Advanced One-Pot Synthesis of N-Acyl Indoles: A Breakthrough for Scalable Pharmaceutical Intermediate Production
Advanced One-Pot Synthesis of N-Acyl Indoles: A Breakthrough for Scalable Pharmaceutical Intermediate Production
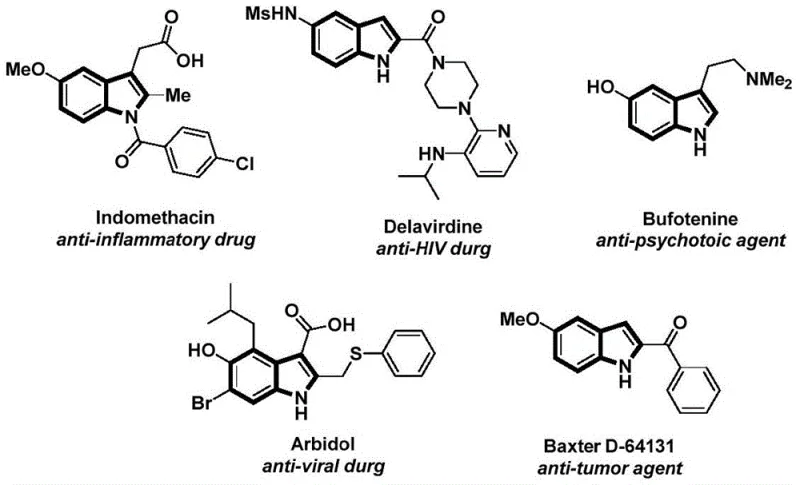
The structural motif of the indole ring is ubiquitous in medicinal chemistry, serving as the core scaffold for a vast array of bioactive molecules ranging from anti-inflammatory agents like Indomethacin to potent anti-HIV drugs such as Delavirdine. As illustrated in the diverse pharmacological profiles of these compounds, the ability to efficiently construct functionalized indole derivatives is of paramount importance to the global pharmaceutical industry. Recently, Chinese patent CN112898192B disclosed a novel preparation method for N-acyl indole compounds that addresses several critical bottlenecks in traditional synthetic routes. This technology leverages a palladium-catalyzed carbonylation cyclization strategy, utilizing readily available 2-alkynyl anilines and aryl iodides as starting materials. By replacing toxic carbon monoxide gas with a solid surrogate, this innovation not only enhances laboratory safety but also streamlines the manufacturing process, making it an attractive candidate for reliable pharmaceutical intermediates supplier networks seeking robust and scalable technologies.
Traditional methods for synthesizing N-acyl indoles often rely on harsh reaction conditions or multi-step sequences that suffer from poor atom economy and significant waste generation. Conventional carbonylation reactions typically require the use of pressurized carbon monoxide gas, which poses severe safety hazards and necessitates specialized high-pressure reactors that are expensive to maintain and operate. Furthermore, many existing protocols exhibit limited substrate compatibility, failing to tolerate sensitive functional groups that are frequently present in complex drug candidates. These limitations result in prolonged development timelines and increased costs, creating a pressing need for cost reduction in API manufacturing. The novel approach described in the patent circumvents these issues by employing a mild, one-pot procedure that operates at atmospheric pressure using standard glassware or stainless steel reactors, thereby drastically lowering the barrier to entry for commercial production.
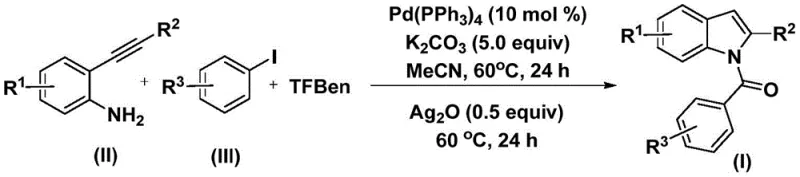
The mechanistic pathway of this transformation involves a sophisticated interplay of organometallic steps initiated by the oxidative addition of the aryl iodide to the palladium(0) catalyst. This generates an aryl-palladium intermediate which subsequently undergoes migratory insertion of carbon monoxide. Crucially, the carbon monoxide is generated in situ from the decomposition of 1,3,5-tricarboxylic acid phenol ester (TFBen), ensuring a steady and controlled release of the carbonyl source without the risks associated with gas cylinders. The resulting acyl-palladium species then reacts with the 2-alkynyl aniline to form an amide intermediate. In the second stage of the reaction, the addition of silver oxide promotes an intramolecular cyclization, effectively closing the indole ring to yield the final N-acyl product. This dual-stage mechanism allows for precise control over the reaction trajectory, minimizing the formation of side products and ensuring high-purity N-acyl indole derivatives suitable for downstream applications.
