Scalable Synthesis Of Complex Macrocyclic FXIa Inhibitor Intermediates For Commercial Production
The pharmaceutical industry's relentless pursuit of safer antithrombotic therapies has placed Factor XIa (FXIa) inhibitors at the forefront of cardiovascular research, offering the potential for anticoagulation with a reduced risk of bleeding compared to traditional agents. Patent CN114375290A discloses novel synthetic options for the preparation of complex macrocyclic compounds, specifically targeting the efficient production of Compound I, a potent FXIa inhibitor. This patent represents a significant technological leap by addressing the inherent scalability challenges associated with macrocyclization and stereocenter installation in large molecule synthesis. For global pharmaceutical manufacturers, the methods described herein provide a robust framework for transitioning from laboratory-scale discovery to commercial-grade production, ensuring that critical drug candidates can be supplied reliably to meet clinical and market demands without the bottlenecks typically associated with complex organic synthesis.
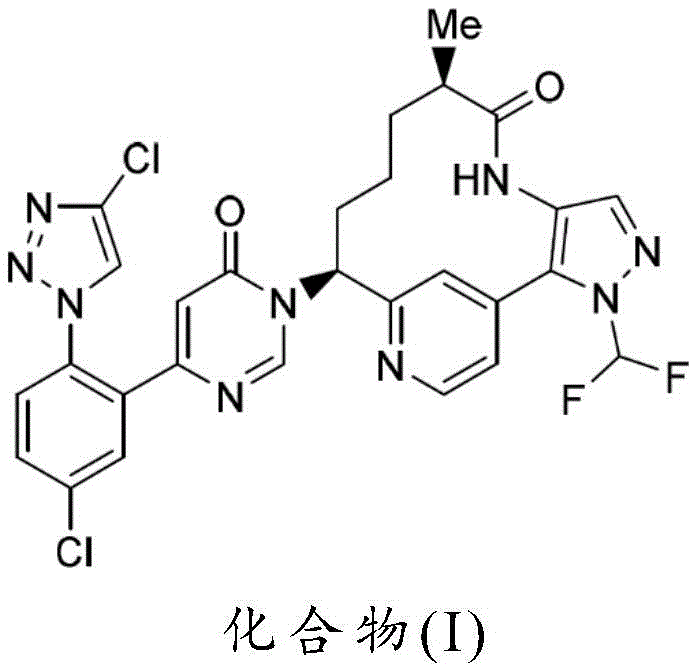
The development of this synthetic pathway is particularly relevant for supply chain stakeholders who must navigate the complexities of sourcing high-purity intermediates for late-stage clinical trials and commercial launches. By optimizing the construction of the pyrazolo-diazacyclotetradecyn core, the patent outlines a strategy that minimizes reliance on scarce reagents and maximizes overall process efficiency. This approach not only secures the intellectual property landscape around these vital therapeutics but also establishes a clear precedent for manufacturing excellence in the realm of specialized pharmaceutical intermediates. Understanding the nuances of this patent is essential for any organization looking to partner with a reliable pharmaceutical intermediates supplier capable of executing high-complexity chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing macrocyclic FXIa inhibitors often relied heavily on ring-closing metathesis (RCM) strategies using catalysts such as Grubbs (II). While effective on a small scale, these methodologies present substantial hurdles when translated to industrial manufacturing environments. The primary constraint lies in the cost and availability of the ruthenium-based catalysts, which can drive up the cost of goods significantly and introduce supply chain vulnerabilities due to the limited number of global suppliers for such specialized reagents. Furthermore, the removal of residual heavy metals from the final active pharmaceutical ingredient (API) requires additional purification steps, increasing processing time and waste generation. These factors collectively render conventional metathesis routes less economically viable and operationally risky for large-scale production campaigns.
