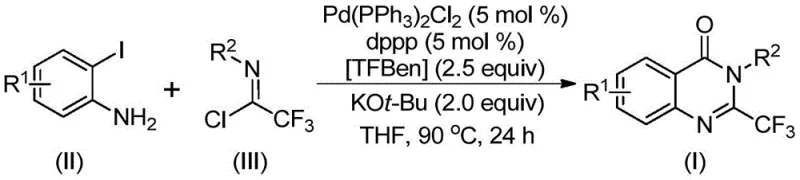
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Commercial API Production
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN112125856A introduces a significant technological advancement in the synthesis of 2-trifluoromethyl substituted quinazolinone derivatives, a class of compounds renowned for their diverse biological activities including anti-inflammatory, antiviral, and anticancer properties. This innovation addresses long-standing challenges in heterocyclic chemistry by employing a transition metal palladium-catalyzed carbonylation tandem reaction that utilizes a solid carbon monoxide substitute. By replacing toxic gaseous carbon monoxide with 1,3,5-tricarboxylate phenol ester (TFBen), the process not only enhances operational safety but also simplifies the engineering requirements for reactor systems. For R&D directors and process chemists, this represents a pivotal shift towards greener and more manageable synthetic routes that maintain high efficiency while mitigating hazardous risks associated with traditional carbonylation protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core, particularly with trifluoromethyl substitution at the 2-position, has relied on methodologies that present substantial logistical and safety hurdles for industrial application. Traditional routes often involve the cyclization of anthranilamides with ethyl trifluoroacetate or trifluoroacetic anhydride, which can suffer from harsh reaction conditions and limited substrate tolerance. Furthermore, methods requiring direct carbon monoxide gas insertion necessitate specialized high-pressure equipment and rigorous safety protocols due to the extreme toxicity and flammability of CO gas. These constraints significantly increase capital expenditure and operational complexity, making scale-up difficult and costly. Additionally, many existing protocols exhibit narrow substrate scopes, failing to accommodate diverse functional groups without extensive protection-deprotection strategies, thereby reducing overall process efficiency and increasing waste generation in a commercial setting.
The Novel Approach
The methodology disclosed in the patent offers a transformative solution by leveraging a palladium-catalyzed tandem reaction that seamlessly integrates C-N bond formation and carbonylation in a single pot. This approach utilizes readily available o-iodoaniline and trifluoroethylimidoyl chloride as starting materials, reacting them in the presence of a palladium catalyst and a solid CO source. The use of TFBen as a carbon monoxide surrogate is particularly groundbreaking, as it releases CO in situ under thermal conditions, effectively bypassing the need for gas cylinders and pressure reactors. This not only democratizes access to carbonylation chemistry for standard laboratory and pilot plant setups but also ensures a controlled release of the reactive species, potentially improving selectivity and yield. The reaction operates at a moderate temperature of 90°C in common organic solvents like THF, demonstrating excellent compatibility with various substituents on the aromatic rings.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The mechanistic pathway of this transformation is a sophisticated orchestration of organometallic steps that ensure high fidelity in product formation. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond coupling between the amine and the imidoyl chloride, generating a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst inserts into the carbon-iodine bond of the aromatic ring, forming a crucial divalent palladium species. As the temperature rises to 90°C, the TFBen additive decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to create an acyl-palladium intermediate. This acyl species undergoes further intramolecular coordination where the base facilitates the formation of a palladium-nitrogen bond, resulting in a seven-membered ring palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone derivative and regenerates the active palladium catalyst for the next turnover.
