Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-One Intermediates for Commercial Scale-Up
Introduction to Next-Generation Triazole Synthesis
The pharmaceutical industry continuously seeks robust methodologies for constructing nitrogen-containing heterocycles, particularly the 1,2,4-triazole-3-one scaffold, which serves as a critical pharmacophore in numerous bioactive agents. As detailed in patent CN112538054B, a groundbreaking preparation method has been developed that addresses long-standing inefficiencies in synthesizing these valuable compounds. This innovation leverages a transition metal palladium-catalyzed carbonylation tandem cyclization reaction, transforming simple chlorohydrazones and sodium azide into complex heterocyclic structures with remarkable efficiency. The significance of this technology extends beyond academic interest; it provides a viable pathway for the commercial scale-up of complex pharmaceutical intermediates, offering a solution that is both economically attractive and operationally straightforward for modern manufacturing facilities.
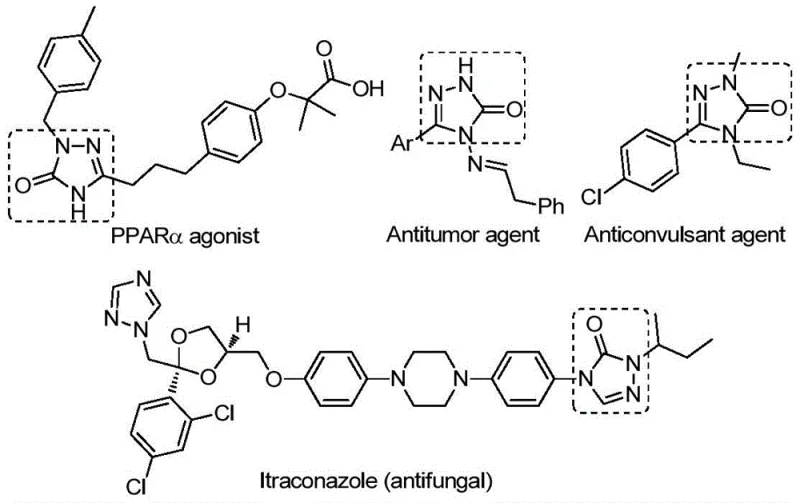
The versatility of the 1,2,4-triazole-3-one core cannot be overstated, as it underpins a wide array of therapeutic applications including antifungal, anti-inflammatory, antitumor, and anticonvulsant activities. By enabling the rapid assembly of this core structure with diverse substituents, this patented method empowers medicinal chemists to explore broader chemical spaces for drug discovery. For procurement and supply chain leaders, the adoption of such efficient synthetic routes translates directly into cost reduction in API manufacturing, as it minimizes waste, reduces reaction times, and utilizes inexpensive starting materials. This report analyzes the technical merits and commercial implications of this synthesis, positioning it as a key technology for reliable pharmaceutical intermediate supplier networks aiming to enhance their portfolio capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazole-3-one compounds has been plagued by significant operational hurdles that hinder large-scale production. Traditional literature reports describe methods such as the cyclization of benzoyl hydrazide with urea under potassium hydroxide action, or the tandem cyclization of hydrazides with isocyanates. These legacy processes often suffer from harsh reaction conditions, requiring extreme temperatures or pressures that pose safety risks and increase energy consumption. Furthermore, many conventional routes necessitate the pre-activation of reaction substrates, adding extra synthetic steps that lower the overall atom economy and increase the generation of chemical waste. The narrow substrate scope of these older methods also limits their utility, as they frequently fail to tolerate sensitive functional groups, thereby restricting the structural diversity accessible to researchers and forcing reliance on low-yielding alternatives.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN112538054B introduces a streamlined catalytic cycle that bypasses these traditional bottlenecks. By employing a palladium catalyst system in conjunction with TFBen (1,3,5-tricarboxylic acid phenol ester) as a safe solid carbon monoxide substitute, the reaction proceeds smoothly in organic solvents like 1,4-dioxane at moderate temperatures of 100°C. This approach eliminates the need for handling hazardous CO gas cylinders, significantly enhancing workplace safety and simplifying reactor setup. The reaction demonstrates exceptional substrate compatibility, accommodating various aryl and alkyl groups without the need for protective group strategies. This operational simplicity, combined with high reaction efficiency, establishes a new standard for the high-purity 1,2,4-triazole-3-one derivatives production, making it an ideal candidate for integration into existing fine chemical manufacturing lines.

Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize the process for specific analogues. The reaction is believed to initiate with the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, generating a reactive divalent palladium intermediate. Simultaneously, the TFBen additive undergoes thermal decomposition to release carbon monoxide in situ, which then inserts into the carbon-palladium bond to form an acyl-palladium species. This acyl intermediate subsequently reacts with sodium azide to generate an acyl azide compound, which is the precursor for the key ring-closing step. The elegance of this mechanism lies in its seamless integration of carbonylation and cyclization events within a single pot, avoiding the isolation of unstable intermediates.