The Novel Approach
In contrast, the novel approach detailed in the patent circumvents these issues by employing a convergent linear synthesis that builds the macrocyclic structure through robust amide bond formations and precise stereochemical control early in the sequence. This strategy eliminates the need for expensive olefin metathesis catalysts, thereby drastically simplifying the purification profile and reducing the environmental footprint of the manufacturing process. By focusing on the assembly of key intermediates like Compound 7 and Compound 10 through classical organic transformations such as Claisen condensations and chiral alkylations, the process ensures higher yields and better reproducibility. This shift in synthetic logic provides a more stable foundation for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to predict costs and timelines with greater accuracy.
Mechanistic Insights into Chiral Auxiliary Alkylation and Enzymatic Resolution
A critical component of this synthetic route is the establishment of the chiral center at the methyl-substituted carbon, which is essential for the biological activity of the final inhibitor. The patent describes the use of chiral auxiliaries, such as oxazolidinones, which are temporarily attached to the substrate to direct the stereochemical outcome of alkylation reactions. This method involves the formation of an enolate species that reacts with an alkyl halide in a highly diastereoselective manner, ensuring that the desired enantiomer is produced with high fidelity. The auxiliary is subsequently removed under mild hydrolytic conditions, recovering the chiral acid intermediate without racemization. This level of control is paramount for R&D directors who must ensure that the impurity profile of the API remains within strict regulatory limits throughout the product lifecycle.
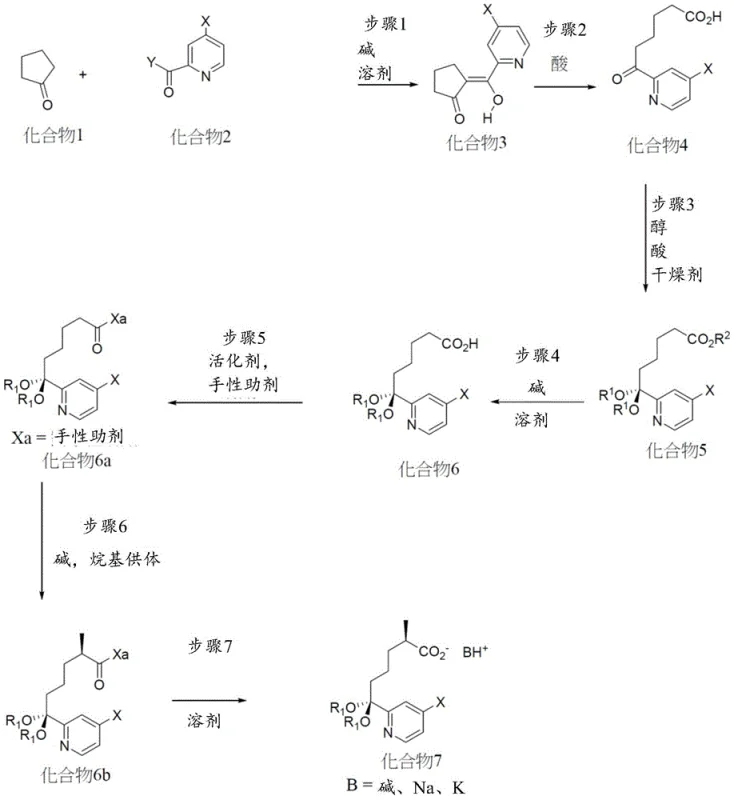
Alternatively, the patent highlights the utility of biocatalytic processes, specifically transaminase-mediated reactions, to introduce chirality. Enzymatic resolution or asymmetric amination offers a green chemistry alternative that operates under mild aqueous conditions, often achieving superior enantiomeric excess compared to traditional chemical methods. The use of engineered transaminases allows for the conversion of ketone intermediates directly into chiral amines, streamlining the synthesis and reducing the number of isolation steps required. This mechanistic flexibility provides manufacturers with options to tailor the process based on available infrastructure and cost targets, ensuring that the production of high-purity pharmaceutical intermediates can be optimized for both technical performance and economic efficiency.
