Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Compounds for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access privileged heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. A significant breakthrough in this domain is detailed in patent CN112538054B, which discloses a highly efficient preparation method for 1,2,4-triazole-3-one compounds. These five-membered nitrogen-containing heterocycles are not merely academic curiosities; they are critical structural motifs found in a vast array of bioactive molecules exhibiting potent antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant properties. As illustrated in the biological context below, these cores are integral to PPARα agonists, antitumor agents, and even established drugs like Itraconazole.
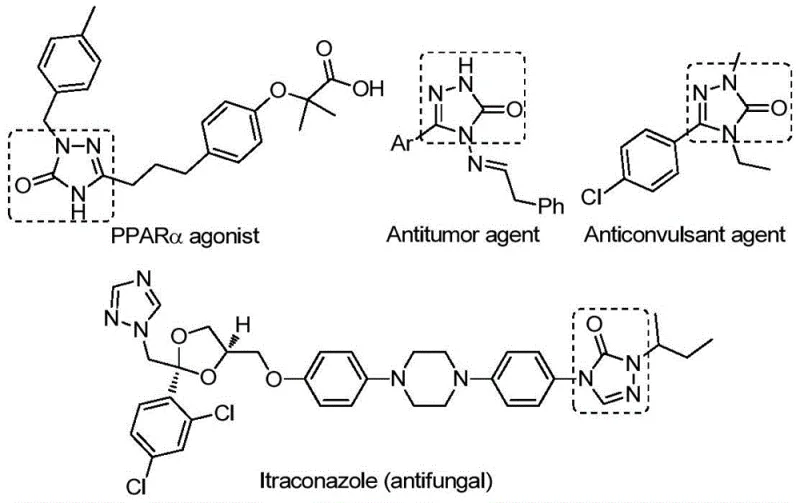
The innovation presented in this patent addresses a long-standing bottleneck in the synthesis of these valuable intermediates. By leveraging a transition metal palladium-catalyzed carbonylation tandem cyclization reaction, the process transforms cheap and readily available chlorohydrazones and sodium azide into complex triazole ketones with remarkable efficiency. For procurement managers and R&D directors alike, this represents a paradigm shift towards more sustainable and cost-effective pharmaceutical intermediate manufacturing, offering a reliable pathway to high-purity building blocks essential for drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthetic landscape for constructing the 1,2,4-triazole-3-one ring has been fraught with significant operational and economic challenges. Traditional literature methods often rely on harsh reaction conditions that are difficult to control on a large scale, such as the cyclization of benzoyl hydrazide with urea under strong basic conditions or the condensation of thioamides with hydrazines at elevated temperatures. These legacy routes frequently suffer from low atom economy, requiring multiple steps and the pre-activation of substrates, which inherently drives up the cost of goods sold (COGS). Furthermore, the narrow substrate scope of these older methodologies means that introducing specific functional groups often requires entirely different synthetic strategies, complicating the supply chain for diverse analog libraries needed in modern medicinal chemistry.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a sophisticated palladium-catalyzed cascade that streamlines the synthesis into a single, high-yielding operation. The core of this innovation is the use of chlorohydrazones as stable precursors which, under the influence of a Pd(0) catalyst and a specialized ligand system, undergo oxidative addition followed by carbonylation. Crucially, the reaction employs TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide surrogate, thereby circumventing the safety hazards and infrastructure costs associated with handling gaseous CO. As shown in the general reaction scheme below, this method allows for the direct conversion of simple starting materials into the target heterocycle with exceptional substrate compatibility.

This streamlined protocol not only simplifies the operational workflow but also drastically reduces the environmental footprint by minimizing waste generation. The ability to tolerate a wide range of functional groups—from electron-rich alkyls to electron-deficient halogens—means that a single standardized process can be applied to generate a vast library of derivatives, significantly accelerating the timeline for cost reduction in API manufacturing and process development.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
To fully appreciate the robustness of this technology, one must delve into the intricate mechanistic pathway that drives the transformation. The reaction initiates with the oxidative addition of the low-valent palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the thermal decomposition of TFBen releases carbon monoxide in situ, which then inserts into the carbon-palladium bond to form a key acyl-palladium species. This step is critical as it builds the carbonyl functionality directly into the growing molecular framework without external gas feeds.
