Scalable Synthesis of Polysubstituted Furans Using Dual Metal Catalysis for Pharmaceutical Applications
Introduction to Advanced Furan Synthesis Technology
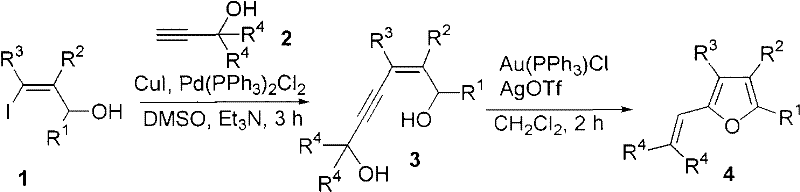
The landscape of organic synthesis for heterocyclic compounds is constantly evolving, driven by the need for more efficient, selective, and scalable methodologies. A pivotal advancement in this field is documented in Chinese Patent CN101792427A, which discloses a novel method for synthesizing polysubstituted furans through a sophisticated dual-catalyst system. This technology represents a significant leap forward for the production of high-value pharmaceutical intermediates, addressing long-standing challenges regarding regioselectivity and functional group tolerance. By leveraging a sequential Sonogashira coupling followed by a gold-catalyzed isomerization cyclization, this process enables the construction of complex furan scaffolds with unprecedented precision. For R&D directors and process chemists, understanding the nuances of this patent is crucial, as it offers a pathway to access chemical space that was previously difficult or expensive to navigate using conventional strong-base mediated reactions. The ability to introduce multiple substituents simultaneously at the 3, 4, and 5 positions, while extending the 2-position substituent from simple alkyl groups to versatile alkenyl moieties, opens new doors for drug discovery and material science applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the furan skeleton has relied heavily on substrates such as allenones, ynones, cyclopropyl ketones, or functionalized epoxides, often necessitating harsh reaction conditions that limit their utility in complex molecule synthesis. Prior art, such as the work by Marshall et al., demonstrated the synthesis of 2-alkenyl furans but was severely constrained by the requirement for strongly basic reaction systems. These alkaline environments are inherently incompatible with base-sensitive functional groups, leading to decomposition or side reactions that drastically reduce overall yield and purity. Furthermore, traditional methods often utilize methoxymethyl ethers as leaving groups, which results in poor substrate compatibility and limits the diversity of structures that can be accessed. For procurement managers, these limitations translate into higher costs due to the need for specialized protecting groups and extensive purification steps to remove byproducts generated under such aggressive conditions. The inability to effectively functionalize the 3, 4, and 5 positions simultaneously has also been a persistent bottleneck, restricting the structural variety available for lead optimization in medicinal chemistry programs.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in CN101792427A employs a mild, metal-catalyzed strategy that circumvents the need for strong bases entirely. This innovative approach utilizes 3-iodo-2-propen-1-ol and terminal propargyl alcohols as starting materials, which undergo a palladium and copper-catalyzed Sonogashira coupling to form a key diol intermediate. This intermediate is then subjected to a gold and silver co-catalyzed isomerization cyclization, efficiently closing the furan ring under neutral conditions. This shift from base-mediated to metal-catalyzed chemistry not only expands the scope of compatible substrates but also significantly simplifies the downstream processing requirements. For supply chain heads, this means a more robust process that is less prone to batch-to-batch variability caused by sensitive reaction parameters. The method's ability to introduce diverse substituents at multiple positions on the furan ring provides a versatile platform for generating libraries of compounds, thereby accelerating the development timeline for new active pharmaceutical ingredients (APIs) and agrochemical agents without compromising on quality or safety standards.
Mechanistic Insights into Au-Catalyzed Isomerization Cyclization
The core of this technological breakthrough lies in the elegant orchestration of two distinct catalytic cycles that work in tandem to build the target molecular architecture. The first stage involves a classic Sonogashira cross-coupling reaction, where the oxidative addition of the aryl or vinyl iodide to the palladium center is followed by transmetallation with the copper-acetylide species formed in situ. This step is critical for establishing the carbon-carbon bond that links the allylic and propargylic fragments, setting the stage for the subsequent ring closure. The second stage, which is the true innovation of this patent, involves the activation of the alkyne moiety by the cationic gold species generated from Au(PPh3)Cl and AgOTf. This activation triggers a cascade of electronic rearrangements, including a 1,2-acyl shift or similar isomerization, which facilitates the nucleophilic attack of the hydroxyl group onto the activated triple bond. This intramolecular cyclization proceeds with high regioselectivity to form the polysubstituted furan ring, driven by the restoration of aromaticity and the stability of the final product. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters, as the interplay between the gold catalyst and the silver salt is essential for generating the active cationic gold species required for efficient turnover.
From an impurity control perspective, this mechanistic pathway offers distinct advantages over traditional acid or base-catalyzed dehydrations. Because the reaction proceeds through a well-defined organometallic cycle rather than a chaotic carbocation or carbanion manifold, the formation of polymeric byproducts or rearrangement isomers is significantly suppressed. The mild conditions, typically ranging from room temperature to 45°C, further minimize thermal degradation of sensitive functional groups, ensuring a cleaner crude reaction profile. This inherent selectivity translates directly into reduced burden on the purification team, as fewer chromatographic fractions are required to isolate the desired product to specification. For quality assurance professionals, the predictability of this mechanism means that potential genotoxic impurities associated with harsh alkylating agents or strong bases are largely avoided. The use of triphenylphosphine ligands on both the palladium and gold centers also helps to stabilize the metal complexes, preventing the formation of metallic precipitates that could complicate filtration and increase heavy metal residues in the final API intermediate.
