Scalable Manufacturing of Gefitinib Intermediates via Convergent Synthesis Routes
Scalable Manufacturing of Gefitinib Intermediates via Convergent Synthesis Routes
The pharmaceutical industry constantly seeks robust manufacturing pathways for critical oncology therapeutics, and the tyrosine kinase inhibitor Gefitinib (ZD1839) remains a cornerstone in this domain. Patent CN102030722A discloses a transformative chemical process for the preparation of 4-(3'-chloro-4'-fluoroanilino)-7-methoxy-6-(3-morpholinopropoxy)quinazoline, widely known by its brand name Iressa. This document details a sophisticated convergent synthesis strategy that addresses the inherent inefficiencies of earlier linear routes, specifically targeting the production of high-purity intermediates essential for API formulation. The core innovation lies in the streamlined construction of the quinazoline core, bypassing traditional bottlenecks associated with purification and yield loss. By focusing on the synthesis of key intermediates such as 7-methoxy-6-(3-morpholinopropoxy)-3,4-dihydroquinazolin-4-one (Formula II), this technology offers a viable pathway for commercial scale-up.
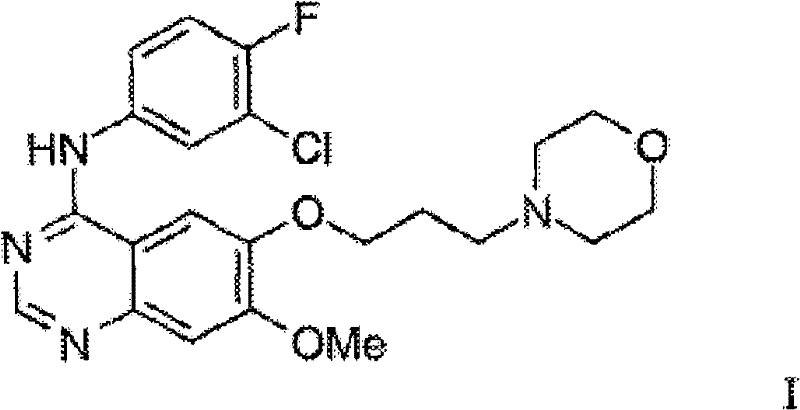
For R&D directors and process chemists, the structural integrity and purity profile of the final active pharmaceutical ingredient are paramount. The molecule depicted above represents the final target, characterized by a substituted quinazoline ring linked to a morpholine-containing side chain and a halogenated aniline moiety. Achieving this structure with minimal impurities requires precise control over each synthetic transformation. The patent emphasizes that previous methods, such as those disclosed in WO 96/33980, relied heavily on linear sequences that necessitated the isolation of multiple intermediates and extensive chromatographic purification. In contrast, the methodology presented in CN102030722A utilizes a convergent approach where complex side chains are introduced earlier in the sequence, allowing for a more efficient assembly of the final heterocyclic system. This shift not only improves the overall mass balance but also simplifies the impurity profile, making downstream processing significantly more manageable for quality control teams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Gefitinib and its precursors has been plagued by operational inefficiencies that hinder cost-effective manufacturing. The prior art routes typically involved building the 3-morpholinopropoxy side chain at the very end of the synthesis, after the quinazoline ring had already been formed. This linear strategy meant that every step leading up to the final coupling had to be performed on a fully functionalized, often sensitive, intermediate. Consequently, these methods required repeated isolation and purification steps, frequently relying on column chromatography to remove trace impurities that accumulated throughout the long sequence. For a procurement manager, this translates to higher consumption of silica gel, solvents, and labor, driving up the cost of goods sold (COGS). Furthermore, the low overall yields associated with linear synthesis result in significant material wastage, which is economically unsustainable for large-volume API production. The reliance on chromatographic separation also introduces variability in batch quality and extends the manufacturing cycle time, creating bottlenecks in the supply chain.
