Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Oncology Drug Development
Advanced Organocatalytic Synthesis of Chiral Indoxazinones for Oncology Drug Development
The pharmaceutical industry is constantly seeking robust, scalable, and stereoselective pathways to access complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. A pivotal advancement in this domain is detailed in Chinese Patent CN112209947A, which discloses a novel synthesis method for chiral indoxazinone compounds. These compounds, characterized by a fused indole-oxazinone core, have garnered significant attention due to their diverse biological profiles, including potential roles as S1P1 receptor antagonists, antimalarial agents, and androgen receptor modulators. The patent highlights a breakthrough organocatalytic strategy that overcomes the limitations of traditional transition-metal catalysis, offering a greener, safer, and highly efficient route to these valuable pharmaceutical intermediates. By leveraging a dual catalytic system involving chiral phosphoric acids and hexafluoroisopropanol, this technology enables the construction of quaternary stereocenters with exceptional precision.
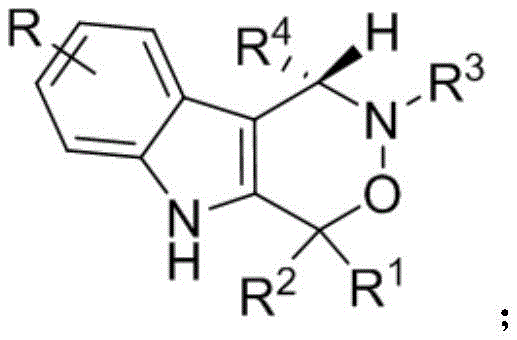
For R&D directors and process chemists, the significance of this invention lies in its ability to generate structural diversity while maintaining rigorous stereochemical control. The general formula (1) depicted above illustrates the core scaffold where various substituents can be introduced to tune biological activity. This flexibility is crucial for medicinal chemistry campaigns aiming to optimize potency and pharmacokinetic properties. Furthermore, the disclosed compounds exhibit notable cytotoxic activity against PC-3 human prostate cancer cells, positioning them as promising candidates for novel antitumor drug research. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such synthetic methodologies allows us to better support clients in bringing these complex molecules from bench to bedside.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral indole-fused ring systems has been fraught with challenges that hinder their widespread adoption in commercial manufacturing. Traditional approaches often rely on transition metal catalysts, which can introduce issues related to heavy metal contamination—a critical concern for regulatory compliance in API production. Moreover, many existing protocols require harsh reaction conditions, such as elevated temperatures or strong acidic/basic environments, which can lead to decomposition of sensitive substrates and the formation of complex impurity profiles. These violent conditions not only pose safety risks in large-scale operations but also result in poor atom economy and lower yields. Additionally, achieving high enantioselectivity with conventional methods frequently necessitates expensive chiral ligands or resolution steps, driving up the overall cost of goods and extending development timelines significantly.
The Novel Approach
In stark contrast, the methodology presented in CN112209947A represents a paradigm shift towards sustainable and efficient organic synthesis. The novel approach utilizes a metal-free organocatalytic system that operates under remarkably mild conditions, typically between 20°C and 30°C. This ambient temperature range eliminates the need for energy-intensive heating or cooling infrastructure, thereby simplifying process engineering requirements. The reaction employs 2-indolemethanol and nitrones as readily available starting materials, which are coupled in the presence of a chiral phosphoric acid catalyst and hexafluoroisopropanol (HFIP) in toluene solvent. This combination facilitates a highly stereoselective cycloaddition or rearrangement process, delivering the target indoxazinones with excellent yields often exceeding 90% and enantiomeric excess (ee) values frequently surpassing 90%. The mildness of the protocol ensures the integrity of functional groups, allowing for a broader substrate scope and reducing the burden on downstream purification processes.
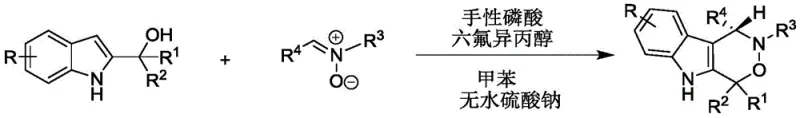
Mechanistic Insights into Chiral Phosphoric Acid Catalysis
The success of this synthetic transformation hinges on the sophisticated interplay between the chiral phosphoric acid catalyst and the hydrogen-bond donor solvent, hexafluoroisopropanol. The chiral phosphoric acid, typically derived from a BINOL (1,1'-bi-2-naphthol) skeleton, acts as a Brønsted acid that activates the electrophilic nitrone species through hydrogen bonding. Simultaneously, the 3,3'-substituents on the phosphoric acid framework (such as the bulky 9-phenanthryl groups shown in the preferred catalyst structure) create a well-defined chiral pocket. This steric environment dictates the facial selectivity of the nucleophilic attack by the 2-indolemethanol derivative, ensuring that the reaction proceeds through a single dominant transition state to yield one specific enantiomer preferentially.
