Advanced Resolution of Axially Chiral Oxindole Styrenes for High-Purity Pharmaceutical Intermediates
Advanced Resolution of Axially Chiral Oxindole Styrenes for High-Purity Pharmaceutical Intermediates
The landscape of asymmetric synthesis is continually evolving, driven by the urgent need for efficient access to structurally complex chiral scaffolds that serve as critical building blocks in modern drug discovery. Patent CN111848322B introduces a groundbreaking methodology for the resolution of axially chiral oxindole-substituted styrene compounds, addressing a significant gap in the current literature regarding the synthesis of these valuable motifs. Traditionally, the construction of axially chiral styrene skeletons has relied heavily on limited strategies such as nucleophilic additions to alkynes or transition-metal catalyzed couplings, which often suffer from narrow substrate scope and rigorous reaction requirements. This innovation leverages a chiral phosphoric acid catalyzed kinetic resolution strategy, utilizing readily available racemic precursors to generate high-value chiral products with exceptional stereocontrol. By establishing a robust protocol that operates under mild conditions with high atom economy, this technology offers a compelling solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring that research teams can access diverse chiral libraries without the bottlenecks associated with traditional de novo asymmetric synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this development, the synthetic community faced substantial hurdles in accessing axially chiral styrene derivatives, as existing methodologies were often restricted to specific substrate classes or required expensive and air-sensitive transition metal catalysts. For instance, earlier reports described the use of palladium or titanium complexes to forge these chiral axes, processes that inherently introduce the risk of heavy metal contamination—a critical concern for high-purity pharmaceutical intermediate manufacturing where strict regulatory limits apply. Furthermore, these conventional routes frequently demanded harsh reaction conditions, including elevated temperatures or strong bases, which could compromise the integrity of sensitive functional groups present on the molecular scaffold. The lack of a general, mild, and metal-free resolution method meant that many potential drug candidates containing these motifs remained inaccessible or prohibitively expensive to produce, limiting the exploration of their biological potential and creating supply chain vulnerabilities for downstream applications in medicinal chemistry.
The Novel Approach
The methodology disclosed in CN111848322B revolutionizes this space by employing an organocatalytic resolution strategy that bypasses the need for transition metals entirely, utilizing a chiral phosphoric acid derivative to differentiate between enantiomers of the racemic starting material. This approach allows for the direct conversion of simple, commercially accessible precursors into highly enantioenriched axially chiral oxindole-substituted styrenes through a highly selective addition reaction. The process is characterized by its operational simplicity, proceeding in dichloromethane at low temperatures (0°C to 10°C) with the aid of molecular sieves to maintain anhydrous conditions, thereby significantly reducing lead time for high-purity pharmaceutical intermediates by minimizing complex workup procedures. The versatility of this system is evidenced by its tolerance to a wide array of substituents on the aromatic rings, enabling the generation of a diverse library of chiral compounds that were previously difficult to synthesize, thus providing a reliable pharmaceutical intermediate supplier with a powerful tool for expanding their portfolio of chiral building blocks.
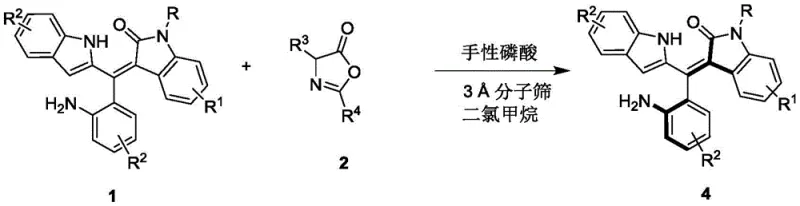
Mechanistically, the success of this transformation hinges on the precise dual-activation mode facilitated by the chiral phosphoric acid catalyst, which simultaneously activates both the electrophilic and nucleophilic partners through a well-defined hydrogen-bonding network. The catalyst, typically a binaphthyl-derived phosphoric acid substituted with bulky aromatic groups like 2-naphthyl, creates a confined chiral environment that dictates the facial selectivity of the incoming nucleophile. This supramolecular assembly ensures that the reaction proceeds through a lower energy transition state for one enantiomer over the other, effectively locking the axial chirality during the bond-forming event. Such a mechanism not only explains the high enantiomeric excess values observed (up to 96% ee) but also highlights the importance of catalyst design in achieving cost reduction in fine chemical manufacturing by maximizing the efficiency of each catalytic cycle. The ability to fine-tune the steric and electronic properties of the catalyst backbone allows for further optimization, making this a adaptable platform for various asymmetric transformations beyond the specific examples provided in the patent data.
