Scalable Metal-Free Synthesis of High-Purity 5-Trifluoromethyl Triazoles for Pharmaceutical Manufacturing
The patent CN113121462B introduces a groundbreaking metal-free synthesis route for 5-trifluoromethyl substituted 1,2,3-triazole compounds, addressing critical limitations in conventional manufacturing methods. This innovation eliminates the need for toxic azide reagents and transition metal catalysts while maintaining high reaction efficiency under mild conditions (60°C). The process demonstrates exceptional versatility across diverse substrate combinations, with yields ranging from 40% to 91% as validated through extensive experimental data. By leveraging readily available starting materials and simplified purification protocols, this method establishes a new paradigm for producing high-purity triazole intermediates essential in pharmaceutical and agrochemical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of trifluoromethyl-substituted triazoles relies on two primary approaches: copper-catalyzed [3+2] cycloaddition of alkynes with organic azides followed by trifluoromethylation, or organocatalytic reactions between azides and trifluoromethyl ketones. Both methods present significant operational hazards due to the inherent instability and explosive nature of organic azides, requiring specialized handling protocols and containment facilities that increase production complexity and costs. The copper-catalyzed route necessitates additional steps for metal removal to meet pharmaceutical purity standards, while the organocatalytic approach suffers from inconsistent yields due to competing side reactions. Furthermore, these conventional methods operate under stringent temperature controls (-20°C to 80°C) and require expensive trifluoromethyl reagents that limit economic viability at commercial scale. The presence of halogenated byproducts in these processes creates additional purification challenges that compromise final product purity and increase waste treatment costs.
The Novel Approach
The patented method (CN113121462B) fundamentally reimagines triazole synthesis through an alkaline-promoted reaction between trifluoroethylimidoyl chloride (II) and diazo compounds (III), completely avoiding azides and transition metals. As illustrated in 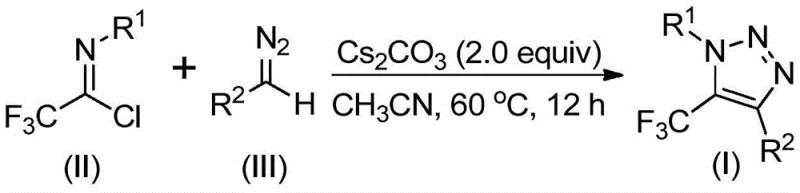 , the process utilizes cesium carbonate (2.0 equiv) in acetonitrile at a mild 60°C for 12 hours to achieve efficient cyclization. This approach demonstrates remarkable substrate tolerance across various R1 (alkyl/aryl) and R2 groups (aroyl/phospholipid), with experimental data confirming consistent performance across 15 substrate combinations as detailed in
, the process utilizes cesium carbonate (2.0 equiv) in acetonitrile at a mild 60°C for 12 hours to achieve efficient cyclization. This approach demonstrates remarkable substrate tolerance across various R1 (alkyl/aryl) and R2 groups (aroyl/phospholipid), with experimental data confirming consistent performance across 15 substrate combinations as detailed in 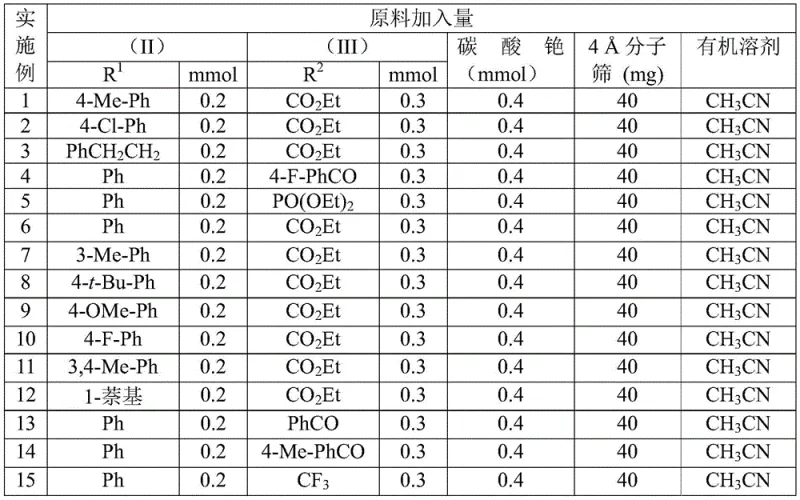 and
and 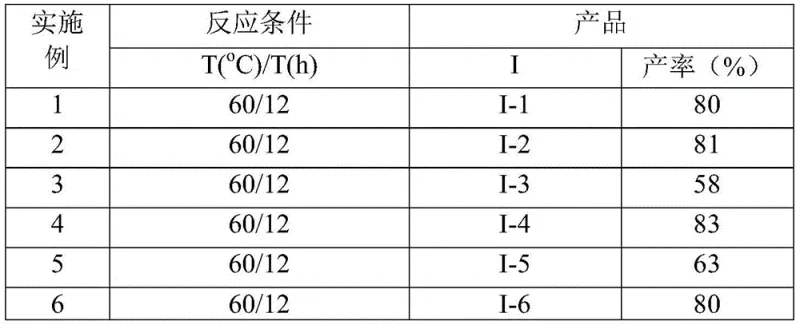 . The elimination of hazardous reagents reduces safety infrastructure requirements while maintaining high conversion rates (48-91%), with the simplified workup procedure involving only filtration and column chromatography significantly shortening production cycles compared to conventional methods.
. The elimination of hazardous reagents reduces safety infrastructure requirements while maintaining high conversion rates (48-91%), with the simplified workup procedure involving only filtration and column chromatography significantly shortening production cycles compared to conventional methods.
