to traditional transition-metal catalyzed routes. The significance of this development extends beyond academic interest, providing a tangible solution for reliable pharmaceutical intermediate supplier networks aiming to reduce dependency on scarce resources while maintaining high purity standards.
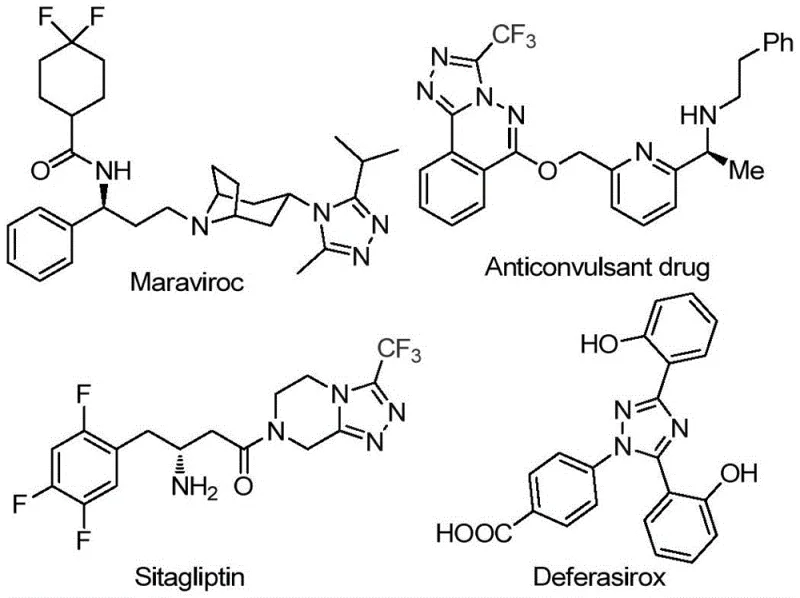
The strategic importance of the 1,2,4-triazole motif cannot be overstated, as evidenced by its presence in blockbuster medications like Maraviroc, Sitagliptin, and Deferasirox shown in the structural overview. The ability to introduce a trifluoromethyl group directly into this heterocyclic system significantly modulates the physicochemical properties of the parent compound, including electronegativity and lipophilicity. Patent CN113105402B capitalizes on this by offering a direct route to these valuable structures without the need for complex multi-step sequences often required in legacy processes. This streamlined approach not only accelerates the timeline from discovery to development but also aligns with the growing demand for green chemistry principles in industrial manufacturing. For procurement managers and supply chain heads, this represents a shift towards more resilient sourcing strategies where raw material availability and process simplicity drive cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles has relied heavily on the use of transition metal catalysts or harsh reaction conditions that pose significant challenges for large-scale implementation. Traditional methods often necessitate strict anhydrous and oxygen-free environments, requiring specialized equipment such as gloveboxes or Schlenk lines which drastically increase capital expenditure and operational complexity. Furthermore, the reliance on heavy metal catalysts introduces severe downstream purification burdens, as residual metal levels must be reduced to parts-per-million (ppm) levels to meet stringent regulatory guidelines for active pharmaceutical ingredients (APIs). These purification steps, often involving scavengers or extensive chromatography, result in substantial yield losses and generate significant hazardous waste streams. Additionally, many conventional routes struggle to simultaneously install both trifluoromethyl and acyl groups at the 3 and 5 positions of the triazole ring, limiting the chemical space accessible for medicinal chemistry optimization and forcing chemists to settle for suboptimal analogs.