How to Synthesize FXIa Inhibitor Intermediate Efficiently
The synthesis of the core intermediate involves a sequence of carefully orchestrated reactions beginning with the condensation of cyclic ketones and pyridine derivatives. This initial step sets the stage for the subsequent functionalization of the carbon chain, requiring precise control over reaction temperatures and stoichiometry to avoid side reactions. Following the formation of the beta-keto ester scaffold, the process moves through protection group chemistry to mask reactive ketones, allowing for selective transformations elsewhere in the molecule. The detailed standardized synthesis steps see the guide below.
- React cyclopentanone derivatives with pyridine esters under basic conditions to form beta-keto esters.
- Perform retro-Claisen reaction and subsequent ketalization to protect ketone functionalities.
- Utilize chiral auxiliaries or enzymatic resolution to establish critical stereocenters with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond mere technical feasibility. The elimination of precious metal catalysts directly correlates to a reduction in raw material costs and simplifies the waste management protocols required for manufacturing compliance. By utilizing commodity chemicals and widely available reagents, the process mitigates the risk of supply disruptions that often plague specialized catalyst markets. This stability is crucial for maintaining continuous production schedules and ensuring that drug development timelines are not compromised by material shortages. The robust nature of the intermediates also allows for longer shelf lives and easier transportation, further enhancing supply chain reliability.
- Cost Reduction in Manufacturing: The removal of expensive Grubbs catalysts and the associated heavy metal scavenging steps leads to substantial cost savings in the overall production budget. By relying on more economical starting materials and avoiding complex purification protocols for metal removal, the process economics are significantly improved. This efficiency allows for a more competitive pricing structure for the final intermediate, making it an attractive option for cost-sensitive pharmaceutical projects. The streamlined workflow also reduces labor and utility consumption, contributing to a leaner manufacturing operation that maximizes resource utilization without compromising quality standards.
- Enhanced Supply Chain Reliability: Sourcing strategies are strengthened by the use of common organic building blocks that are available from multiple vendors globally. This diversification of the supply base reduces dependency on single-source suppliers and minimizes the impact of geopolitical or logistical disruptions. The stability of the intermediates ensures that inventory can be managed more effectively, allowing for strategic stockpiling without the risk of degradation. Consequently, lead times for high-purity pharmaceutical intermediates can be optimized, providing customers with greater certainty regarding delivery schedules and project milestones.
- Scalability and Environmental Compliance: The synthetic route is designed with scale-up in mind, utilizing reactions that are exothermically manageable and compatible with standard reactor configurations. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, reducing the burden of disposal and compliance reporting. This environmental stewardship not only protects the manufacturer from regulatory risks but also enhances the corporate sustainability profile. The ability to scale from kilogram to multi-ton quantities without significant process re-engineering ensures that the supply can grow in tandem with clinical and commercial demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of these complex intermediates. The answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on process capabilities and quality standards. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains.
Q: Why is the Grubbs catalyst method less suitable for large-scale production?
A: The patent indicates that Grubbs (II) reagents are costly and difficult to adapt for commercial scale synthesis, prompting the need for alternative linear routes.
Q: How is stereochemistry controlled in the new process?
A: Stereocontrol is achieved through the use of chiral auxiliaries like oxazolidinones or via enzymatic transamination and resolution steps.
Q: What are the supply chain benefits of this synthetic route?
A: The route utilizes readily available starting materials and avoids expensive transition metal catalysts, enhancing supply continuity and reducing procurement complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Factor XIa Inhibitor Intermediate Supplier
The technological potential of the synthetic routes described in Patent CN114375290A is immense, offering a pathway to produce life-saving antithrombotic agents with greater efficiency and purity. NINGBO INNO PHARMCHEM stands ready to leverage this intellectual property, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to your project. Our facility is equipped with stringent purity specifications and rigorous QC labs to ensure that every batch of intermediate meets the exacting standards required for global pharmaceutical registration. We understand the critical nature of these supply chains and are committed to delivering consistency and quality at every stage of the manufacturing process.
We invite you to engage with our technical procurement team to discuss how we can support your specific development needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing capabilities can optimize your budget and timeline. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements. Let us partner with you to navigate the complexities of fine chemical manufacturing and bring your therapeutic candidates to market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
