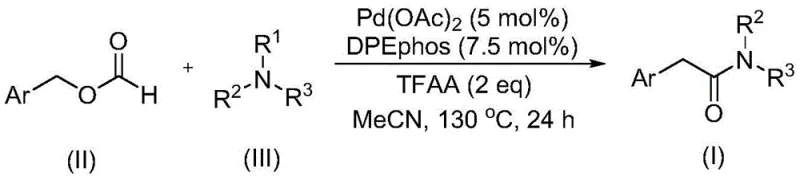
Advanced Palladium-Catalyzed Synthesis of Aryl Acetamides for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand robust synthetic routes for complex amide scaffolds, particularly aryl acetamides, which serve as critical building blocks in numerous bioactive molecules. A significant technological advancement in this domain is detailed in Chinese Patent CN111978194B, which discloses a novel preparation method for aryl acetamide compounds. This innovation addresses long-standing challenges in amide bond formation by utilizing a transition metal-catalyzed carbonylation strategy that bypasses the limitations of traditional amidation. The core of this methodology lies in the strategic use of benzyl formate as a dual-function reagent, acting simultaneously as a carbon monoxide source and a reactant, paired with readily available tertiary amines. For R&D directors and process chemists seeking reliable aryl acetamide supplier partnerships, this patent represents a pivotal shift towards safer, more efficient, and highly designable synthetic pathways that avoid the logistical nightmares associated with gaseous carbon monoxide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl acetamide compounds has relied heavily on the direct amidation of phenylacetic acid and its derivatives, a process often plagued by harsh reaction conditions and poor atom economy. Alternatively, transition metal-catalyzed carbonylation reactions have emerged as a powerful alternative; however, these traditional methods predominantly utilize primary and secondary amines as nitrogen sources. The activation of the C-N bond in tertiary amines remains a formidable obstacle in organic synthesis due to the lack of an N-H proton and the stability of the tertiary amine structure. Furthermore, existing protocols for tertiary amine carbonylation frequently necessitate the use of excessive oxidants or explosive mixtures of carbon monoxide and oxygen, posing severe safety risks and complicating the scale-up process for commercial manufacturing. These inherent drawbacks limit the substrate scope and increase the operational costs, making the production of complex aryl acetamides derived from tertiary amines economically and technically unfeasible for many applications.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN111978194B introduces a streamlined catalytic system that effectively overcomes the barriers of tertiary amine activation. By employing benzyl formate, a stable liquid surrogate for carbon monoxide, the reaction eliminates the need for high-pressure gas infrastructure and mitigates explosion hazards. The method utilizes a palladium catalyst in conjunction with a specialized ligand and trifluoroacetic anhydride to facilitate the cleavage of the tertiary amine C-N bond without the addition of external oxidants. This breakthrough not only simplifies the reaction setup but also significantly broadens the tolerance for various functional groups on the aryl ring, including alkyl, alkoxy, trifluoromethyl, and halogen substituents. For procurement managers focused on cost reduction in pharmaceutical intermediates manufacturing, this approach offers a pathway to high-purity products using cheap and widely available starting materials, thereby optimizing the overall supply chain economics.
Mechanistic Insights into Pd-Catalyzed Carbonylation with Benzyl Formate
The mechanistic elegance of this synthesis lies in the in situ generation of reactive species that drive the carbonylation forward under relatively mild thermal conditions. The reaction initiates with the coordination of the palladium catalyst, typically palladium acetate, with the bidentate phosphine ligand DPEphos, forming an active catalytic species capable of oxidative addition or insertion processes. Benzyl formate undergoes decomposition or activation within this catalytic cycle to release carbon monoxide equivalents directly at the metal center, which then inserts into the palladium-carbon or palladium-nitrogen bond. Crucially, the presence of trifluoroacetic anhydride likely assists in activating the formate or stabilizing intermediates, facilitating the difficult C-N bond cleavage of the tertiary amine. This intricate interplay allows the transformation to proceed with high efficiency, converting simple precursors into valuable amide structures while maintaining the integrity of sensitive functional groups that might otherwise degrade under oxidative conditions.
From an impurity control perspective, the absence of external oxidants is a major advantage, as it prevents the formation of over-oxidized byproducts such as N-oxides or degraded aromatic rings that are common in oxidative carbonylation protocols. The specificity of the palladium-DPEphos system ensures that the reaction proceeds selectively towards the desired aryl acetamide, minimizing the generation of complex impurity profiles that are difficult to remove during downstream processing. This high level of chemoselectivity is paramount for producing high-purity aryl acetamides required for regulatory compliance in drug substance manufacturing. The ability to tolerate diverse substituents on the aryl group, such as electron-withdrawing trifluoromethyl groups or electron-donating methoxy groups, further demonstrates the robustness of the catalytic cycle, allowing for the synthesis of a wide library of analogues without needing to re-optimize reaction conditions for each new substrate.