How to Synthesize Polysubstituted Furans Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires strict adherence to the optimized conditions described in the patent to ensure maximum yield and reproducibility. The process is divided into two distinct operational phases: the initial coupling to generate the acyclic precursor and the subsequent cyclization to form the heterocycle. Operators must maintain an inert atmosphere, typically using nitrogen protection, to prevent oxidation of the sensitive phosphine ligands and copper catalysts during the first step. The precise stoichiometry of the catalysts, particularly the 1:1 molar ratio of the gold complex to the silver salt, is critical for generating the active catalytic species without wasting expensive precious metals. Detailed standard operating procedures (SOPs) should be developed to monitor the reaction progress, likely via TLC or HPLC, to determine the exact quenching point and avoid over-reaction which could lead to decomposition. The following guide outlines the critical procedural steps derived directly from the experimental examples provided in the intellectual property documentation.
- Perform Sonogashira coupling between 3-iodo-2-propen-1-ol and terminal propargyl alcohol using Pd(PPh3)2Cl2 and CuI in DMSO/Et3N at 40-45°C.
- Quench the reaction with saturated ammonium chloride, extract with ether, and purify the intermediate 4-alkyne-2-hexene-1,6-diol via flash column chromatography.
- Subject the intermediate to isomerization cyclization using Au(PPh3)Cl and AgOTf in dichloromethane at room temperature to yield the final polysubstituted furan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this synthetic methodology offers compelling economic and logistical benefits that extend beyond simple yield metrics. The transition from harsh chemical reagents to catalytic systems fundamentally alters the cost structure of manufacturing these valuable intermediates. By eliminating the need for strong bases and specialized leaving groups, the process reduces the consumption of hazardous consumables and minimizes the generation of toxic waste streams, leading to substantial cost savings in waste disposal and environmental compliance. Furthermore, the use of readily available starting materials such as substituted iodo-allyl alcohols and terminal propargyl alcohols ensures a stable supply chain, reducing the risk of production delays caused by sourcing bottlenecks. The mild reaction temperatures, ranging from ambient to moderately warm conditions, also imply lower energy consumption for heating and cooling compared to processes requiring cryogenic temperatures or high-pressure reactors. These factors collectively contribute to a more sustainable and cost-effective manufacturing model that aligns with modern green chemistry principles and corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous strong bases, along with the reduction in purification steps due to cleaner reaction profiles, drives down the overall cost of goods sold (COGS). The catalytic nature of the reaction means that expensive metals are used in small quantities relative to the substrate, and the high turnover number ensures that the cost per kilogram of product remains competitive. Additionally, the avoidance of complex protecting group strategies, which are often necessary in base-sensitive syntheses, further streamlines the synthetic route and reduces material costs. This efficiency allows manufacturers to offer high-purity polysubstituted furans at a more attractive price point, enhancing competitiveness in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and robust catalytic systems mitigates the risks associated with supply chain disruptions. Since the reagents involved are stable and widely produced, there is less dependency on single-source suppliers for exotic precursors. The operational simplicity of the process, which does not require specialized high-pressure equipment or extreme temperature control, allows for greater flexibility in manufacturing locations. This decentralization capability strengthens the supply chain resilience, ensuring consistent delivery schedules even in the face of regional logistical challenges. For long-term planning, this reliability is invaluable for securing the raw material needs of multi-year drug development projects.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions on millimole scales with consistent results that can be extrapolated to kilogram and tonne levels. The mild conditions reduce the thermal load on reactors, making scale-up safer and more predictable from an engineering standpoint. From an environmental perspective, the reduction in hazardous waste and the use of less toxic solvents like dichloromethane (which can potentially be replaced with greener alternatives in further optimization) support stricter environmental regulations. This compliance reduces the regulatory burden and potential fines, safeguarding the company's reputation and operational license.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this furan synthesis technology. These answers are derived from a detailed analysis of the patent specifications and are intended to provide clarity for stakeholders evaluating this route for their specific applications. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation. The responses cover aspects ranging from reaction scope to purification strategies, ensuring a comprehensive overview of the technology's capabilities and limitations.
Q: What are the primary advantages of this gold-catalyzed method over traditional strong base protocols?
A: Unlike traditional methods requiring harsh strong bases which limit substrate compatibility, this patent describes a mild, metal-catalyzed approach that tolerates various functional groups and allows for substitution at the 2, 3, 4, and 5 positions of the furan ring.
Q: What yields can be expected from this synthetic route for commercial production?
A: The patent data indicates robust yields ranging from 68% to 87% across multiple examples, demonstrating high efficiency and reliability suitable for scaling up complex pharmaceutical intermediates.
Q: Is this process scalable for industrial manufacturing of fine chemicals?
A: Yes, the process utilizes readily available raw materials and operates under mild conditions (40-45°C and room temperature), which significantly simplifies thermal management and safety protocols required for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating the development of next-generation therapeutics. Our team of expert chemists has thoroughly analyzed the potential of the gold-catalyzed furan synthesis described in CN101792427A and is fully prepared to leverage this technology for your custom synthesis needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted furan delivered meets the highest industry standards for pharmaceutical intermediates. We understand that consistency and quality are paramount in the supply of fine chemicals, and our robust quality management systems are designed to provide you with total confidence in our products.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall project costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data for our existing furan derivatives or to discuss route feasibility assessments for your proprietary targets. By partnering with NINGBO INNO PHARMCHEM, you gain access not just to a supplier, but to a strategic partner committed to driving your success through chemical innovation and operational excellence.