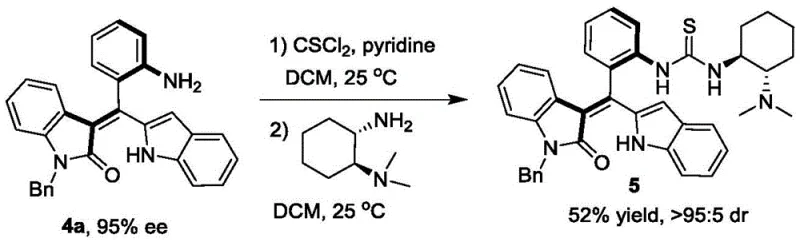
Furthermore, the utility of the resolved products extends far beyond their role as static intermediates, as they serve as privileged scaffolds for the construction of next-generation organocatalysts. The patent details a seamless derivatization pathway where the resolved axially chiral styrene (Formula 4a) is converted into a chiral tertiary amine catalyst (Formula 5) via a thiourea linkage. This downstream application underscores the strategic value of the initial resolution step, as the resulting catalyst demonstrates superior performance in promoting asymmetric [4+2] cyclization reactions compared to existing commercial alternatives. By integrating the synthesis of the chiral scaffold with its application in catalysis, the technology creates a closed-loop value proposition where the intermediate is not just a commodity but a key enabler for further chemical innovation, reinforcing the importance of securing a stable supply of these specialized fine chemical intermediates for advanced R&D programs.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
The practical implementation of this resolution technology is designed for scalability and ease of operation, making it highly attractive for process chemistry teams aiming to transition from milligram-scale discovery to kilogram-scale production. The standard protocol involves dissolving the racemic substrate and the azlactone coupling partner in dry dichloromethane, followed by the addition of activated 3A molecular sieves to scavenge trace moisture that could deactivate the catalyst. Upon cooling the mixture to 0°C, the chiral phosphoric acid catalyst is introduced, and the reaction is allowed to proceed with stirring until thin-layer chromatography indicates complete consumption of the limiting reagent. Detailed standardized synthesis steps for optimizing yield and enantioselectivity are provided in the guide below, ensuring reproducibility across different laboratory settings.
- Prepare the reaction mixture by combining the racemic starting material (Formula 1) and the azlactone derivative (Formula 2) in dichloromethane solvent with 3A molecular sieves.
- Add the chiral phosphoric acid catalyst (Formula 3, preferably 10 mol%) to the mixture and stir at a controlled low temperature between 0°C and 10°C.
- Monitor the reaction progress via TLC until completion, then filter off the molecular sieves, concentrate the filtrate, and purify the resulting axially chiral product (Formula 4) using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this organocatalytic resolution method offers distinct strategic advantages that align with the goals of cost efficiency and supply security. By eliminating the reliance on precious transition metals like palladium or platinum, manufacturers can avoid the volatile pricing and geopolitical supply risks associated with these critical raw materials, leading to a more stable and predictable cost structure for the final intermediate. Additionally, the mild reaction conditions reduce the energy footprint of the manufacturing process, as there is no need for energy-intensive heating or cryogenic cooling beyond standard refrigeration, contributing to overall operational expenditure savings. The simplicity of the workup procedure, which primarily involves filtration and standard chromatography, minimizes solvent usage and waste generation, facilitating compliance with increasingly stringent environmental regulations without compromising throughput.
- Cost Reduction in Manufacturing: The transition to an organocatalytic system fundamentally alters the cost drivers of the synthesis by removing the need for expensive metal salts and specialized ligands that often account for a significant portion of the raw material bill. Since the chiral phosphoric acid catalysts can be recovered and potentially recycled, and the starting materials are derived from abundant commodity chemicals, the overall cost of goods sold is significantly optimized. Moreover, the high selectivity of the reaction reduces the formation of diastereomeric impurities, which simplifies the purification process and increases the overall yield of the desired isomer, thereby maximizing the value extracted from every kilogram of input material.
- Enhanced Supply Chain Reliability: Securing a consistent supply of high-quality chiral intermediates is critical for maintaining uninterrupted drug development timelines, and this method enhances reliability by utilizing robust, shelf-stable reagents that are less susceptible to degradation than sensitive metal complexes. The broad substrate scope means that a single catalytic platform can be used to generate a wide variety of analogues, reducing the need to qualify multiple different synthetic routes for different projects. This flexibility allows supply chain managers to consolidate vendors and streamline inventory management, ensuring that critical materials are available on demand to support rapid iteration in medicinal chemistry campaigns.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being easily manageable at low temperatures and the absence of hazardous gaseous byproducts often associated with metal-catalyzed couplings. The use of dichloromethane, while requiring careful handling, is a well-understood solvent in industrial settings with established recovery protocols, and the solid waste generated (primarily molecular sieves and silica) is non-toxic and easy to dispose of. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is increasingly becoming a key criterion in supplier selection processes for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology, drawing directly from the experimental data and scope defined in the patent documentation. Understanding these nuances is essential for project managers evaluating the feasibility of integrating this route into their existing pipelines, as it clarifies the operational boundaries and potential customization options available. The answers provided reflect the current state of the art as described in the intellectual property, offering a realistic baseline for performance expectations.
Q: What represents the primary advantage of this resolution method over traditional transition metal catalysis?
A: Unlike traditional methods relying on palladium or titanium catalysts which often require harsh conditions and complex ligand synthesis, this organocatalytic approach utilizes chiral phosphoric acids under mild temperatures (0-10°C), eliminating heavy metal contamination risks and simplifying purification protocols for pharmaceutical grade intermediates.
Q: Can the resolved axially chiral styrenes be further functionalized into active catalysts?
A: Yes, the patent demonstrates that the resolved compound (Formula 4a) can be efficiently derivatized into a chiral tertiary amine catalyst (Formula 5) through a thiourea linkage, which subsequently exhibits superior performance in asymmetric [4+2] cyclization reactions compared to commercial benchmarks.
Q: What is the typical enantiomeric excess (ee) achievable with this protocol?
A: The methodology consistently delivers high optical purity, with experimental data showing enantiomeric excess values reaching up to 96% ee across a diverse range of substrates, ensuring the production of high-purity chiral building blocks essential for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
As the demand for sophisticated chiral building blocks continues to surge in the pharmaceutical sector, partnering with an experienced CDMO capable of navigating complex asymmetric syntheses is paramount for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory curiosity to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of axially chiral oxindole-substituted styrene meets the exacting standards required for clinical trial materials and eventual commercial API manufacturing.
We invite you to engage with our technical procurement team to discuss how this innovative resolution technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol for your specific target molecules. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering high-quality, cost-effective solutions that accelerate your drug development timeline.
