Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Drug Discovery
Scalable Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Drug Discovery
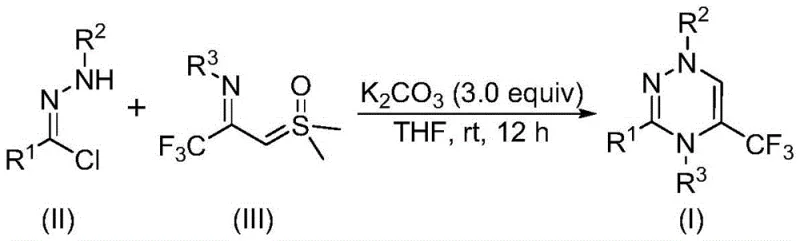
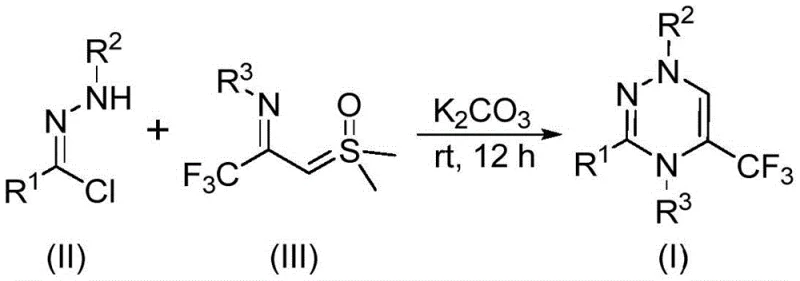
The rapid evolution of medicinal chemistry demands efficient access to fluorinated heterocyclic scaffolds, particularly those capable of enhancing metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in current synthetic methodologies. This innovation leverages a base-promoted [3+3] cycloaddition strategy that operates under remarkably mild conditions, utilizing inexpensive potassium carbonate as a promoter in an air atmosphere. For R&D directors and procurement specialists alike, this represents a paradigm shift away from costly transition-metal catalysis toward sustainable, operationally simple chemistry. The ability to construct these privileged structures at room temperature without inert gas protection not only lowers the barrier to entry for laboratory synthesis but also paves the way for robust industrial scale-up, ensuring a reliable supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied on condensation reactions between amidrazones and 1,2-diketones or alkynes, often requiring elevated temperatures and extended reaction times that degrade sensitive functional groups. Alternative multicomponent reactions involving hydrazides and dicarbonyl compounds frequently suffer from poor atom economy and generate significant amounts of waste, complicating downstream purification processes. Furthermore, many existing protocols necessitate the use of expensive heavy metal catalysts or strictly anhydrous and anaerobic conditions, which impose severe constraints on manufacturing infrastructure and operational costs. These traditional pathways often lack the structural diversity required for modern drug discovery, limiting the exploration of chemical space around the triazine scaffold and hindering the optimization of pharmacokinetic properties in lead compounds.
The Novel Approach
In stark contrast, the methodology disclosed in the patent utilizes readily available chlorohydrazones and trifluoroacetyl sulfur ylides as starting materials, reacting them in the presence of potassium carbonate within common organic solvents like tetrahydrofuran. This novel route proceeds efficiently at temperatures between 20°C and 40°C, completely eliminating the need for energy-intensive heating or cooling systems. The reaction tolerates exposure to air, removing the logistical burden of maintaining nitrogen or argon atmospheres, which is a significant advantage for both small-scale screening and large-batch production. By avoiding heavy metal catalysts, the process inherently produces cleaner crude products, drastically reducing the complexity of impurity profiles and minimizing the need for rigorous metal scavenging steps that often plague pharmaceutical manufacturing.
Mechanistic Insights into Base-Promoted [3+3] Cycloaddition
The mechanistic elegance of this transformation lies in the in situ generation of a reactive nitrile imine intermediate from the chlorohydrazone precursor under the promotion of potassium carbonate. This dipolar species then engages in a synergistic [3+3] cycloaddition with the trifluoroacetyl sulfur ylide, a process that is both regioselective and highly efficient. The elimination of dimethyl sulfoxide as a byproduct drives the equilibrium forward, ensuring high conversion rates without the need for exotic reagents. Alternatively, the reaction may proceed through a stepwise pathway involving intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution, ultimately yielding the stable 1,2,4-triazine ring system. Understanding this mechanism is crucial for process chemists, as it highlights the robustness of the reaction against moisture and oxygen, factors that typically derail similar cycloaddition strategies.