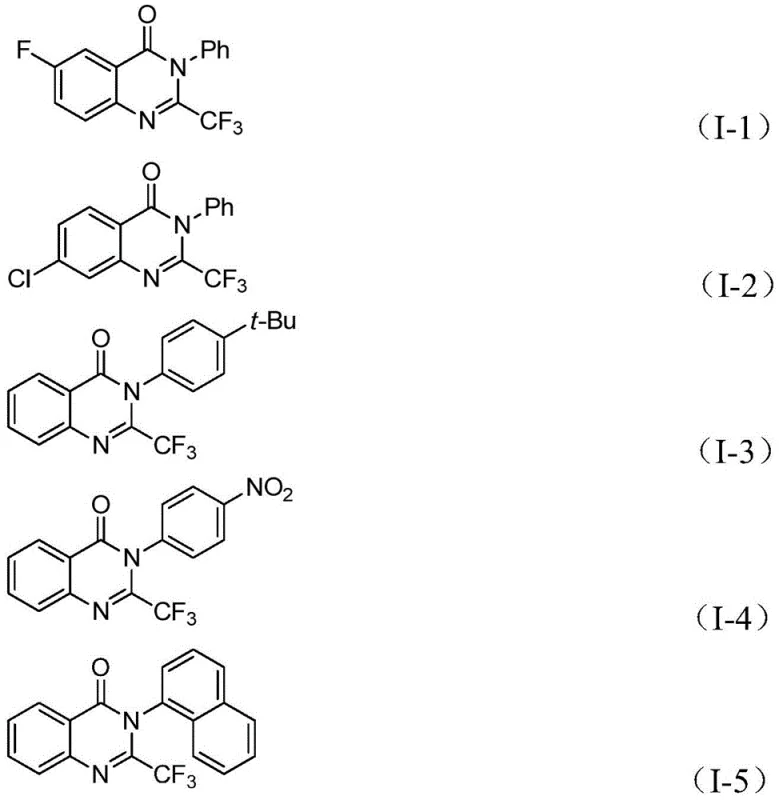
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. By conducting the C-N coupling and carbonylation in a tandem fashion, the accumulation of unstable intermediates is minimized, reducing the likelihood of side reactions such as hydrolysis or polymerization. The use of a specific ligand system, typically involving dppp alongside the palladium source, stabilizes the catalytic cycle and prevents the formation of palladium black, which can lead to catalyst deactivation and metal contamination in the final product. The broad substrate scope demonstrated in the patent, accommodating electron-withdrawing and electron-donating groups alike, suggests that the electronic demands of the oxidative addition and migratory insertion steps are well-balanced. This robustness ensures that the impurity profile remains consistent and manageable across different batches, a critical factor for meeting stringent pharmaceutical quality standards.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling, making it an ideal candidate for technology transfer from lab to plant. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, base, solid CO source, and the two primary organic substrates in an aprotic solvent. The mixture is then heated to facilitate the tandem reaction sequence. Detailed standardized synthesis steps follow below, outlining the precise molar ratios and workup procedures required to achieve the high yields reported in the patent data. This streamlined process eliminates the need for specialized gas handling infrastructure, allowing facilities to produce high-value intermediates with existing equipment.
- Combine palladium catalyst, ligand, base, solid CO source, and reactants in solvent.
- Heat the mixture to 90°C and maintain reaction for 16 to 30 hours.
- Perform filtration and column chromatography to isolate the pure derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route translates into tangible strategic benefits regarding cost structure and risk management. The elimination of toxic carbon monoxide gas from the supply chain removes a significant regulatory and safety burden, reducing the need for specialized storage facilities and emergency response protocols. This simplification of the hazard profile allows for more flexible manufacturing site selection and lowers insurance and compliance costs associated with handling extremely hazardous substances. Furthermore, the reliance on commodity chemicals like o-iodoaniline and commercially available palladium catalysts ensures a stable and competitive supply base, mitigating the risk of raw material shortages that often plague niche synthetic routes.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the replacement of expensive or hazardous reagents with cost-effective alternatives. By utilizing a solid CO source, the process avoids the capital intensity associated with high-pressure carbonylation reactors, leading to significant depreciation savings. Additionally, the high conversion rates and broad substrate tolerance minimize the loss of valuable starting materials, optimizing the overall material balance. The simplified workup procedure, involving basic filtration and chromatography, reduces solvent consumption and processing time, contributing to a lower cost of goods sold without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials that are not subject to the same geopolitical or logistical constraints as specialized gases. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by minor variations in utility supply or environmental conditions. This reliability ensures consistent delivery timelines to downstream customers, fostering stronger long-term partnerships. The ability to scale the reaction without fundamental changes to the chemistry further supports the seamless transition from clinical trial material to commercial volume production.
- Scalability and Environmental Compliance: From an environmental standpoint, the use of a solid CO surrogate aligns with green chemistry principles by reducing the potential for atmospheric release of toxic gases. The process generates less hazardous waste compared to traditional methods that might require stoichiometric amounts of activating agents. The scalability is inherent in the design, as the reaction does not rely on mass transfer limitations typical of gas-liquid reactions. This makes the process highly adaptable to larger reactor volumes, ensuring that increased demand can be met efficiently while maintaining strict adherence to environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on the operational parameters and expected outcomes. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What is the primary safety advantage of this synthesis method?
A: The method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide substitute, eliminating the need for handling toxic and hazardous carbon monoxide gas directly.
Q: What are the typical reaction conditions for this transformation?
A: The reaction typically proceeds in an organic solvent like THF at 90°C for 16 to 30 hours, using a palladium catalyst system with a phosphine ligand and potassium tert-butoxide base.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the process uses commercially available starting materials, operates under relatively mild conditions without high-pressure gas equipment, and involves simple post-treatment steps, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization. Our team of expert process chemists has extensively evaluated the palladium-catalyzed carbonylation route described in CN112125856A and is fully prepared to execute this chemistry at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle sensitive catalytic reactions with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards.
We invite you to collaborate with us to leverage this innovative technology for your next project. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise in complex heterocycle synthesis can drive value and efficiency for your organization.