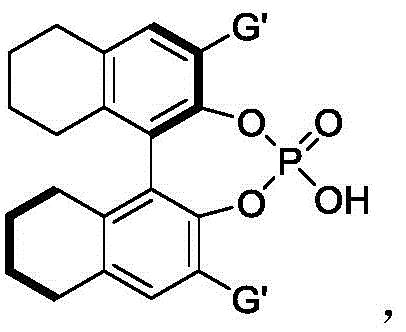
The role of hexafluoroisopropanol (HFIP) is equally critical in this mechanistic landscape. HFIP is known for its unique ability to enhance hydrogen bonding networks and stabilize charged intermediates without acting as a nucleophile itself. In this reaction, HFIP likely cooperates with the chiral phosphoric acid to form a supramolecular catalytic assembly that further rigidifies the transition state. This cooperative catalysis not only accelerates the reaction rate but also amplifies the stereoinduction, leading to the observed high enantioselectivity. From an impurity control perspective, the specificity of this organocatalytic mechanism minimizes side reactions such as polymerization or non-selective background reactions. Consequently, the crude reaction mixtures are cleaner, facilitating easier isolation of the pure product and reducing the generation of hazardous waste, which aligns perfectly with the principles of green chemistry.
How to Synthesize Chiral Indoxazinone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent quality and reaction monitoring to ensure optimal outcomes. The process is designed to be operationally simple, avoiding the need for inert atmosphere techniques or specialized high-pressure equipment, which makes it accessible for most synthetic facilities. The following section outlines the standardized procedure derived from the patent examples, providing a clear roadmap for reproducing these high-value compounds. For detailed standard operating procedures and safety data sheets, please refer to the technical documentation provided below.
- Prepare the reaction mixture by combining 2-indolemethanol and nitrone in toluene solvent with a molar ratio between 1: 1 and 2:1.
- Add anhydrous sodium sulfate as an additive, along with chiral phosphoric acid and hexafluoroisopropanol as the dual catalytic system.
- Stir the reaction at a mild temperature of 20-30°C until completion monitored by TLC, then filter, concentrate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this organocatalytic technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The shift from metal-based catalysis to organocatalysis fundamentally alters the cost structure and risk profile of the manufacturing process. By eliminating the need for expensive transition metals and complex ligand systems, the raw material costs are significantly optimized. Furthermore, the mild reaction conditions reduce energy consumption and mitigate safety hazards associated with exothermic runaway reactions, leading to lower insurance and operational overheads. The robustness of the method across a wide range of substrates ensures supply continuity, as alternative starting materials can be sourced without requiring extensive re-optimization of the process parameters.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive, commercially available organocatalysts and solvents like toluene. Unlike precious metal catalysts that require costly recovery and recycling steps to meet residual metal specifications, the organic catalysts used here simplify the workup procedure. The high yields reported in the patent examples mean that less raw material is wasted, directly improving the mass balance and reducing the cost per kilogram of the final API intermediate. Additionally, the simplified purification steps, often achievable via standard silica gel chromatography or crystallization, lower the processing time and solvent usage, contributing to substantial overall cost savings.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the accessibility of the key starting materials, 2-indolemethanols and nitrones, which can be synthesized from commodity chemicals. The tolerance of the reaction to various functional groups allows for flexibility in sourcing; if one supplier of a specific substituted indole is unavailable, alternative analogs can often be substituted with minimal impact on the reaction outcome. This flexibility reduces the risk of bottlenecks caused by single-source dependencies. Moreover, the stability of the reagents and the simplicity of the reaction setup facilitate easier technology transfer between different manufacturing sites, ensuring consistent quality and availability regardless of geographic location.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the absence of sensitive metal catalysts and the use of standard organic solvents. The mild thermal profile (20-30°C) means that heat management is straightforward, even in large reactors, reducing the risk of thermal incidents. From an environmental standpoint, the metal-free nature of the synthesis simplifies waste treatment, as there is no need for specialized heavy metal scrubbing systems. The high atom economy and reduced solvent requirements align with increasingly stringent environmental regulations, helping companies meet their sustainability goals and avoid potential regulatory fines or delays associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial potential of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this route into your existing production pipelines.
Q: What are the key advantages of this chiral indoxazinone synthesis method?
A: The method described in patent CN112209947A offers extremely high enantioselectivity and yields under mild reaction conditions (20-30°C), avoiding the harsh conditions and safety risks associated with conventional metal-catalyzed routes.
Q: What is the biological activity of the synthesized compounds?
A: The synthesized chiral indoxazinone compounds demonstrate significant cytotoxic activity against PC-3 human prostate cancer cells, with specific derivatives showing IC50 values as low as 40.08 μg/mL, indicating strong potential for oncology drug development.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available raw materials, simple post-treatment procedures (filtration and chromatography), and operates at ambient temperatures, making it highly scalable and economically viable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalytic methods like the one described in CN112209947A for the development of next-generation oncology therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific indoxazinone derivatives or scale-up of the patented route, our infrastructure is designed to support your most demanding projects.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget. Let us be your partner in turning innovative chemistry into commercial reality.
