Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production with Safe, Metal-Free Catalysis
Revolutionizing 5-Trifluoromethyl-1,2,3-Triazole Production with Safe, Metal-Free Catalysis
In the rapidly evolving landscape of fine chemical manufacturing, the demand for efficient, safe, and scalable synthetic routes for heterocyclic scaffolds is paramount. Patent CN113121462B introduces a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical bottlenecks in traditional synthesis. This technology leverages a base-promoted cyclization strategy that completely bypasses the need for transition metal catalysts and hazardous organic azides. For R&D directors and procurement managers alike, this represents a paradigm shift towards greener, more cost-effective manufacturing of high-value pharmaceutical and agrochemical intermediates. The ability to construct these privileged structures under mild conditions using readily available starting materials positions this methodology as a cornerstone for next-generation process development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,3-triazoles has relied heavily on two primary strategies, both of which present significant operational and safety challenges for industrial scale-up. The first conventional approach involves copper-catalyzed [3+2] cycloaddition reactions between alkynes and organic azides, followed by trifluoromethylation. This pathway is fraught with difficulties, primarily due to the inherent instability and explosive nature of organic azides, which pose severe safety risks in large-scale reactors. Furthermore, the reliance on copper catalysts introduces the persistent issue of heavy metal contamination, necessitating expensive and time-consuming purification steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients. The second traditional method utilizes organocatalytic 1,3-dipolar cycloaddition involving azides and trifluoromethyl ketones, which similarly suffers from the safety hazards associated with handling bulk quantities of azide reagents. These legacy methods often result in complex impurity profiles and elevated production costs due to the specialized safety infrastructure and extensive downstream processing required.
The Novel Approach
In stark contrast to these hazardous legacy protocols, the novel methodology disclosed in the patent utilizes a metal-free, azide-free strategy that fundamentally alters the risk-reward profile of triazole synthesis. By employing trifluoroethylimidoyl chlorides and diazo compounds as starting materials, promoted simply by cesium carbonate, the process achieves high efficiency without the baggage of toxic metals or explosive precursors. 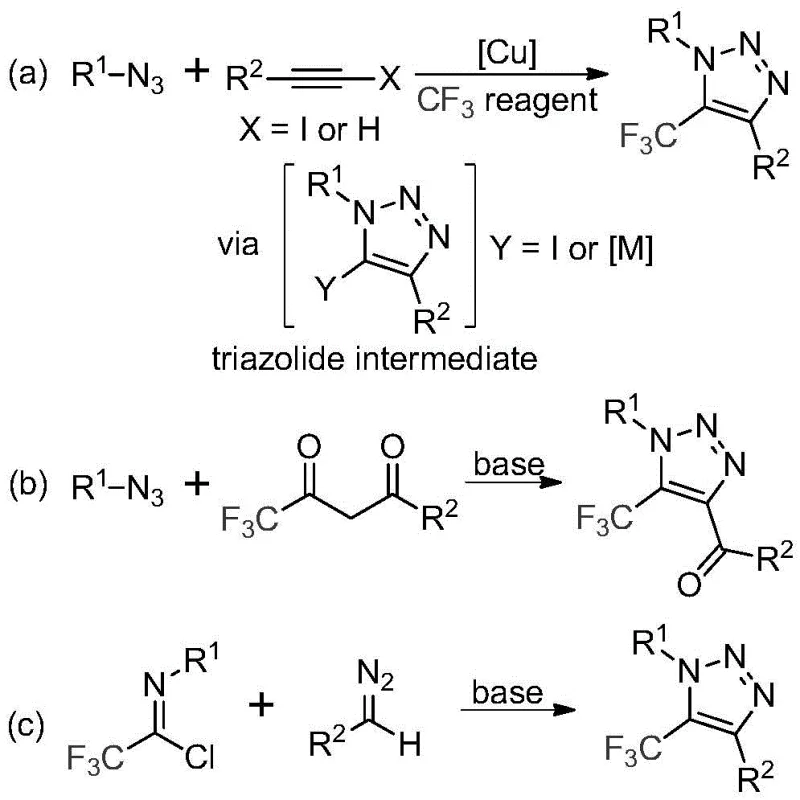 As illustrated in the reaction overview, this new route (Scheme c) offers a direct and atom-economical pathway that avoids the formation of triazole-metal complexes entirely. This simplification of the reaction mechanism translates directly into operational simplicity, allowing for milder reaction temperatures and reduced pressure requirements. For supply chain heads, this means a drastic reduction in the complexity of raw material sourcing, as diazo compounds and imidoyl chlorides are generally more stable and commercially accessible than specialized azide reagents. The elimination of copper also removes the need for scavenger resins or complex extraction protocols, streamlining the workflow from reaction to isolation.
