Advanced Manufacturing of 5-Cyanoindole: A Safe and Scalable Seven-Step Synthetic Strategy
The pharmaceutical industry continuously seeks robust synthetic pathways for critical heterocyclic intermediates, particularly those serving as scaffolds for treating depression, malignancies, and cardiovascular diseases. Patent CN116082213A introduces a groundbreaking preparation method for 5-cyanoindole, addressing the severe safety limitations inherent in traditional manufacturing protocols. This technical disclosure outlines a sophisticated seven-step sequence that transforms inexpensive o-chloroaniline into high-purity 5-cyanoindole through a series of controlled catalytic reactions. By shifting away from hazardous diazotization processes, this innovation offers a paradigm shift in how fine chemical manufacturers approach the production of complex indole derivatives. The methodology emphasizes environmental friendliness, operational safety, and scalability, making it an ideal candidate for commercial adoption by global supply chains demanding rigorous compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-cyanoindole has relied heavily on the diazotization of p-cyanoaniline followed by reduction to form cyanophenylhydrazine, which is subsequently cyclized with aldehydes. While theoretically concise, this legacy pathway presents unacceptable risks for modern industrial applications. The generation of diazonium salts is notoriously unstable, carrying a high potential for thermal runaway and explosive decomposition, especially when handled in large quantities. Furthermore, the handling of hydrazine intermediates introduces significant toxicity concerns, requiring specialized containment infrastructure that drives up capital expenditure. These safety bottlenecks severely restrict the ability to scale production to meet the growing global demand for antidepressant and anticancer medications derived from this scaffold.
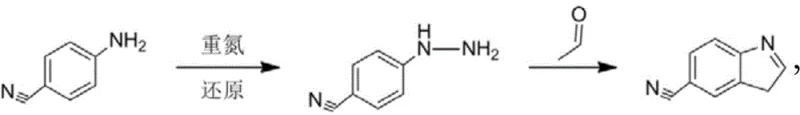
The Novel Approach
In stark contrast, the methodology disclosed in CN116082213A employs a linear, step-wise construction of the indole core starting from stable aromatic amines. This novel approach bypasses the formation of explosive diazonium species entirely, utilizing a strategic sequence of nucleophilic substitution, hydrolysis, and intramolecular cyclization. The process leverages common industrial solvents such as ethanol and DMF, alongside standard reagents like dimethyl malonate and N-bromosuccinimide, ensuring that raw material sourcing remains resilient against market fluctuations. By decomposing the synthesis into seven distinct, manageable transformations, the protocol allows for precise control over reaction parameters at each stage, thereby minimizing impurity carryover and maximizing overall yield without compromising operator safety.
Mechanistic Insights into the Seven-Step Catalytic Cascade
The core of this synthetic strategy lies in the efficient construction of the indole ring system through a carefully orchestrated redox and condensation sequence. The process initiates with the nucleophilic attack of o-chloroaniline on dimethyl malonate, facilitated by a basic catalyst to form the key malonate intermediate. Subsequent hydrolysis and decarboxylation steps cleanly generate o-aminophenylacetic acid, setting the stage for regioselective bromination. The introduction of the bromine atom at the para-position relative to the amino group is critical, as it serves as the handle for the final cyanation. The intramolecular condensation step, mediated by coupling agents like CDI or EDCI, effectively closes the five-membered ring to form the oxindole skeleton, demonstrating high atom economy and selectivity under mild thermal conditions.
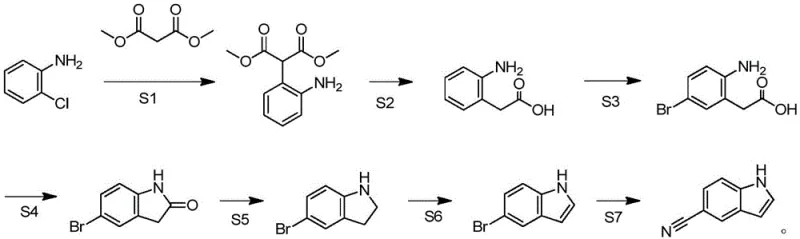
Following ring closure, the pathway employs a reduction-oxidation strategy to aromatize the system. The reduction of the oxindole carbonyl to the indoline using borane complexes proceeds with high fidelity, avoiding over-reduction of the aromatic ring. The subsequent oxidation step, whether utilizing activated manganese dioxide or catalytic oxygen systems, restores the aromaticity to yield 5-bromoindole. Finally, the displacement of the bromine atom with a cyano group using cuprous cyanide completes the synthesis. This final substitution is particularly robust, tolerating the indole nitrogen and ensuring that the nitrile functionality is installed without degrading the sensitive heterocyclic core, resulting in a product with exceptional chemical purity.