How to Synthesize N-Acyl Indole Compounds Efficiently
The operational simplicity of this protocol makes it highly accessible for process chemists aiming to implement this technology. The procedure begins with the combination of the palladium catalyst, base, solid CO source, and substrates in an organic solvent such as acetonitrile. The detailed standardized synthesis steps see the guide below, which outlines the precise stoichiometry and thermal profiles required to achieve optimal conversion rates. By adhering to these parameters, manufacturers can consistently reproduce the high yields reported in the patent examples, ensuring a stable supply of critical intermediates.
- Combine palladium catalyst (Pd(PPh3)4), potassium carbonate, solid CO source (TFBen), 2-alkynyl aniline, and aryl iodide in acetonitrile solvent.
- Heat the reaction mixture at 60°C for 24 hours to facilitate the initial carbonylation and amide formation.
- Add silver oxide (Ag2O) to the mixture and continue heating at 60°C for another 24 hours to induce cyclization, followed by filtration and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from gaseous reagents to solid surrogates represents a paradigm shift in supply chain risk management. The reliance on TFBen, a stable and storable solid, eliminates the logistical complexities and regulatory burdens associated with transporting and storing compressed carbon monoxide. This transition significantly enhances supply chain reliability by decoupling production schedules from the availability of hazardous gas supplies, which can be subject to strict transportation regulations and delivery delays. Moreover, the use of commercially available starting materials like aryl iodides and 2-alkynyl anilines ensures that raw material sourcing remains straightforward and cost-effective, reducing the lead time for high-purity heterocyclic scaffolds.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the associated safety infrastructure leads to substantial capital expenditure savings. Additionally, the use of a solid CO source allows for more efficient reagent utilization, reducing waste disposal costs. The mild reaction temperature of 60°C further contributes to energy savings compared to processes requiring reflux or cryogenic conditions, driving down the overall operational expenditure per kilogram of product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that production is less susceptible to interruptions caused by equipment failure or environmental fluctuations. The broad substrate scope allows for the synthesis of a wide variety of analogues using the same core process, providing flexibility to adapt to changing market demands without requalifying entirely new synthetic routes. This versatility is crucial for maintaining continuity of supply in the fast-paced pharmaceutical sector.
- Scalability and Environmental Compliance: The one-pot nature of the synthesis minimizes solvent usage and intermediate isolation steps, aligning with green chemistry principles. The simplified workup procedure, involving filtration and chromatography, reduces the volume of chemical waste generated. This environmental friendliness facilitates easier regulatory approval and permits for commercial scale-up of complex heterocyclic scaffolds, ensuring long-term sustainability of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this patented technology. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this method for their specific pipeline projects. The answers are derived directly from the experimental data and technical disclosures within the patent documentation.
Q: What is the advantage of using TFBen over carbon monoxide gas?
A: Using 1,3,5-tricarboxylic acid phenol ester (TFBen) as a solid carbon monoxide substitute eliminates the need for handling hazardous high-pressure CO gas, significantly improving operational safety and simplifying equipment requirements for industrial scale-up.
Q: What is the substrate scope of this N-acyl indole synthesis method?
A: The method demonstrates excellent functional group tolerance, accommodating various substituents such as methyl, methoxy, halogens (F, Cl, Br), and trifluoromethyl groups on both the aniline and aryl iodide components, yielding products with moderate to high efficiency.
Q: Is the palladium catalyst reusable in this process?
A: While the patent specifies the use of tetrakis(triphenylphosphine)palladium(0) at 10 mol%, the process is designed for high efficiency in a single batch. For large-scale commercial production, catalyst recovery systems can be implemented to further reduce costs, though the primary economic benefit comes from the mild reaction conditions and cheap reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acyl Indole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN112898192B for accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of N-acyl indole intermediates meets the exacting standards required by global regulatory bodies.
We invite potential partners to engage with our technical procurement team to discuss how this technology can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this safer, more efficient synthetic route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your research timeline and your bottom line.