The Novel Approach
The innovative process described in the patent fundamentally restructures the synthetic logic by adopting a convergent strategy that builds the side chain onto a simpler aromatic precursor before ring closure. This approach allows for the preparation of the key intermediate, 4-methoxy-5-(3-morpholinopropoxy)-2-nitrobenzonitrile (Formula III), which serves as a versatile building block. By introducing the morpholinepropoxy group early, the subsequent transformations—reduction, hydration, and cyclization—can be telescoped effectively. This means that intermediates like the amino benzonitrile (Formula IV) and the benzamide (Formula V) do not necessarily need to be isolated as solid products but can be processed in solution. This telescoping capability drastically reduces the number of unit operations, eliminating the need for time-consuming filtration and drying steps between reactions. For supply chain heads, this reduction in processing steps directly correlates to shorter lead times and increased throughput capacity, enabling manufacturers to respond more agilely to market demand without compromising on the stringent purity specifications required for oncology drugs.
Mechanistic Insights into the Convergent Synthesis of Quinazolinone Intermediates
The heart of this technological advancement lies in the efficient conversion of the nitro-benzonitrile precursor into the quinazolinone core. The process begins with the preparation of Formula III, which involves the alkylation of 3-hydroxy-4-methoxybenzonitrile with a morpholine derivative. As illustrated in the reaction scheme below, this step establishes the ether linkage that will eventually become the solubilizing side chain of the final drug. The use of a displaceable group Z, such as a halogen or sulfonate, on the propyl chain facilitates this nucleophilic substitution under basic conditions. Following this, the nitro group is selectively reduced to an amine. The patent highlights the versatility of this reduction, noting that it can be achieved via catalytic hydrogenation using palladium on carbon or through chemical reduction using activated metals like iron or reagents such as V-Brite B. This flexibility allows manufacturers to choose the most cost-effective and safe reduction method available at their facility, ensuring supply chain resilience against catalyst shortages.
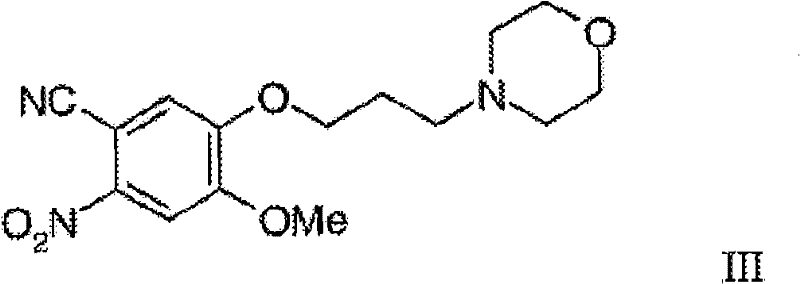
Following the reduction to Formula IV, the nitrile group undergoes hydration to form the corresponding benzamide (Formula V). This transformation is critical as it sets up the functionality required for the subsequent ring closure. The patent specifies that this hydration can be carried out under alkaline conditions, preferably using potassium hydroxide in a solvent like tertiary amyl alcohol. The choice of solvent is crucial here, as it must withstand the basic conditions and elevated temperatures without degrading the sensitive amine functionality. Once the benzamide is formed, the final cyclization to the quinazolinone (Formula II) is effected using formic acid or its reactive derivatives, such as formamide. This cyclodehydration step closes the heterocyclic ring, generating the core scaffold of the drug. The ability to perform these three distinct chemical transformations—reduction, hydration, and cyclization—in a telescoped manner without isolating the intermediate solids is a major mechanistic advantage. It minimizes exposure of the reactive amine to air and moisture, thereby reducing the formation of oxidation byproducts and ensuring a cleaner reaction profile for the final API.
How to Synthesize 7-methoxy-6-(3-morpholinopropoxy)-3,4-dihydroquinazolin-4-one Efficiently
The synthesis of the key quinazolinone intermediate is a multi-step sequence that benefits greatly from process optimization and telescoping. To achieve high yields and purity, operators must carefully control reaction parameters such as temperature, pH, and solvent composition during the reduction and hydration phases. The patent provides detailed guidance on using specific reagents like V-Brite B for reduction and potassium hydroxide for hydration, emphasizing the importance of maintaining specific temperature ranges to prevent side reactions. For a comprehensive understanding of the operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Reduce the nitro group of Formula III using V-Brite B or catalytic hydrogenation to obtain the amino benzonitrile (Formula IV).
- Perform hydration of the nitrile group in Formula IV using potassium hydroxide in tertiary amyl alcohol to generate the benzamide (Formula V).