How to Synthesize 5-Cyanoindole Efficiently
The implementation of this seven-step protocol requires strict adherence to the optimized reaction conditions detailed in the patent examples to ensure maximum efficiency and safety. From the initial condensation temperatures to the specific workup procedures for the bromination step, every parameter has been tuned to minimize waste and maximize throughput. For R&D teams looking to replicate or adapt this process, understanding the stoichiometry of the coupling agents and the precise control of pH during the hydrolysis phase is paramount. The detailed standardized synthesis steps below provide a comprehensive guide for executing this route in a pilot or production environment.
- Condense o-chloroaniline with dimethyl malonate using a basic catalyst to form 2-(2-aminophenyl)dimethyl malonate.
- Hydrolyze the malonate ester with alkali followed by acid-catalyzed high-temperature decarboxylation to yield o-aminophenylacetic acid.
- Perform para-bromination on the amino group using NBS to obtain 2-(2-amino-5-bromophenyl)acetic acid.
- Execute intramolecular condensation with a coupling agent like CDI to close the ring, forming 5-bromooxindole.
- Reduce the oxindole carbonyl group using borane dimethyl sulfide to generate 5-bromoindoline.
- Oxidize 5-bromoindoline using manganese dioxide or catalytic oxygen systems to produce 5-bromoindole.
- Substitute the bromine atom with a cyano group using cuprous cyanide to finalize the 5-cyanoindole structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers profound strategic benefits beyond mere technical feasibility. The elimination of hazardous diazotization steps fundamentally alters the risk profile of the manufacturing process, leading to substantial reductions in insurance premiums and safety compliance costs. Furthermore, the reliance on commodity chemicals rather than specialized, high-risk reagents ensures that the supply chain remains agile and resistant to disruptions caused by regulatory crackdowns on dangerous substances. This stability is crucial for maintaining continuous production schedules in the highly regulated pharmaceutical sector.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive starting materials like o-chloroaniline and avoiding the need for cryogenic conditions often required for diazonium stability. The high conversion rates observed in the condensation and cyclization steps reduce the volume of solvent and energy required for purification, directly lowering the cost of goods sold. Additionally, the avoidance of expensive transition metal catalysts in favor of more accessible reagents further contributes to a leaner cost structure.
- Enhanced Supply Chain Reliability: By sourcing raw materials that are widely produced for other industrial applications, manufacturers can mitigate the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to minor variations in utility supply or environmental conditions, ensuring consistent delivery timelines. This reliability is essential for downstream API manufacturers who operate on tight just-in-time inventory models and cannot afford delays in intermediate supply.
- Scalability and Environmental Compliance: The mild reaction temperatures and absence of toxic hydrazine by-products simplify waste treatment protocols, aligning with increasingly stringent environmental regulations. The process is inherently designed for scale-up, with exothermic risks managed through controlled addition rates rather than complex engineering controls. This ease of scaling allows producers to rapidly respond to market demand surges without the lengthy validation periods associated with hazardous chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on safety, purity, and operational feasibility for potential partners.
Q: Why is the conventional diazotization route for 5-cyanoindole considered unsafe for large-scale production?
A: The traditional method involves the diazotization of p-cyanoaniline, which generates unstable diazonium salts. These intermediates pose significant explosion risks and require stringent temperature control, making them unsuitable for industrial scale-up due to safety hazards and operational complexity.
Q: How does the new seven-step synthesis improve supply chain reliability for pharmaceutical manufacturers?
A: The novel route utilizes readily available starting materials like o-chloroaniline and dimethyl malonate, avoiding specialized hazardous reagents. The mild reaction conditions and absence of explosive intermediates ensure consistent batch-to-batch quality and uninterrupted production schedules, significantly enhancing supply security.
Q: What are the key purification advantages of the intramolecular condensation step in this process?
A: The intramolecular condensation using coupling agents like CDI proceeds under mild conditions with high conversion rates. This specificity minimizes the formation of polymeric by-products and side-reaction impurities, simplifying downstream purification and resulting in a final product with superior purity profiles suitable for API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Cyanoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality intermediates for your drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 5-cyanoindole meets the exacting standards required for pharmaceutical synthesis, guaranteeing performance in your downstream reactions.
We invite you to engage with our technical procurement team to discuss how this advanced synthetic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and reliability in your manufacturing operations.
