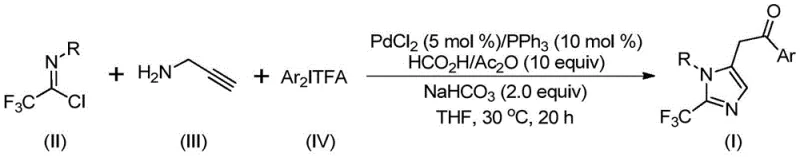
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazole Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds that possess enhanced biological profiles. Patent CN111423381A discloses a groundbreaking preparation method for 2-trifluoromethyl substituted imidazole compounds, addressing a critical need in modern medicinal chemistry. Imidazole derivatives are ubiquitous in bioactive molecules, serving as core structures in numerous drugs and functional materials. The strategic incorporation of a trifluoromethyl group into these heterocycles is known to drastically improve pharmacokinetic properties, including metabolic stability and lipophilicity. This patent presents a transition metal palladium-catalyzed carbonylation tandem reaction that efficiently synthesizes these valuable motifs using cheap and readily available starting materials. By leveraging trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts, this novel route offers a significant departure from traditional, often hazardous synthetic pathways. For R&D directors and procurement managers alike, this technology represents a viable pathway to high-purity intermediates with improved cost-efficiency and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrogen-containing heterocycles bearing trifluoromethyl functional groups has relied heavily on the use of specific trifluoromethyl synthons reacting directly with suitable substrates. Literature reports frequently cite the use of trifluorodiazoethane as a primary reagent for these transformations. However, diazo compounds are inherently unstable and potentially explosive, posing significant safety risks during handling and storage, particularly on a commercial scale. Furthermore, alternative methods utilizing trifluoroethylimide acid halides have not been widely explored or optimized, leaving a gap in practical, safe, and scalable synthetic protocols. Conventional approaches often suffer from harsh reaction conditions, limited substrate scope, and the generation of difficult-to-remove impurities, which complicates downstream purification and increases the overall cost of goods. These limitations create bottlenecks for reliable pharmaceutical intermediate suppliers aiming to deliver consistent quality at scale.
The Novel Approach
The methodology described in the patent overcomes these historical barriers by employing a sophisticated palladium-catalyzed carbonylation series reaction. This approach utilizes trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts as the foundational building blocks. The reaction proceeds under remarkably mild conditions, typically at 30°C for 16 to 24 hours, which contrasts sharply with the high temperatures or cryogenic conditions often required by older methods. The use of diaryliodonium salts as oxidants allows for the efficient construction of the imidazole ring while incorporating the trifluoromethyl group in a single pot. This strategy not only simplifies the operational procedure but also broadens the utility of the method by accommodating a wide range of functional groups. The ability to synthesize diversely substituted imidazole compounds through simple substrate design makes this a powerful tool for cost reduction in API manufacturing.
Mechanistic Insights into Pd-Catalyzed Carbonylation Tandem Reaction
The mechanistic pathway of this transformation is a testament to the elegance of modern organometallic catalysis. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation, yielding a trifluoroacetamidine compound. This intermediate subsequently undergoes isomerization, followed by palladium-catalyzed alkynyl amine palladation to generate a vinyl palladium intermediate. Further isomerization leads to an alkyl palladium species, which is then subjected to a carbonylation reaction. Crucially, the carbon monoxide required for this step is released in situ from a mixture of formic acid and acetic anhydride, eliminating the need for handling toxic CO gas cylinders. The resulting acyl palladium intermediate then undergoes oxidative addition with the diaryliodonium salt to form a tetravalent palladium species. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted imidazole compound and regenerates the active catalyst.