Following the carbonylation event, the acyl-palladium intermediate interacts with sodium azide to generate an acyl azide compound. This unstable intermediate rapidly undergoes a Curtius rearrangement, a thermally driven process that migrates the acyl group to the nitrogen, expelling nitrogen gas and forming an isocyanate intermediate. The final stage involves an intramolecular nucleophilic attack by the remaining hydrazine nitrogen onto the electrophilic isocyanate carbon, closing the ring to yield the stable 1,2,4-triazole-3-one product. This elegant cascade ensures high regioselectivity and minimizes the formation of byproducts, which is paramount for maintaining high-purity pharmaceutical intermediates required for regulatory compliance.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires precise adherence to the optimized conditions defined in the patent to ensure maximum yield and reproducibility. The process is designed to be operationally simple, utilizing standard Schlenk techniques or sealed vessel reactors to maintain the integrity of the reaction environment. The following guide outlines the critical parameters for executing this transformation effectively, ensuring that the complex catalytic cycle proceeds to completion.
- Combine palladium catalyst (Pd2(dba)3), Xantphos ligand, TFBen, chlorohydrazone substrate, and sodium azide in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 100°C and maintain stirring for 16 to 30 hours to allow the carbonylation and cyclization cascade to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity 1,2,4-triazole-3-one product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this palladium-catalyzed methodology offers tangible strategic benefits that extend beyond mere chemical yield. The shift from multi-step, harsh traditional syntheses to this one-pot cascade significantly de-risks the supply of critical heterocyclic building blocks. By utilizing commodity chemicals like sodium azide and easily synthesized chlorohydrazones, manufacturers can secure a more resilient supply chain that is less susceptible to the volatility of specialized reagent markets.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the elimination of expensive and hazardous reagents. By replacing high-pressure carbon monoxide gas with the solid surrogate TFBen, facilities can avoid the capital expenditure associated with specialized high-pressure reactors and gas handling safety systems. Furthermore, the high catalytic efficiency means that precious metal loading is minimized, and the simplified workup procedure reduces solvent consumption and labor hours, leading to substantial overall cost savings in production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply continuity. The method's tolerance for various functional groups implies that a single manufacturing line can be adapted to produce a wide variety of substituted triazole derivatives without extensive retooling. This flexibility allows for rapid response to changing demand patterns in drug development, ensuring that commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal lead time adjustments.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior. The avoidance of toxic gases and the use of relatively benign solvents like 1,4-dioxane (which can be recycled) align with green chemistry principles. The high conversion rates reduce the burden on waste treatment facilities, and the straightforward purification via column chromatography or crystallization ensures that the final product meets stringent purity specifications with minimal environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, aimed at clarifying the practical aspects of adopting this technology for industrial applications.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-tricarboxylic acid phenol ester) serves as a safe and solid carbon monoxide substitute, eliminating the need for handling hazardous high-pressure CO gas while effectively driving the carbonylation step.
Q: What represents the optimal catalyst loading for this transformation?
A: The patent specifies a molar ratio where the palladium catalyst (Pd2(dba)3) is used at approximately 2.5 mol% relative to the substrate, balanced with 5 mol% of the Xantphos ligand for optimal turnover.
Q: Does this method support diverse substrate scopes?
A: Yes, the methodology demonstrates excellent functional group tolerance, successfully accommodating various aryl groups including those with electron-withdrawing halogens and electron-donating alkyl or alkoxy substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
The technological potential of this palladium-catalyzed carbonylation route is immense, offering a clear path to high-quality heterocyclic intermediates essential for modern drug discovery. At NINGBO INNO PHARMCHEM, we pride ourselves on our capability to translate such innovative academic and patent literature into robust, industrial-scale processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from gram-scale optimization to full commercial supply. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,2,4-triazole-3-one delivered meets the exacting standards required by global regulatory bodies.
We invite you to leverage our technical expertise to optimize your supply chain for these critical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