The Novel Approach
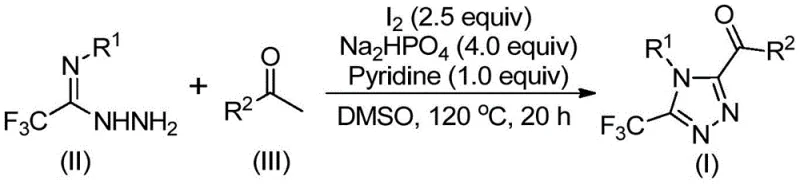
In stark contrast, the methodology disclosed in patent CN113105402B presents a paradigm shift by employing elemental iodine as a benign promoter in dimethyl sulfoxide (DMSO). This novel approach eliminates the need for toxic heavy metals entirely, thereby simplifying the impurity profile and removing the costly metal removal steps from the downstream processing workflow. The reaction proceeds efficiently under aerobic conditions without the necessity for rigorous exclusion of moisture or oxygen, making it exceptionally amenable to standard reactor setups found in most multipurpose chemical plants. The process utilizes a tandem sequence involving iodination and Kornblum oxidation of aryl ketones followed by cyclization with trifluoroethylimide hydrazide, allowing for the direct construction of the target scaffold in a single pot. This operational simplicity translates directly into reduced cycle times and lower utility consumption, positioning this method as a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation events driven by the unique reactivity of iodine in DMSO. Initially, the aryl ethyl ketone undergoes an iodination event followed by a Kornblum oxidation, effectively converting the methyl ketone moiety into an alpha-dicarbonyl or similar oxidized intermediate in situ. This activated species then engages in a dehydration condensation with the trifluoroethylimide hydrazide to form a hydrazone intermediate. The presence of sodium dihydrogen phosphate and pyridine acts as a crucial buffer system, facilitating the subsequent intramolecular cyclization that closes the triazole ring. The iodine serves a dual role, acting both as an oxidant for the initial activation of the ketone and as a promoter for the final ring closure, ensuring high atom economy. Understanding this mechanism is vital for R&D directors as it highlights the robustness of the reaction against various electronic substituents on the aromatic rings, allowing for broad substrate scope without compromising yield.
From an impurity control perspective, the absence of transition metals significantly reduces the risk of metal-catalyzed side reactions such as homocoupling or over-oxidation which are common pitfalls in palladium or copper-mediated processes. The use of DMSO as the solvent ensures excellent solubility for both polar and non-polar intermediates, preventing precipitation issues that can lead to incomplete conversions. The reaction conditions, typically heating to 110-130°C for 12-20 hours, provide sufficient thermal energy to overcome activation barriers while maintaining selectivity for the desired 1,2,4-triazole isomer over potential 1,2,3-triazole byproducts. This high level of regioselectivity is critical for ensuring the biological activity of the final drug substance, as the position of the nitrogen atoms in the ring profoundly affects binding affinity to biological targets. The detailed structural confirmation data provided in the patent, including NMR and HRMS analysis, confirms the formation of the desired 3,4,5-trisubstituted architecture with high fidelity.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory scale optimization and pilot plant campaigns. The protocol involves a sequential addition strategy where the aryl ketone is first activated before the introduction of the nitrogen source, minimizing side reactions. Detailed standardized operating procedures for this specific transformation are outlined below to ensure reproducibility and safety during scale-up operations.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat the mixture to 90-110°C for 4-6 hours to initiate Kornblum oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iodine-promoted synthesis offers profound strategic advantages that extend well beyond simple reaction yields. The primary value proposition lies in the drastic simplification of the supply chain for raw materials; elemental iodine and aryl ketones are commodity chemicals available from multiple global sources, mitigating the risk of single-source dependency often associated with specialized ligands or organometallic catalysts. This diversification of supply sources enhances supply chain reliability and provides significant leverage in price negotiations. Furthermore, the elimination of heavy metal catalysts removes the need for expensive metal scavengers and the associated disposal costs for hazardous metal waste, leading to substantial cost savings in waste management and environmental compliance. The robustness of the reaction conditions means that production can be scheduled with greater flexibility, as there is no need for complex drying of solvents or inert gas purging, thereby increasing overall plant throughput and asset utilization.