As illustrated in the reaction overview, this new route (Scheme c) offers a direct and atom-economical pathway that avoids the formation of triazole-metal complexes entirely. This simplification of the reaction mechanism translates directly into operational simplicity, allowing for milder reaction temperatures and reduced pressure requirements. For supply chain heads, this means a drastic reduction in the complexity of raw material sourcing, as diazo compounds and imidoyl chlorides are generally more stable and commercially accessible than specialized azide reagents. The elimination of copper also removes the need for scavenger resins or complex extraction protocols, streamlining the workflow from reaction to isolation.
Mechanistic Insights into Base-Promoted Cyclization
The mechanistic elegance of this transformation lies in its reliance on basic promotion rather than transition metal activation. The reaction is hypothesized to proceed through a base-promoted intermolecular nucleophilic addition-elimination sequence. Initially, the diazo compound acts as a nucleophile, attacking the electrophilic carbon of the trifluoroethylimidoyl chloride. This step facilitates the formation of a critical carbon-carbon bond while eliminating a chloride ion, driven by the presence of the inorganic base. Following this initial coupling, the intermediate undergoes an intramolecular 5-endo-dig cyclization. This cyclization step is crucial for closing the triazole ring and establishing the aromatic character of the final product. The use of cesium carbonate is particularly advantageous here; its moderate basicity is sufficient to deprotonate the diazo species or facilitate the elimination without causing decomposition of the sensitive diazo functionality, a balance that stronger bases might fail to maintain. This mechanistic pathway ensures high regioselectivity, predominantly yielding the desired 5-trifluoromethyl isomer with minimal formation of regioisomeric byproducts.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed variants. In copper-catalyzed systems, side reactions often involve Glaser coupling of alkynes or incomplete cycloadditions leading to open-chain byproducts. In this base-promoted system, the primary impurities are likely derived from the hydrolysis of the imidoyl chloride or dimerization of the diazo compound, both of which are chemically distinct and easier to separate via standard chromatographic techniques. The absence of metal-ligand complexes means there is no risk of ligand-derived impurities leaching into the final product. Furthermore, the reaction demonstrates broad functional group tolerance, accommodating various substituents on the aryl rings of both the imidoyl chloride and the diazo compound. This robustness suggests that the electronic properties of the substrates do not drastically alter the reaction pathway, providing a reliable platform for synthesizing diverse libraries of triazole derivatives for SAR studies.
How to Synthesize 5-Trifluoromethyl-1,2,3-Triazole Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of operation, making it highly suitable for both laboratory discovery and pilot plant campaigns. The protocol typically involves charging a reactor with the trifluoroethylimidoyl chloride, the specific diazo compound, and cesium carbonate in an aprotic solvent such as acetonitrile. The mixture is then heated to a moderate temperature range, typically between 50°C and 70°C, and stirred for a duration of 8 to 16 hours. 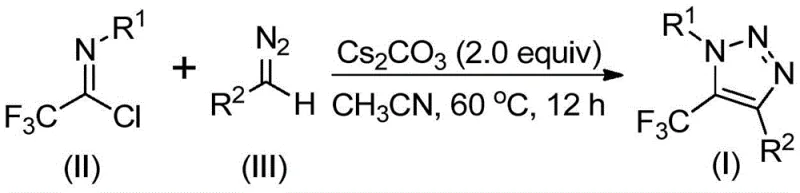 Monitoring the reaction progress via TLC or HPLC ensures complete consumption of the starting materials before proceeding to workup. The post-treatment process is remarkably straightforward, involving simple filtration to remove inorganic salts followed by concentration and purification via column chromatography. This simplicity underscores the process's potential for commercial adoption, as it minimizes unit operations and solvent usage compared to multi-step metal-catalyzed sequences.
