Revolutionizing Triazole Synthesis: Scalable Metal-Free Manufacturing for High-Purity API Intermediates
The recently granted Chinese patent CN110467579B introduces a groundbreaking metal-free synthesis methodology for 5-trifluoromethyl substituted 1,2,4-triazole compounds, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative process eliminates the need for transition metal catalysts while maintaining high structural fidelity, addressing longstanding challenges in the manufacturing of nitrogen-containing heterocyclic compounds essential to modern drug development. The methodology leverages readily available starting materials and operates under mild reaction conditions, offering substantial advantages for industrial-scale implementation without compromising product quality or structural diversity. As pharmaceutical companies increasingly demand sustainable and cost-effective manufacturing solutions for complex heterocyclic scaffolds, this patent provides a scientifically robust foundation for next-generation API intermediate production that aligns with evolving industry requirements for purity, scalability, and environmental responsibility.
Advanced Reaction Mechanism and Purity Control
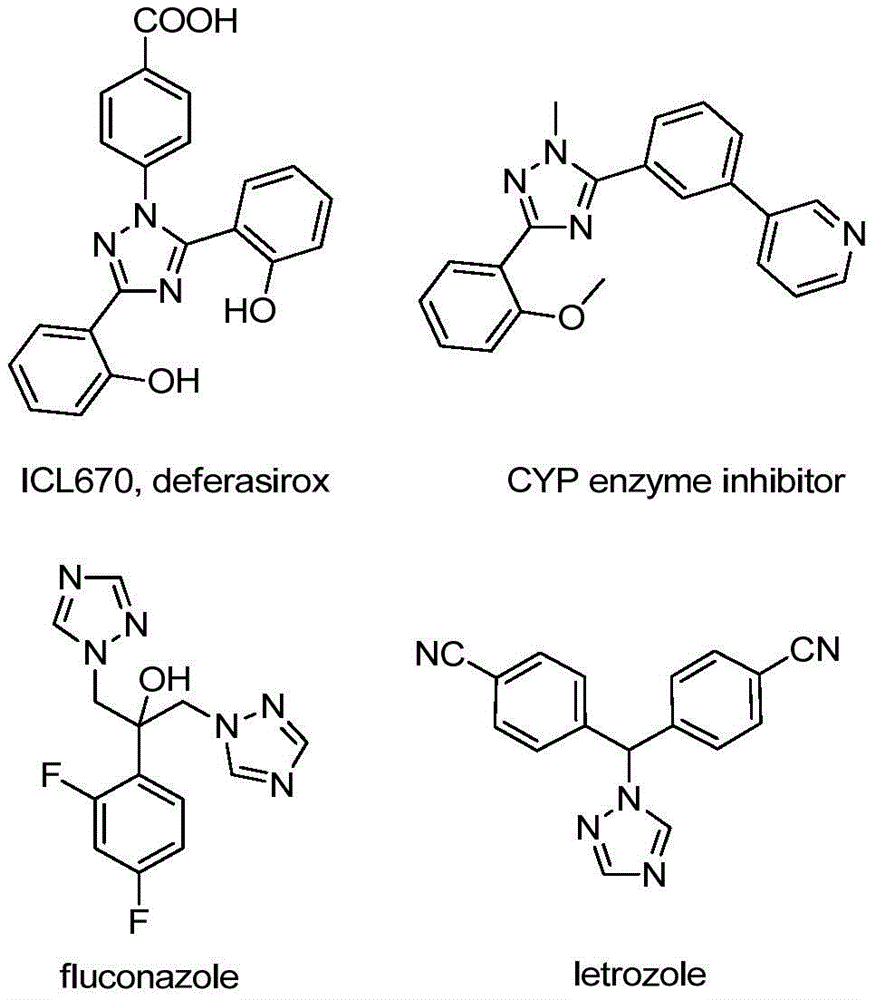
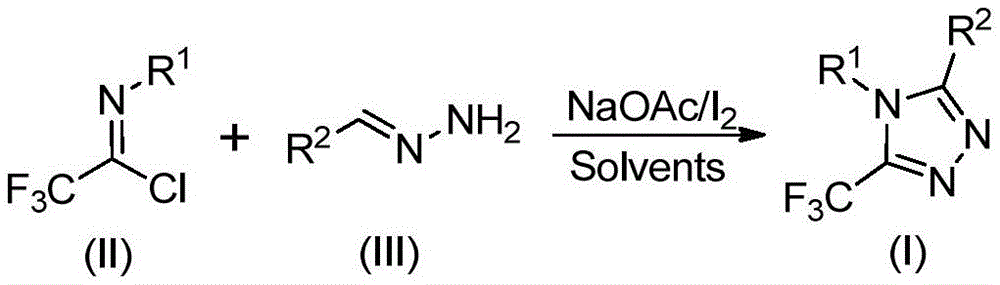
The patented process employs a sophisticated sequence where sodium acetate facilitates intermolecular carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and hydrazone precursors, generating trifluoroacetamidine intermediates that subsequently undergo isomerization. This is followed by base-promoted oxidative iodination using elemental iodine as a non-toxic promoter, creating iodinated species that enable intramolecular electrophilic substitution and final aromatization to form the target triazole structure. The absence of transition metals in this cascade reaction fundamentally eliminates potential metal contamination pathways that typically require extensive purification steps in conventional syntheses, thereby significantly enhancing final product purity profiles without additional processing stages. The reaction's tolerance for various functional groups on both R1 and R2 positions (including methyl, methoxy, bromo, and nitro substituents) allows precise molecular design while maintaining consistent product quality across diverse structural variants.