- Cyclize Formula V with formic acid or formamide at elevated temperatures to yield the final quinazolinone intermediate (Formula II).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this convergent synthesis route offers substantial benefits that extend beyond mere technical feasibility. For procurement managers tasked with optimizing the cost structure of API manufacturing, the elimination of chromatographic purification is a game-changer. Traditional methods that rely on column chromatography consume vast quantities of silica gel and organic solvents, both of which represent significant variable costs and waste disposal liabilities. By replacing these purification steps with crystallization and extraction protocols, the new process drastically reduces the consumption of consumables. Furthermore, the telescoped nature of the synthesis means fewer isolation steps, which translates to lower labor costs and reduced equipment occupancy time. This efficiency gain allows for a more competitive pricing structure for the final intermediate, providing a buffer against raw material price fluctuations in the global chemical market.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this process is the removal of chromatographic purification and the reduction of isolation steps. In conventional linear synthesis, each intermediate isolation incurs costs related to filtration, drying, and analysis, alongside the inevitable yield loss associated with physical handling. By processing intermediates like Formula IV and V in solution, the manufacturer avoids these losses entirely. Additionally, the use of commodity chemicals such as formic acid, potassium hydroxide, and common solvents like toluene and ethyl acetate ensures that the raw material basket remains affordable and stable. The qualitative improvement in yield, driven by the avoidance of degradation during isolation, further amplifies the cost efficiency, ensuring that more of the starting material ends up in the final product rather than in the waste stream.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that have multiple points of failure. The simplified workflow of this novel method reduces the number of critical control points, thereby enhancing overall process robustness. Since fewer intermediates need to be stored and transported between different production suites or even different sites, the logistical burden is significantly lightened. The ability to telescope reactions also means that the total cycle time from raw material to finished intermediate is compressed. This agility allows manufacturers to react more swiftly to changes in demand forecasts, reducing the risk of stockouts. Moreover, the flexibility in choosing reduction methods (hydrogenation vs. chemical reduction) provides a contingency plan; if hydrogen gas or specific catalysts are unavailable, the process can still proceed using alternative reductants, ensuring uninterrupted production schedules.
- Scalability and Environmental Compliance: Scaling a chemical process from the laboratory to multi-ton production often reveals hidden challenges related to heat transfer and mixing, particularly in exothermic reactions like nitration or reduction. The conditions described in the patent, such as performing reductions at moderate temperatures (e.g., 50-70°C) and using standard solvents, are inherently scalable. The absence of chromatography also simplifies the environmental footprint of the manufacturing process. Chromatographic waste is difficult to treat and recycle, whereas the aqueous and organic waste streams generated by extraction and crystallization are more amenable to standard wastewater treatment protocols. This alignment with green chemistry principles not only reduces disposal costs but also ensures compliance with increasingly stringent environmental regulations in major pharmaceutical manufacturing hubs, safeguarding the long-term viability of the supply source.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer.
Q: How does this process improve purity compared to conventional linear synthesis?
A: The novel convergent route eliminates the need for chromatographic purification steps required in older linear methods. By utilizing telescoped reactions and specific crystallization techniques, such as DMSO solvate formation, the process achieves high-purity specifications suitable for API manufacturing without expensive resin-based cleaning.
Q: What are the key advantages for supply chain stability?
A: This method significantly reduces the number of isolated intermediates. By processing compounds like Formula IV and V in solution without isolation, the overall cycle time is shortened, and material loss is minimized. This leads to enhanced supply chain reliability and consistent batch-to-batch quality for large-scale production.
Q: Is the synthesis scalable for commercial API production?
A: Yes, the patent explicitly describes industrial acceptable yields and conditions suitable for scale-up. The use of common solvents like toluene, ethyl acetate, and alcohols, along with standard reagents like phosphorus oxychloride and formic acid, ensures the process is robust and adaptable for multi-ton manufacturing campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gefitinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team specializes in the scale-up of complex pharmaceutical intermediates, including the quinazoline derivatives discussed herein. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of intermediate meets the high standards required for oncology API manufacturing. We are committed to delivering high-purity Gefitinib intermediates that facilitate seamless downstream processing for our global clients.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to request specific COA data and route feasibility assessments to validate the technical and economic benefits of this convergent approach. Let us collaborate to enhance the efficiency and reliability of your pharmaceutical manufacturing operations.