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the replacement of precious metal catalysts with inexpensive elemental iodine, which represents a fraction of the cost per mole. By avoiding the use of palladium, copper, or rhodium, manufacturers eliminate the volatility associated with precious metal markets and the high cost of catalyst recovery systems. Additionally, the simplified workup procedure, which often requires only filtration and standard chromatography rather than complex extraction or metal scavenging, reduces labor hours and solvent consumption. These factors combine to lower the overall cost of goods sold (COGS), allowing for more competitive pricing of the final high-purity pharmaceutical intermediates in the global market.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like sodium dihydrogen phosphate and pyridine ensures that production schedules are not disrupted by the degradation of sensitive catalysts or ligands. Since the reaction does not require anhydrous conditions, the quality specifications for the solvent (DMSO) are less stringent, allowing for the use of technical grade solvents in early stages which further drives down costs. This tolerance to ambient conditions reduces the technical barrier for contract manufacturing organizations (CMOs) to adopt the process, expanding the pool of qualified suppliers and reducing lead time for high-purity intermediates. Consequently, pharmaceutical companies can maintain leaner inventories and respond more rapidly to market demands without fearing supply interruptions due to specialized reagent shortages.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or tonne levels is straightforward due to the absence of exothermic risks associated with strong oxidants or pyrophoric reagents. The use of DMSO, a high-boiling polar aprotic solvent, facilitates heat transfer and mixing in large reactors, ensuring consistent reaction performance across different batch sizes. From an environmental standpoint, the process aligns with green chemistry principles by avoiding toxic heavy metals, thereby simplifying the environmental impact assessment and permitting process for new manufacturing facilities. The reduced generation of hazardous waste and the potential for solvent recycling contribute to a lower carbon footprint, which is increasingly becoming a key criterion for supplier selection by major multinational pharmaceutical corporations committed to sustainability goals.
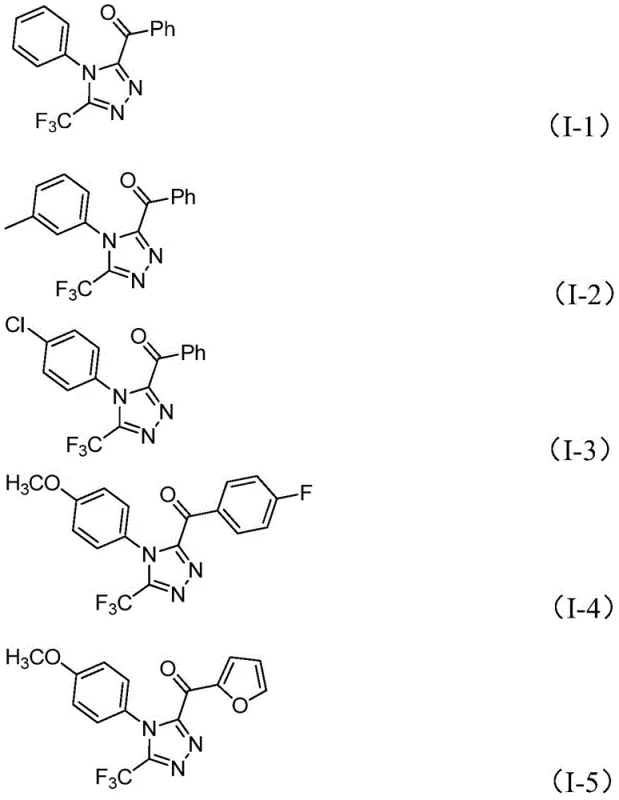
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams evaluating this route for their specific projects.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metal promoter, completely avoiding the use of toxic and costly heavy metal catalysts typically found in traditional triazole synthesis.
Q: What are the reaction conditions regarding moisture and oxygen sensitivity?
A: The process is highly robust and does not require strict anhydrous or oxygen-free conditions, significantly simplifying operational requirements and reducing equipment costs for large-scale production.
Q: Is this method suitable for producing trifluoromethyl-containing drug intermediates?
A: Yes, the method specifically targets the synthesis of 3,4,5-trisubstituted 1,2,4-triazoles containing both trifluoromethyl and acyl groups, which are critical scaffolds in drugs like Sitagliptin and Deferasirox.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iodine-promoted synthesis described in patent CN113105402B for the production of advanced pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest international standards. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process engineering team is ready to optimize this route for your specific target molecule, ensuring maximum yield and minimal impurity formation.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development program. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team today to request specific COA data for similar triazole derivatives and to discuss route feasibility assessments for your proprietary compounds. Let us help you accelerate your path to market with a reliable, cost-effective, and scalable supply solution for your critical 1,2,4-triazole building blocks.