From an impurity control perspective, the absence of transition metals means that the primary impurities are likely to be unreacted starting materials or simple hydrolysis products, which are generally easier to separate than metal-complexed side products. The use of potassium carbonate, a mild inorganic base, ensures that acid-sensitive functional groups on the aromatic rings remain intact, preserving the integrity of complex molecular architectures. This selectivity is paramount when synthesizing libraries of analogs where subtle changes in electronic properties can dictate biological activity. The broad substrate scope, accommodating various substituents such as halogens, alkoxy groups, and trifluoromethyl groups on the phenyl rings, further demonstrates the versatility of this mechanistic pathway in generating diverse chemical entities for biological evaluation.
How to Synthesize Trifluoromethyl Substituted 1,2,4-Triazine Efficiently
To implement this synthesis effectively, one must adhere to the optimized stoichiometry and conditions detailed in the patent examples, which demonstrate consistent high yields across a variety of substrates. The process begins with the precise weighing of chlorohydrazone and trifluoroacetyl sulfur ylide, typically in a molar ratio favoring the ylide to ensure complete consumption of the hydrazone. These reagents are suspended in an aprotic solvent such as THF, which provides the ideal polarity for solubilizing the organic reactants while facilitating the heterogeneous catalysis by the solid potassium carbonate base. The mixture is then stirred at ambient temperature for a duration of 10 to 14 hours, allowing sufficient time for the cycloaddition to reach completion without the risk of thermal decomposition.
- Mix potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent like THF.
- Stir the reaction mixture at room temperature (20-40°C) in an air atmosphere for 10 to 14 hours.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic benefits that extend far beyond the laboratory bench. The elimination of precious metal catalysts such as palladium or rhodium removes a major cost driver and supply chain vulnerability, as these metals are subject to volatile market prices and geopolitical sourcing risks. Furthermore, the reliance on commodity chemicals like potassium carbonate and THF ensures that raw material availability remains stable and predictable, safeguarding production schedules against disruptions. The simplified operational requirements, specifically the ability to run reactions in air at room temperature, translate directly into reduced capital expenditure on specialized reactor vessels and utility systems, making this technology accessible for manufacturers of all scales.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts fundamentally alters the cost structure of the synthesis by eliminating the need for expensive catalyst loading and the subsequent, often costly, metal removal steps required to meet regulatory limits. This simplification of the downstream processing workflow significantly reduces the consumption of scavengers and filtration media, leading to substantial overall cost savings in the manufacturing of complex pharmaceutical intermediates. Additionally, the use of inexpensive inorganic bases instead of organic amines or strong alkalis further drives down the raw material costs, enhancing the economic viability of producing these high-value heterocycles.
- Enhanced Supply Chain Reliability: By utilizing starting materials that are commercially available or easily synthesized from bulk commodities, this method mitigates the risk of supply shortages associated with specialized reagents. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures related to inert gas systems or temperature control units, ensuring a more consistent and reliable output. This stability is critical for maintaining continuous supply lines to downstream API manufacturers, who depend on timely deliveries to meet their own production targets and regulatory filing deadlines.
- Scalability and Environmental Compliance: The green chemistry attributes of this process, including the avoidance of toxic heavy metals and the use of mild conditions, align perfectly with increasingly stringent environmental regulations and corporate sustainability goals. Scaling this reaction from gram to kilogram quantities does not require complex engineering modifications, as the heat management and safety profiles are favorable due to the ambient operating temperature. This ease of scale-up facilitates rapid technology transfer from R&D to pilot and commercial plants, accelerating the time-to-market for new drug candidates while minimizing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl triazine synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on reaction scope, safety, and scalability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios or research pipelines.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive heavy metal catalysts and harsh reaction conditions. It operates at room temperature in air, significantly simplifying equipment requirements and reducing energy consumption compared to traditional high-temperature or inert atmosphere protocols.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is highly scalable. The use of inexpensive inorganic bases like potassium carbonate and common solvents like THF, combined with the absence of sensitive catalysts, makes it ideal for scaling from gram-level discovery to multi-kilogram commercial manufacturing without complex engineering controls.
Q: What is the structural diversity achievable with this method?
A: The method supports a wide range of substituents. R1 can be alkyl, phenyl, naphthyl, or furyl; R2 can be phenyl or naphthyl; and R3 can be alkyl or aryl groups with various electron-donating or withdrawing substituents, allowing for the creation of diverse libraries for SAR studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the development of next-generation therapeutics and agrochemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of trifluoromethyl substituted 1,2,4-triazine compound delivered meets the highest industry standards for quality and consistency.
We invite you to engage with our technical procurement team to discuss how this innovative chemistry can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this protocol for your specific target molecules. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your development timeline and your bottom line.