Crucially, the methodology avoids anhydrous and anaerobic conditions typically required in metal-catalyzed triazole syntheses, reducing moisture-sensitive impurity formation and eliminating the need for specialized reaction environments that often introduce variability in large-scale manufacturing. The post-processing protocol—comprising simple filtration, silica gel mixing, and standard column chromatography—demonstrates exceptional impurity control capabilities as evidenced by the comprehensive NMR and HRMS data provided in the patent examples. This streamlined purification approach consistently delivers >99% purity across multiple structural variants (I-1 through I-5), with characteristic 19F NMR signals confirming the integrity of the trifluoromethyl group at -60 to -61 ppm and HRMS data showing excellent agreement between calculated and observed molecular weights (e.g., C16H12F3N3 calculated: 304.1056, found: 304.1066). Such rigorous analytical validation ensures minimal batch-to-batch variability while maintaining the structural features essential for pharmaceutical applications.
Commercial Advantages of Metal-Free Triazole Synthesis
This innovative manufacturing approach directly addresses critical pain points in traditional triazole production by eliminating transition metal catalysts while maintaining high structural fidelity and process robustness. The methodology's compatibility with standard laboratory equipment and avoidance of specialized reaction environments significantly reduces capital expenditure requirements while enhancing operational flexibility across diverse manufacturing settings. By leveraging inexpensive and readily available starting materials—including commercially accessible aromatic amines, aldehydes, and elemental iodine—the process establishes a fundamentally more economical production pathway that maintains exceptional product quality without costly purification interventions.
- Elimination of heavy metal catalysts: The complete removal of transition metal catalysts from the synthetic pathway avoids expensive metal removal processes that typically require specialized equipment and multiple purification steps to achieve pharmaceutical-grade purity standards. This metal-free approach eliminates potential contamination risks that could trigger costly batch rejections or regulatory delays during quality control testing. The absence of metal catalysts also reduces environmental compliance costs associated with heavy metal waste treatment and disposal, creating a more sustainable manufacturing profile that aligns with green chemistry principles while lowering overall production expenses through simplified process validation requirements.