How to Synthesize Aryl Acetamide Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly suitable for both laboratory-scale optimization and industrial production. The process involves combining the palladium catalyst, ligand, benzyl formate, tertiary amine, and trifluoroacetic anhydride in a polar aprotic solvent such as acetonitrile. The mixture is then heated in a sealed vessel to temperatures ranging from 120°C to 140°C, typically around 130°C, for a duration of approximately 24 hours. This thermal profile ensures complete conversion of the starting materials while maintaining the stability of the catalytic system. Following the reaction, the workup procedure is straightforward, involving filtration to remove catalyst residues, followed by silica gel treatment and standard column chromatography purification. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Combine palladium acetate, DPEphos ligand, benzyl formate, tertiary amine, and trifluoroacetic anhydride in an organic solvent such as acetonitrile.
- Heat the reaction mixture to 130°C in a sealed tube and maintain stirring for approximately 24 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target aryl acetamide compound.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this patented methodology offers substantial strategic benefits beyond mere chemical efficiency. The reliance on benzyl formate and tertiary amines as starting materials leverages a supply chain of commodity chemicals that are abundant and cost-effective, reducing dependency on specialized or hazardous reagents. By eliminating the need for high-pressure carbon monoxide cylinders and the associated safety infrastructure, facilities can significantly lower their capital expenditure and operational risk profiles. This shift translates into a more resilient supply chain capable of sustaining continuous production without the interruptions often caused by the logistics of handling dangerous gases. Furthermore, the simplified post-treatment process reduces the consumption of solvents and purification media, contributing to a greener manufacturing footprint that aligns with increasingly stringent environmental regulations.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive raw materials such as formic acid derivatives and common tertiary amines, which are produced on a massive global scale. By avoiding the use of expensive oxidants and specialized high-pressure equipment, the overall cost of goods sold (COGS) for the final aryl acetamide product is drastically reduced. The high reaction efficiency and yield reported in the patent data mean that less raw material is wasted, maximizing the output per batch and improving the return on investment for manufacturing campaigns. Additionally, the simplified purification workflow lowers the labor and utility costs associated with downstream processing, further enhancing the margin potential for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term production contracts, and this method excels by utilizing reagents with established, multi-vendor supply chains. Benzyl formate and the requisite palladium catalysts are commercially available from numerous global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions allows for flexibility in manufacturing scheduling, as the process is less sensitive to minor variations in reagent quality compared to more finicky oxidative protocols. This reliability ensures consistent delivery timelines for clients, reducing lead time for high-purity aryl acetamides and enabling just-in-time inventory strategies that minimize warehousing costs.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often introduces unforeseen challenges, but the homogeneous nature of this catalytic system facilitates smooth scale-up in standard stainless steel reactors. The absence of explosive gas mixtures removes a major barrier to increasing batch sizes, allowing for safer operation at higher volumes. From an environmental standpoint, the atom economy of using benzyl formate as a CO source is superior to traditional methods, resulting in less chemical waste. The ability to conduct the reaction without external oxidants also reduces the load on wastewater treatment systems, as there are fewer oxidative byproducts to neutralize, supporting sustainable manufacturing practices and compliance with green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for process development teams. Understanding these nuances is essential for evaluating the feasibility of this route for specific target molecules and for planning effective technology transfer activities.
Q: What is the primary advantage of using benzyl formate in this synthesis?
A: Benzyl formate serves as a safe and effective in situ source of carbon monoxide, eliminating the need for handling hazardous high-pressure CO gas while acting as a reactant in the carbonylation process.
Q: Can tertiary amines be utilized directly in this carbonylation reaction?
A: Yes, this method uniquely enables the cleavage of the C-N bond in tertiary amines without requiring additional external oxidants, which is a significant challenge in conventional amide synthesis.
Q: What catalyst system is recommended for optimal yield?
A: The patent specifies a system using Palladium(II) acetate (Pd(OAc)2) as the catalyst and DPEphos as the ligand, which provides high efficiency and broad functional group tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Acetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111978194B for accelerating drug discovery and development pipelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with the highest safety standards, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of aryl acetamide meets the exacting requirements of the global pharmaceutical market. We are committed to delivering high-quality intermediates that empower our partners to bring life-saving therapies to patients faster.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for cost reduction in pharmaceutical intermediates manufacturing.
