Advanced Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up of Complex Pharmaceutical Intermediates
Introduction to Novel Quinolinone Synthesis Technology
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing privileged heterocyclic scaffolds, among which quinolin-2(1H)one derivatives hold a paramount position due to their pervasive presence in bioactive natural products and therapeutic agents. As detailed in the recent intellectual property disclosure CN113045489B, a groundbreaking preparation method has been established that leverages palladium-catalyzed aminocarbonylation to access these valuable structures with unprecedented efficiency. This technology addresses critical bottlenecks in traditional synthetic routes by utilizing benzisoxazole not merely as a nitrogen donor but uniquely as a dual source for both nitrogen and the formyl group required for ring construction. The strategic integration of molybdenum hexacarbonyl as a solid carbon monoxide surrogate further enhances the operational safety and simplicity of the process, eliminating the hazards associated with handling gaseous CO. For R&D directors and process chemists, this represents a significant leap forward in designing safer, more atom-economical pathways for high-purity pharmaceutical intermediates.
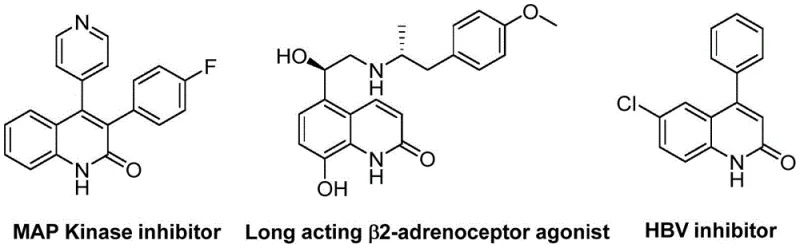
The structural versatility of the resulting 3-arylquinolin-2(1H)one core is exemplified by its occurrence in potent MAP Kinase inhibitors, long-acting β2-adrenoceptor agonists, and HBV inhibitors, highlighting the immense commercial value of mastering this synthetic transformation. By adopting the methodology outlined in patent CN113045489B, manufacturers can access a broad chemical space previously difficult to navigate economically. The reaction operates under relatively mild thermal conditions compared to classical cyclization methods, thereby preserving sensitive functional groups that are often essential for downstream biological activity. This capability is crucial for the development of next-generation therapeutics where molecular complexity continues to increase. Furthermore, the use of commercially available starting materials ensures that the supply chain remains resilient against raw material shortages, a key consideration for procurement managers aiming to secure long-term production stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations, which, while effective, suffer from significant drawbacks in a modern industrial context. These traditional protocols often necessitate the use of harsh reagents, extreme temperatures, or toxic solvents that complicate waste management and increase the environmental footprint of the manufacturing process. Moreover, many conventional routes require pre-functionalized aniline derivatives or specific beta-keto esters, limiting the scope of accessible substitution patterns and often requiring multi-step sequences to install the necessary precursors. Transition metal-catalyzed improvements have been attempted, yet many still rely on external carbon monoxide gas, posing severe safety risks and requiring specialized high-pressure equipment that is not universally available in standard pilot plants. Additionally, the regioselectivity in some of these older methods can be problematic, leading to difficult-to-separate isomeric mixtures that drastically reduce overall yield and purity.
The Novel Approach
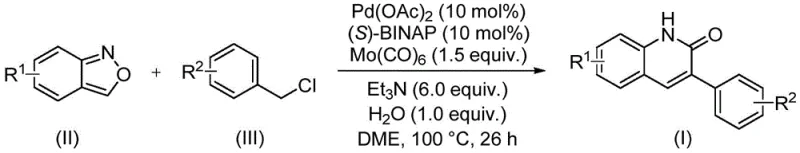
In stark contrast, the novel approach disclosed in the patent utilizes a sophisticated palladium-catalyzed aminocarbonylation strategy that fundamentally reimagines the construction of the quinolinone core. By employing benzisoxazole as a latent source of both the amine nitrogen and the carbonyl carbon, the reaction bypasses the need for separate formylation steps or hazardous gas handling. The inclusion of molybdenum hexacarbonyl allows for the in situ generation of carbon monoxide under controlled conditions, facilitating the carbonylation step safely within a standard sealed tube setup. This methodology exhibits exceptional functional group tolerance, accommodating electron-donating groups like methoxy and tert-butyl as well as electron-withdrawing groups such as cyano and trifluoromethyl without significant loss in efficiency. The operational simplicity is further enhanced by the use of common organic bases like triethylamine and solvents like DME, making the transition from laboratory discovery to commercial scale-up of complex pharmaceutical intermediates seamless and cost-effective.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The mechanistic pathway of this transformation involves a intricate dance of organometallic steps initiated by the oxidative addition of the benzyl chloride substrate to the active palladium(0) species generated in situ from palladium acetate and the chiral ligand (S)-BINAP. Following oxidative addition, the coordination and subsequent insertion of carbon monoxide—released from the decomposition of molybdenum hexacarbonyl—into the palladium-carbon bond forms an acyl-palladium intermediate. This acyl species then undergoes nucleophilic attack by the nitrogen atom derived from the ring-opening of the benzisoxazole moiety, a process facilitated by the basic conditions provided by triethylamine. The unique ability of benzisoxazole to act as a dual donor is central to this mechanism, as the cleavage of the N-O bond provides the necessary nitrogen for the amide linkage while the adjacent carbon framework integrates into the newly forming heterocyclic ring. Water plays a critical role as an additive, likely assisting in the proton transfer steps necessary to finalize the cyclization and regenerate the active catalyst species.