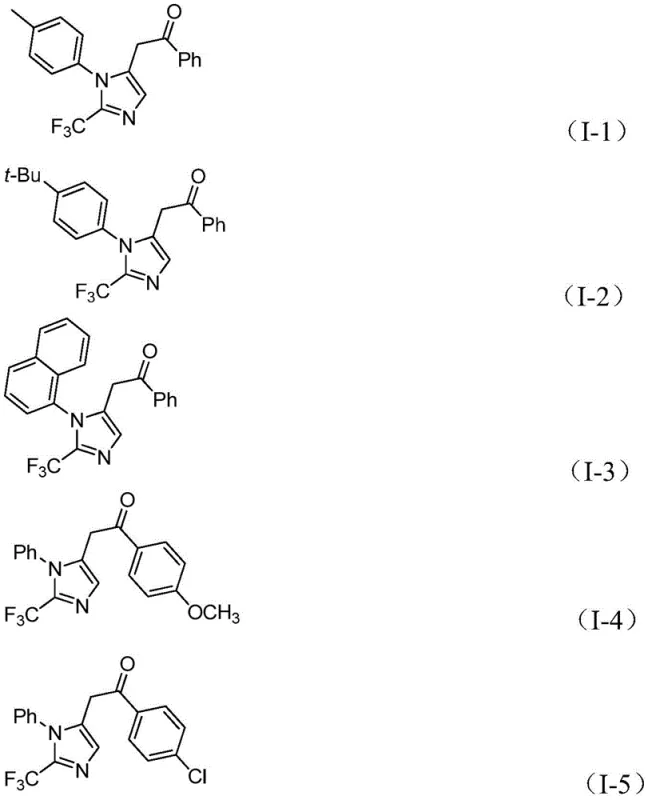
Understanding this mechanism is vital for controlling the impurity profile of the final product. The specific choice of ligands, such as triphenylphosphine, and the precise molar ratios of reagents play a pivotal role in steering the reaction towards the desired product while minimizing side reactions. The patent highlights that the method exhibits excellent substrate compatibility, tolerating various substituents on the aryl rings, including methyl, tert-butyl, chloro, bromo, trifluoromethyl, and nitro groups. This tolerance ensures that the electronic and steric properties of the starting materials do not detrimentally affect the catalytic cycle. For quality control teams, this mechanistic robustness translates to a more predictable impurity spectrum, facilitating easier purification via standard column chromatography and ensuring the delivery of high-purity OLED material or pharmaceutical intermediates.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The experimental protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale synthesis. The process begins by charging a reaction vessel with palladium chloride, triphenylphosphine, sodium bicarbonate, and a mixture of acetic anhydride and formic acid in an organic solvent, preferably tetrahydrofuran (THF). To this mixture, the key reactants—trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt—are added. The reaction is maintained at a constant temperature of 30°C for a duration of 16 to 24 hours to ensure complete conversion. Upon completion, the crude mixture is filtered and mixed with silica gel before undergoing purification via column chromatography. This straightforward workup procedure minimizes waste and maximizes recovery. Detailed standardized synthesis steps follow below for technical reference.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction mixture under stirring.
- React at 30°C for 16 to 24 hours, then filter, mix with silica gel, and purify via column chromatography to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The shift towards this palladium-catalyzed method addresses several pain points associated with traditional heterocycle synthesis, specifically regarding raw material availability, safety, and process scalability. By utilizing starting materials that are commercially available and inexpensive, such as aromatic amines used to prepare the imidoyl chloride, the overall cost structure of the synthesis is significantly optimized. Furthermore, the avoidance of hazardous reagents like trifluorodiazoethane reduces the regulatory burden and safety infrastructure costs associated with manufacturing. This aligns perfectly with the goals of a reliable pharmaceutical intermediate supplier seeking to enhance operational efficiency.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of cheap and readily available starting materials. Trifluoroethylimidoyl chloride can be rapidly synthesized from aromatic amines, triphenylphosphine, carbon tetrachloride, and trifluoroacetic acid, all of which are commodity chemicals. Additionally, the catalyst system employs palladium chloride, which, while a precious metal, is used in low loading (5 mol%) and is more cost-effective than many specialized catalysts. The mild reaction temperature of 30°C significantly reduces energy consumption compared to processes requiring reflux or cryogenic cooling. Eliminating the need for high-pressure carbon monoxide equipment further lowers capital expenditure, leading to substantial cost savings in the final product pricing.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the broad availability of the requisite reagents. The diaryliodonium salts can be conveniently prepared from arylphenylboronic acids and aryl iodides, ensuring a stable upstream supply. The robustness of the reaction against various functional groups means that supply disruptions for specific substituted anilines can often be mitigated by switching to alternative substrates without redesigning the entire process. This flexibility is crucial for maintaining the commercial scale-up of complex polymer additives or drug intermediates. The simplicity of the post-processing, involving filtration and standard chromatography, ensures that production timelines are not delayed by complex purification bottlenecks, thereby reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: The patent explicitly states that the method can be expanded to the gram level, providing a solid foundation for industrial large-scale production applications. The use of THF as a solvent is well-established in the industry, and the reaction generates minimal hazardous waste compared to diazo-based routes. The high reaction efficiency and good substrate compatibility mean that fewer batches are rejected due to poor yield or purity issues, enhancing overall process mass intensity. From an environmental perspective, the avoidance of explosive diazo compounds and the use of formic acid as a CO source represent greener chemistry principles. This facilitates easier compliance with increasingly stringent environmental regulations governing chemical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific technical details and beneficial effects outlined in the patent documentation. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this method into their existing supply chains.
Q: What are the key advantages of introducing a trifluoromethyl group into imidazole compounds?
A: Introducing a trifluoromethyl group significantly enhances the electronegativity, bioavailability, metabolic stability, and lipophilicity of the parent molecule, making it highly valuable for drug design.
Q: Why is this palladium-catalyzed method superior to conventional trifluoromethylation techniques?
A: Unlike conventional methods that often rely on hazardous trifluorodiazoethane, this method utilizes cheap and readily available trifluoroethylimidoyl chloride under mild conditions (30°C) with excellent substrate compatibility.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method features simple operation, inexpensive starting materials, and has been demonstrated to be scalable to the gram level, providing a strong foundation for industrial large-scale production applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111423381A. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, ensuring that every batch of 2-trifluoromethyl imidazole intermediate meets the highest global standards. We understand that transitioning a new synthetic route from the lab to the plant requires deep technical expertise and a partner who values precision and reliability above all else.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective supply of these critical building blocks for your pharmaceutical or agrochemical pipelines.