Following the formation of the acyl azide, the system undergoes a Curtius rearrangement to produce an isocyanate intermediate, a highly reactive species that immediately participates in an intramolecular nucleophilic addition. This final cyclization step closes the five-membered triazole ring, yielding the target 1,2,4-triazole-3-one product with high regioselectivity. The robustness of this catalytic cycle is evidenced by the broad scope of substrates tolerated, as shown in the patent examples where various substituted phenyl and naphthyl groups successfully participate in the reaction. This mechanistic clarity allows for precise tuning of reaction parameters, ensuring that reducing lead time for high-purity pharmaceutical building blocks is achieved without compromising on the structural integrity or purity of the final active pharmaceutical ingredient intermediates.

How to Synthesize 1,2,4-Triazole-3-One Efficiently
The practical execution of this synthesis is designed to be accessible for laboratory and pilot plant settings alike. The protocol involves charging a reaction vessel with the palladium catalyst Pd2(dba)3, the Xantphos ligand, the solid CO source TFBen, the chlorohydrazone starting material, and sodium azide in a suitable aprotic solvent. The mixture is then heated to 100°C and stirred for a period ranging from 16 to 30 hours, depending on the specific electronic nature of the substrate. Upon completion, the workup procedure is remarkably simple, involving filtration to remove inorganic salts followed by silica gel mixing and standard column chromatography purification. This straightforward workflow minimizes the need for specialized equipment or complex quenching procedures, facilitating rapid technology transfer.
- Combine palladium catalyst Pd2(dba)3, Xantphos ligand, TFBen carbon monoxide source, chlorohydrazone substrate, and sodium azide in an organic solvent such as 1,4-dioxane.
- Heat the reaction mixture to 100°C and maintain stirring for a duration of 16 to 30 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product via column chromatography to isolate the target 1,2,4-triazole-3-one compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of adopting this patented synthesis route are profound. The shift from multi-step, harsh traditional methods to this one-pot catalytic process represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. By utilizing cheap and readily available starting materials like chlorohydrazones and sodium azide, the direct material costs are substantially lowered compared to routes requiring exotic or pre-activated reagents. Furthermore, the use of TFBen as a solid CO source removes the logistical burden and safety costs associated with storing and transporting high-pressure carbon monoxide gas, thereby simplifying regulatory compliance and insurance overheads.
- Cost Reduction in Manufacturing: The elimination of pre-activation steps and the high atom economy of the tandem reaction significantly reduce the consumption of reagents and solvents. Since the reaction proceeds with high efficiency and minimal byproduct formation, the downstream purification burden is lessened, leading to lower processing costs per kilogram of product. The ability to achieve high yields, reaching up to 96% in optimized cases, ensures that raw material investment is maximized, providing a clear financial advantage over lower-yielding legacy processes.
- Enhanced Supply Chain Reliability: The reliance on commercially available, stable reagents ensures a consistent supply of inputs, mitigating the risk of production delays caused by raw material shortages. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, resulting in more predictable batch outcomes. This reliability is critical for maintaining continuous production schedules and meeting the stringent delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The method has been demonstrated to be scalable, with the patent noting its applicability for medium-scale production applications. The use of safer reagents and the generation of less hazardous waste align with modern green chemistry principles, simplifying waste disposal and environmental reporting. This ease of scale-up from milligram to multi-kilogram quantities ensures that the technology can grow with demand, supporting the commercial scale-up of complex heterocyclic intermediates without requiring massive capital expenditure on new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a transparent view of the process capabilities. Understanding these details helps stakeholders make informed decisions about integrating this method into their supply chains.
Q: What are the primary advantages of this Pd-catalyzed method over traditional synthesis routes?
A: Unlike traditional methods that require harsh conditions, pre-activation of substrates, or toxic reagents, this novel approach utilizes cheap and readily available chlorohydrazones and sodium azide. It operates under milder conditions with high substrate compatibility and significantly improved reaction efficiency.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the patent explicitly demonstrates that the method is practical for scale-up. The use of stable solid reagents like TFBen as a CO source eliminates the safety hazards of handling carbon monoxide gas, making it highly suitable for large-scale manufacturing environments.
Q: What is the expected purity and yield range for these triazole derivatives?
A: Experimental data indicates isolated yields ranging from 48% to 96% depending on the substrate substituents. The process allows for standard purification via column chromatography, ensuring high-purity specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the Pd-catalyzed carbonylation described in CN112538054B. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole-3-one intermediate delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific drug development programs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your project's unique requirements, demonstrating exactly how this efficient route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market.