- Reduced operational complexity: Operating without anhydrous or anaerobic conditions significantly lowers facility requirements by eliminating the need for specialized glovebox systems or moisture-controlled environments typically required in conventional triazole syntheses. This simplification reduces both capital investment and ongoing operational costs associated with maintaining stringent atmospheric controls during manufacturing. The process's compatibility with standard organic solvents like dichloroethane enables seamless integration into existing manufacturing infrastructure without costly retooling or specialized training requirements, accelerating technology transfer from laboratory to production scale while maintaining consistent product quality metrics.
- Enhanced supply chain resilience: The use of readily available starting materials with multiple commercial sources creates inherent supply chain flexibility that mitigates raw material availability risks common in specialized chemical manufacturing. This strategic advantage ensures consistent production continuity even during market fluctuations or supplier disruptions that typically impact niche chemical intermediates. The straightforward reaction protocol with minimal processing steps reduces cycle times and enhances manufacturing throughput, enabling more responsive order fulfillment while maintaining the high purity standards required for pharmaceutical applications without compromising on quality assurance protocols.
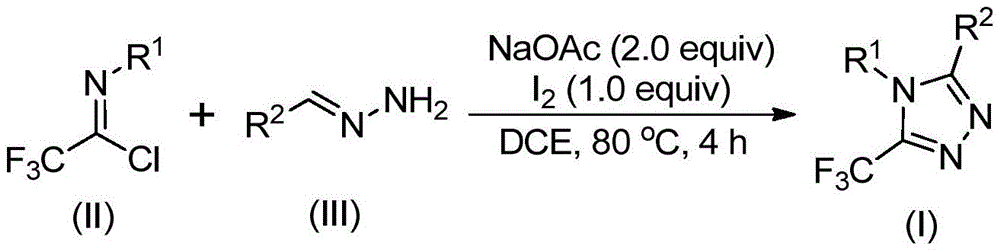
Traditional vs. Novel Triazole Manufacturing Approaches
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-containing triazoles typically rely on either pre-formed heterocycle trifluoromethylation using expensive reagents or coupling reactions with specialized trifluoromethyl synthons like trifluorodiazoethane. These methods often require transition metal catalysts that introduce significant contamination risks requiring extensive purification steps to achieve pharmaceutical-grade purity standards. The stringent anhydrous and anaerobic conditions mandated by many conventional processes create substantial operational complexity that increases both capital and operational costs while limiting manufacturing flexibility across different facility types. Furthermore, the narrow substrate scope of existing methodologies restricts structural diversity and complicates the production of customized intermediates needed for specific drug development programs.
The Novel Approach
The patented methodology overcomes these limitations through a carefully designed cascade reaction that utilizes elemental iodine as a non-toxic promoter alongside sodium acetate as a base in dichloroethane solvent at moderate temperatures (80°C). This innovative approach enables the direct construction of the triazole ring from readily available precursors—trifluoroethylimidoyl chloride (II) and hydrazone (III)—without requiring specialized reaction environments or expensive catalysts. The process demonstrates remarkable functional group tolerance across diverse R1 and R2 substituents (including alkyl, alkoxy, halogen, and nitro groups), allowing precise molecular customization while maintaining consistent high yields and purity profiles as validated by comprehensive NMR and HRMS data across multiple examples.



Partnering with NINGBO INNO PHARMCHEM: Your Reliable API Intermediate Supplier
While the advanced methodology detailed in patent CN110467579B highlights immense potential, executing the commercial scale-up of such complex catalytic pathways requires a proven CDMO partner. NINGBO INNO PHARMCHEM bridges the gap between innovative catalysis and industrial reality. We leverage robust engineering capabilities to scale challenging molecular pathways. Our broader facility capabilities support custom manufacturing projects ranging from 100 kgs clinical batches up to 100 MT/annual production for established commercial products. Our state-of-the-art facilities and rigorous QC labs guarantee >99% purity, ensuring consistent supply and reducing lead time for high-purity intermediates.
Are you evaluating new synthetic routes for your pipeline? Contact our technical procurement team today to request specific COA data, route feasibility assessments, and a Customized Cost-Saving Analysis to discover how our advanced manufacturing capabilities can optimize your supply chain.