Monitoring the reaction progress via TLC or HPLC ensures complete consumption of the starting materials before proceeding to workup. The post-treatment process is remarkably straightforward, involving simple filtration to remove inorganic salts followed by concentration and purification via column chromatography. This simplicity underscores the process's potential for commercial adoption, as it minimizes unit operations and solvent usage compared to multi-step metal-catalyzed sequences.
- Combine cesium carbonate, trifluoroethylimidoyl chloride, and diazo compound in an aprotic organic solvent such as acetonitrile.
- Heat the reaction mixture to a temperature range of 50-70°C and stir for 8 to 16 hours to ensure complete conversion.
- Filter the mixture, concentrate, and purify the crude product via column chromatography to isolate the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this metal-free synthesis route offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The most significant advantage lies in the substantial cost reduction in pharmaceutical intermediate manufacturing achieved by eliminating expensive transition metal catalysts and their associated ligands. Copper salts and specialized phosphine ligands represent a notable portion of the raw material cost in traditional methods, and their removal often requires additional processing steps that consume time and resources. By switching to inexpensive inorganic bases like cesium carbonate, the direct material costs are significantly lowered, improving the overall gross margin of the production process.
- Cost Reduction in Manufacturing: The elimination of transition metals removes the necessity for costly metal scavenging technologies and rigorous testing for residual heavy metals, which are mandatory for API release. This simplification of the downstream processing workflow leads to substantial cost savings in both labor and consumables. Furthermore, the avoidance of explosive azides reduces the capital expenditure required for specialized blast-proof reactor infrastructure and safety monitoring systems. The use of common solvents like acetonitrile, which are easily recovered and recycled, further enhances the economic viability of the process on a multi-ton scale.
- Enhanced Supply Chain Reliability: Sourcing stable, non-hazardous raw materials is a critical factor in maintaining supply chain continuity. Organic azides are often subject to strict transportation regulations and limited supplier availability due to their instability. In contrast, trifluoroethylimidoyl chlorides and diazo compounds are more widely available and easier to transport, reducing the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output even when sourcing from different vendors. This reliability is essential for long-term contracts and just-in-time manufacturing models prevalent in the pharmaceutical industry.
- Scalability and Environmental Compliance: The green chemistry credentials of this method align perfectly with modern environmental, social, and governance (ESG) goals. By generating less hazardous waste and avoiding toxic heavy metals, the process simplifies waste treatment and disposal, lowering environmental compliance costs. The mild reaction conditions reduce energy consumption compared to high-temperature or high-pressure alternatives. Additionally, the high atom economy of the cyclization step minimizes the generation of byproduct waste. These factors collectively make the process highly scalable, allowing for seamless transition from kilogram-level pilot batches to hundred-ton commercial production without the need for major process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, offering clarity for technical teams evaluating this route for their own pipelines.
Q: Why is this metal-free synthesis preferred over copper-catalyzed methods?
A: This method eliminates the need for toxic copper catalysts and explosive organic azides, significantly reducing purification costs and safety risks associated with heavy metal residues in pharmaceutical intermediates.
Q: What are the scalability advantages of using cesium carbonate promotion?
A: The use of inexpensive inorganic bases like cesium carbonate allows for straightforward scale-up from gram to kilogram levels without the complex ligand systems required in transition metal catalysis.
Q: How does this route impact the purity profile of the final API intermediate?
A: By avoiding transition metals, the process inherently reduces the risk of metal contamination, simplifying the impurity profile and facilitating easier compliance with stringent ICH Q3D guidelines for elemental impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the global supply of high-purity pharmaceutical intermediates. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. We possess the rigorous QC labs and analytical capabilities necessary to verify stringent purity specifications, guaranteeing that every batch of 5-trifluoromethyl-1,2,3-triazole meets the exacting standards required for drug substance manufacturing. Our commitment to quality assurance ensures that issues related to residual solvents or trace impurities are proactively managed.
We invite forward-thinking partners to collaborate with us to leverage this advanced synthetic route for their specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this switch can optimize your bill of materials. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most efficient and safe chemical foundations available.