Understanding the impurity profile is vital for R&D teams focused on regulatory compliance, and this catalytic system offers distinct advantages in controlling side reactions. The high selectivity of the palladium catalyst minimizes the formation of homocoupling byproducts often seen in radical-based benzyl chloride reactions. Furthermore, the specific electronic properties of the (S)-BINAP ligand help stabilize the catalytic cycle, preventing premature catalyst deactivation which can lead to incomplete conversions and difficult-to-remove palladium black residues. The reaction conditions, specifically the temperature of 100°C and the 26-hour duration, are optimized to balance reaction kinetics with thermal stability, ensuring that the delicate benzisoxazole ring opens cleanly without decomposing into polymeric tars. This level of control results in a crude reaction mixture that is significantly cleaner than those obtained from traditional acid-mediated cyclizations, thereby reducing the burden on downstream purification processes and improving the overall mass balance of the synthesis.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
To implement this high-efficiency synthesis in a laboratory or pilot setting, operators must adhere to precise stoichiometric ratios and mixing protocols to maximize yield and reproducibility. The process begins with the careful weighing of the palladium catalyst, ligand, and carbonyl source, followed by the addition of the solid and liquid reagents into a pressure-rated vessel capable of withstanding the reaction temperature. It is imperative to ensure thorough mixing of the heterogeneous components before heating to prevent localized hot spots that could degrade the catalyst or substrates. The patent emphasizes the importance of the water additive, which, although used in small amounts (1.0 equiv.), is crucial for the successful turnover of the catalytic cycle. Detailed standardized operating procedures regarding the filtration and silica gel treatment steps are essential to remove metal residues effectively before the final chromatographic purification.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend far beyond simple yield improvements, directly impacting the bottom line through reduced operational complexity and enhanced raw material security. The reliance on benzisoxazole and benzyl chloride derivatives as starting materials is particularly advantageous because these commodities are widely produced on a global scale, ensuring a stable supply chain that is less susceptible to the geopolitical disruptions often affecting exotic reagents. The elimination of gaseous carbon monoxide cylinders from the process inventory removes a significant safety liability and reduces the regulatory overhead associated with storing and transporting toxic gases. Furthermore, the simplified workup procedure, which avoids complex aqueous extractions or cryogenic conditions, translates into lower energy consumption and reduced solvent usage, aligning with modern green chemistry initiatives that are increasingly mandated by corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the dual functionality of benzisoxazole, which effectively replaces two separate reagents (an amine source and a carbonyl source), thereby reducing the total number of raw materials required per batch. By utilizing molybdenum hexacarbonyl as a solid CO surrogate, the need for expensive high-pressure autoclaves is negated, allowing the reaction to proceed in standard glass-lined reactors or sealed tubes, which significantly lowers capital expenditure requirements for new production lines. The high yields reported, reaching up to 97% for certain substrates, mean that less raw material is wasted, and the throughput per reactor volume is maximized, leading to substantial cost savings in large-scale API manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output quality regardless of minor fluctuations in ambient processing parameters, which is critical for maintaining just-in-time delivery schedules to downstream customers. Since the substrates are stable solids or liquids with long shelf lives, inventory management becomes more predictable, reducing the risk of spoilage or degradation that can plague more sensitive reagents. The broad substrate scope means that a single manufacturing platform can be adapted to produce a wide variety of analogues without requiring extensive re-validation of the entire process, providing the flexibility needed to respond quickly to changing market demands for different drug candidates.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram quantities is straightforward due to the absence of exothermic hazards typically associated with gas-liquid carbonylations, making it safer for operators and easier to model for engineering teams. The use of DME as a solvent, while requiring recovery systems, is well-established in the industry, and the overall reduction in waste generation simplifies the effluent treatment process. This alignment with environmental, social, and governance (ESG) criteria makes the technology attractive for companies aiming to reduce their carbon footprint and meet stringent regulatory standards for pharmaceutical production.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this palladium-catalyzed aminocarbonylation technology, drawing directly from the experimental data and specifications provided in the patent documentation. These insights are intended to assist technical teams in evaluating the feasibility of integrating this method into their existing workflows for producing high-value heterocyclic intermediates. Understanding the nuances of catalyst loading, solvent selection, and substrate compatibility is essential for achieving the reported high yields and purity levels in a commercial setting.
Q: What are the key advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (carbonyl) source, simplifying the reaction stoichiometry and reducing the need for external carbon monoxide gas or complex formylating agents.
Q: What is the optimal catalyst system for this transformation?
A: The patent specifies a system comprising palladium acetate (10 mol%), (S)-BINAP (10 mol%), and molybdenum hexacarbonyl (1.5 equiv.) in the presence of triethylamine and water.
Q: Does this method tolerate diverse functional groups on the substrates?
A: Yes, the method demonstrates wide functional group tolerance, successfully accommodating substituents such as methoxy, chloro, cyano, trifluoromethyl, and tert-butyl groups on both the benzisoxazole and benzyl chloride rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in patent CN113045489B for accelerating drug discovery and development pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop innovation to market-ready product is seamless and efficient. Our state-of-the-art facilities are equipped to handle sensitive organometallic chemistry with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of 3-arylquinolin-2(1H)one derivative meets the exacting requirements of the global pharmaceutical industry. We are committed to being a reliable pharmaceutical intermediate supplier that not only delivers quality but also drives innovation through continuous process improvement.
We invite you to collaborate with our technical procurement team to explore how this cutting-edge synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this greener, more efficient route. We encourage potential partners to contact us directly to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for custom targets, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
