Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-one Compounds for Commercial Scale-up
The pharmaceutical and agrochemical industries continuously seek efficient pathways to access privileged scaffolds that exhibit potent biological activities. Patent CN112538054B discloses a groundbreaking preparation method for 1,2,4-triazole-3-one compounds, a versatile five-membered nitrogen-containing heterocyclic core. This structural motif is ubiquitous in medicinal chemistry, serving as a key pharmacophore in molecules exhibiting antifungal, anti-inflammatory, antitumor, antiviral, and anticonvulsant properties. As illustrated in the biological context, these compounds function as tyrosinase inhibitors, CB receptor modulators, and angiotensin AT1 receptor antagonists, underlining their immense value in drug discovery pipelines.
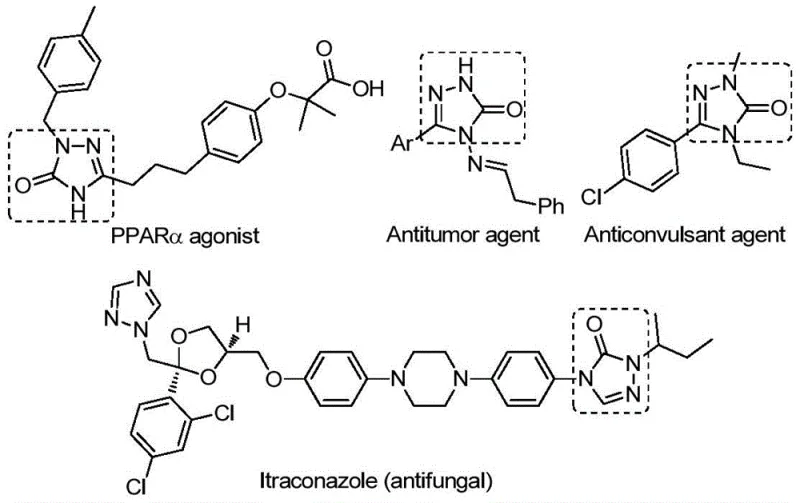
For R&D directors and process chemists, the ability to rapidly access diverse analogues of this scaffold is paramount. The disclosed technology offers a transition metal palladium-catalyzed carbonylation tandem cyclization reaction that transforms readily available chlorohydrazones and sodium azide into the target heterocycles. This approach represents a significant leap forward in synthetic efficiency, addressing long-standing challenges in the construction of triazole rings. By leveraging a robust catalytic system involving Pd2(dba)3 and Xantphos, the method ensures high reaction efficiency and broad substrate compatibility, making it an ideal candidate for the development of reliable pharmaceutical intermediate supplier networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazol-3-one compounds has been plagued by significant operational and economic inefficiencies. Traditional routes often rely on the cyclization of benzoyl hydrazide with urea under strong basic conditions, or the tandem cyclization of hydrazides with isocyanates. These legacy methods frequently suffer from harsh reaction conditions that require extreme temperatures or corrosive reagents, posing safety risks and equipment corrosion issues in large-scale manufacturing. Furthermore, many conventional pathways necessitate the pre-activation of substrates, adding extra synthetic steps that erode overall atom economy and increase waste generation. The narrow substrate scope of these older techniques often limits the chemical diversity accessible to medicinal chemists, while low yields necessitate extensive purification efforts, driving up the cost of goods sold (COGS) for the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the novel palladium-catalyzed protocol described in the patent offers a streamlined, one-pot solution that circumvents these historical bottlenecks. By utilizing cheap and easily obtainable starting materials such as chlorohydrazones and sodium azide, the process drastically simplifies the supply chain logistics. The reaction proceeds through an elegant tandem sequence where carbon monoxide, released in situ from a solid surrogate (TFBen), is inserted into the carbon-palladium bond. This innovation not only avoids the handling of toxic gaseous CO but also facilitates a smooth Curtius rearrangement followed by intramolecular nucleophilic addition to form the triazole ring. The result is a highly efficient transformation that operates under relatively mild conditions (100 °C to 120 °C) in standard aprotic solvents like 1,4-dioxane, delivering high-purity products with excellent yields across a wide range of substrates.

Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Cyclization
Understanding the mechanistic underpinnings of this transformation is critical for process optimization and impurity control. The reaction initiates with the oxidative addition of the palladium(0) catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the solid carbon monoxide source, TFBen, undergoes thermal decomposition to release CO gas within the reaction vessel. This carbon monoxide then inserts into the carbon-palladium bond of the intermediate, forming an acyl-palladium species. This step is pivotal as it introduces the carbonyl functionality required for the triazole ring closure without the need for external pressurized gas cylinders.