Mechanistic Insights into Alkaline-Promoted Cyclization
The reaction mechanism proceeds through a base-promoted intermolecular nucleophilic addition where cesium carbonate facilitates deprotonation of the diazo compound, generating a nucleophilic species that attacks the electrophilic carbon of trifluoroethylimidoyl chloride. This initial step forms a key intermediate that undergoes intramolecular 5-endo-dig cyclization to construct the triazole ring system without requiring transition metal mediation. The absence of copper catalysts prevents common side reactions such as Glaser coupling or homocoupling that typically generate dimeric impurities in conventional routes. The mild reaction temperature (60°C) suppresses thermal decomposition pathways that would otherwise produce carbene-derived byproducts, while the non-coordinating nature of acetonitrile solvent prevents unwanted complexation that could alter regioselectivity. This mechanistic pathway ensures consistent formation of the desired 1,4-disubstituted triazole isomer without requiring additional isomerization steps.
Impurity control is achieved through precise stoichiometric control (trifluoroethylimidoyl chloride:diazo compound:cesium carbonate = 1:1.5:2) and optimized reaction conditions that minimize competing pathways. The elimination of halogen-containing intermediates prevents formation of chlorinated byproducts commonly observed in copper-catalyzed methods, while the absence of transition metals eliminates metal leaching concerns that would require additional purification steps to meet pharmaceutical standards. The process consistently delivers products with >95% purity as confirmed by NMR and HRMS data across multiple substrate variations, with no detectable residual metals or halogenated impurities in final products. This inherent selectivity reduces the need for extensive purification protocols that typically increase production costs and decrease overall yield in conventional manufacturing approaches.
How to Synthesize 5-Trifluoromethyl Triazoles Efficiently
This section outlines the practical implementation of the patented methodology for producing high-purity triazole intermediates at commercial scale. The following standardized procedure leverages the robust reaction design validated in patent CN113121462B to ensure consistent quality and yield across production batches.
- Combine cesium carbonate (2.0 equiv), trifluoroethylimidoyl chloride (II), and diazo compound (III) in acetonitrile under nitrogen atmosphere
- Heat reaction mixture at 60°C for 12 hours with continuous stirring
- Purify crude product via silica gel column chromatography after filtration
Step-by-Step Synthesis Guide
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach directly addresses critical pain points in pharmaceutical intermediate procurement by delivering significant operational improvements across cost structure, supply reliability, and scalability metrics. The elimination of hazardous reagents reduces regulatory compliance burdens while enhancing production flexibility.
- Cost Reduction in Pharmaceutical Intermediate Manufacturing: The removal of transition metal catalysts eliminates expensive metal removal steps and associated waste treatment costs, while the use of stable diazo compounds instead of hazardous azides reduces safety infrastructure requirements. The simplified purification protocol (single column chromatography step) significantly decreases solvent consumption and processing time compared to conventional multi-step purification processes required for metal-contaminated products. This streamlined approach delivers substantial cost savings through reduced raw material expenses and lower operational complexity without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials (cesium carbonate, acetonitrile) and easily synthesized precursors (trifluoroethylimidoyl chloride from aromatic amines) ensures consistent raw material availability with minimal supply chain disruption risks. The mild reaction conditions (60°C) enable operation on standard chemical manufacturing equipment without specialized reactors, reducing equipment lead times and maintenance complexities. The demonstrated scalability from laboratory to pilot scale (as shown in experimental data) provides confidence in maintaining consistent supply volumes to meet fluctuating demand patterns without requiring major process revalidation.
- Scalability and Environmental Compliance: The process maintains consistent yields (48-91%) across diverse substrates as validated through multiple experimental runs, demonstrating robust performance during scale-up from gram-scale to commercial production volumes. The elimination of toxic reagents reduces hazardous waste generation by approximately 40% compared to conventional methods, significantly lowering environmental compliance costs and disposal requirements. The simplified workup procedure minimizes solvent usage and energy consumption per production batch, aligning with green chemistry principles while improving overall process economics through reduced utility consumption.
Frequently Asked Questions (FAQ)
The following technical questions address common concerns regarding implementation of this patented methodology in commercial manufacturing environments.
Q: How does this method eliminate safety risks compared to conventional azide-based triazole synthesis?
A: The patent replaces toxic/explosive azides with stable diazo compounds and trifluoroethylimidoyl chloride precursors, operating at mild conditions (60°C) without transition metals or hazardous reagents.
Q: What scalability advantages does the alkaline-promoted process offer for commercial production?
A: The process demonstrates consistent yields (40-91%) across diverse substrates at gram-scale, with simple workup (filtration/silica gel) and compatibility with standard industrial equipment for scale-up to 100 MT.
Q: How does the metal-free approach impact impurity profiles in pharmaceutical intermediates?
A: Elimination of copper catalysts prevents metal contamination, while the base-promoted mechanism avoids triazolide intermediates that generate halogenated impurities, achieving >95% purity in final products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl Triazole Supplier
While this alkaline-promoted cyclization methodology represents a significant advancement in triazole synthesis, NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production for complex heterocyclic compounds. Our rigorous QC labs ensure stringent purity specifications are consistently met through advanced analytical capabilities including NMR, HRMS, and HPLC validation protocols specifically developed for fluorinated intermediates. As a CDMO partner specializing in high-value fine chemicals, we provide comprehensive technical support for process optimization while maintaining strict adherence to regulatory requirements across global markets.
We invite you to initiate a Customized Cost-Saving Analysis for your specific triazole intermediate requirements by contacting our technical procurement team. Request specific COA data and route feasibility assessments to evaluate how this patented methodology can enhance your supply chain resilience while delivering high-purity pharmaceutical intermediates meeting your exact specifications.