Subsequently, the acyl-palladium intermediate reacts with sodium azide to generate an acyl azide compound in situ. This unstable intermediate rapidly undergoes a Curtius rearrangement, losing nitrogen gas to form a highly reactive isocyanate intermediate. The final ring closure occurs via an intramolecular nucleophilic attack by the adjacent nitrogen atom on the isocyanate carbon, yielding the stable 1,2,4-triazole-3-one core. This detailed mechanistic pathway highlights the elegance of the cascade reaction, where multiple bond-forming events occur in a single operational sequence. For quality control teams, understanding that the reaction relies on the precise stoichiometry of the azide and the efficient release of CO helps in monitoring potential side reactions, ensuring that the final impurity profile meets stringent regulatory standards for clinical candidates.
How to Synthesize 1,2,4-Triazole-3-one Efficiently
The practical implementation of this synthesis is designed for scalability and ease of operation, making it suitable for both laboratory discovery and pilot plant production. The procedure involves charging a reactor with the palladium catalyst system, the ligand, the CO surrogate, the chlorohydrazone substrate, and sodium azide in an appropriate organic solvent. The mixture is then heated to facilitate the tandem cyclization. Detailed standardized operating procedures regarding specific molar ratios, solvent volumes, and workup protocols are essential for reproducibility. For a comprehensive guide on executing this synthesis with optimal parameters, please refer to the technical instructions below.
- Combine palladium catalyst (Pd2(dba)3), ligand (Xantphos), CO source (TFBen), chlorohydrazone substrate, and sodium azide in an aprotic organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to a temperature range of 100 °C to 120 °C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, perform filtration to remove solids, followed by silica gel mixing and column chromatography purification to isolate the high-purity 1,2,4-triazole-3-one product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic advantages over traditional manufacturing routes. The shift towards using commodity chemicals like sodium azide and simple chlorohydrazones as starting materials reduces dependency on complex, custom-synthesized precursors that often have long lead times and volatile pricing. This simplification of the bill of materials (BOM) enhances supply chain resilience, ensuring consistent availability of raw materials even during market fluctuations. Furthermore, the use of a solid CO surrogate eliminates the need for specialized high-pressure equipment and safety infrastructure associated with gaseous carbon monoxide, significantly lowering capital expenditure (CAPEX) for facility upgrades.
- Cost Reduction in Manufacturing: The elimination of multi-step pre-activation sequences and the use of inexpensive, commercially available reagents directly contribute to a lower cost base. By consolidating multiple synthetic transformations into a single pot, the process reduces solvent consumption, energy usage, and labor hours associated with intermediate isolations. Although specific percentage savings depend on the specific substrate, the qualitative reduction in unit operations translates to significant margin improvements for high-volume production campaigns.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system allows for the use of a wide variety of substrates without requiring bespoke optimization for each new analogue. This flexibility means that procurement teams can source generic building blocks rather than specialized intermediates, mitigating the risk of single-source supplier bottlenecks. The high conversion rates reported in the patent data suggest that raw material utilization is efficient, minimizing waste disposal costs and maximizing the throughput of existing reactor capacity.
- Scalability and Environmental Compliance: The reaction conditions are compatible with standard stainless steel reactors and do not require exotic materials of construction. The use of 1,4-dioxane, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols. The high atom economy of the tandem reaction minimizes the generation of hazardous byproducts, aligning with modern green chemistry principles and simplifying the environmental permitting process for new manufacturing lines.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and specifications outlined in the patent documentation, providing clarity on reaction scope and operational parameters. Understanding these details is crucial for assessing the feasibility of adopting this route for your specific project needs.
Q: What are the critical reaction parameters for maximizing yield in this synthesis?
A: The patent specifies that maintaining a reaction temperature between 100 °C and 120 °C is crucial. Additionally, the use of aprotic solvents like 1,4-dioxane significantly enhances conversion rates compared to polar aprotic solvents like DMF or DMSO, which were found to be less conducive to the reaction progress.
Q: How does this method handle diverse substrate functional groups?
A: The methodology demonstrates excellent functional group tolerance. It successfully accommodates various substituents on both the R1 and R2 positions, including alkyl groups (n-Bu, t-Bu), aryl groups (phenyl, naphthyl), and halogenated aromatics (4-F-Ph, 4-Br-Ph), allowing for the synthesis of a wide library of derivatives without extensive protection-deprotection strategies.
Q: Is the carbon monoxide source safe for large-scale operations?
A: Yes, the process utilizes TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide substitute. This eliminates the need for handling hazardous gaseous carbon monoxide directly, thereby significantly improving operational safety and simplifying the engineering controls required for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists has extensively evaluated the palladium-catalyzed carbonylation route described in CN112538054B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole-3-one intermediate delivered meets the highest international quality standards.
We invite you to collaborate with us to leverage this advanced technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to contact us today to discuss your requirements, obtain specific COA data for our catalog compounds, and receive detailed route feasibility assessments that will empower your R&D and supply chain strategies.
