Advanced Resolution of Axially Chiral Oxindole Styrenes for High-Value Catalysis
Advanced Resolution of Axially Chiral Oxindole Styrenes for High-Value Catalysis
The landscape of asymmetric synthesis is continuously evolving, driven by the demand for structurally complex chiral scaffolds that serve as critical building blocks in modern drug discovery and materials science. Patent CN111848322B introduces a groundbreaking methodology for the resolution of axially chiral oxindole-substituted styrene compounds, addressing a significant gap in the availability of these high-value intermediates. Unlike traditional approaches that often rely on harsh conditions or scarce metal catalysts, this invention leverages a sophisticated organocatalytic strategy using chiral phosphoric acids to achieve exceptional stereocontrol. For R&D directors and procurement specialists alike, this technology represents a pivotal shift towards more sustainable and efficient manufacturing pathways for complex chiral molecules. The ability to access these axially chiral frameworks with high optical purity opens new avenues for developing next-generation ligands and catalysts, positioning this patent as a cornerstone for future innovation in fine chemical synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of axially chiral styrene skeletons has been fraught with significant synthetic challenges and limitations that hinder large-scale adoption. Prior art predominantly relied on nucleophilic addition reactions of alkynes or coupling reactions involving aryl halides and alkenes, often necessitating the use of expensive transition metal catalysts such as palladium or titanium. These conventional methods frequently suffer from narrow substrate scope, requiring specific electronic properties on the aromatic rings to proceed efficiently, which limits their utility in diverse medicinal chemistry campaigns. Furthermore, the reliance on heavy metals introduces substantial downstream processing burdens, including the need for rigorous metal scavenging steps to meet stringent regulatory limits for pharmaceutical ingredients. The lack of efficient resolution methods for racemic styrene compounds meant that valuable chiral information was often lost, forcing chemists to resort to lengthy asymmetric synthesis routes that were neither atom-economical nor cost-effective for industrial applications.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN111848322B utilizes a dynamic kinetic resolution or direct enantioselective functionalization strategy mediated by chiral phosphoric acid catalysts. This organocatalytic approach operates under remarkably mild conditions, typically in dichloromethane at temperatures between 0°C and 10°C, thereby preserving sensitive functional groups that might degrade under harsher thermal or metallic conditions. The reaction employs readily available racemic precursors (Formula 1) and reacts them with specific partners (Formula 2) in the presence of 3A molecular sieves to drive the equilibrium towards the desired chiral product (Formula 4). 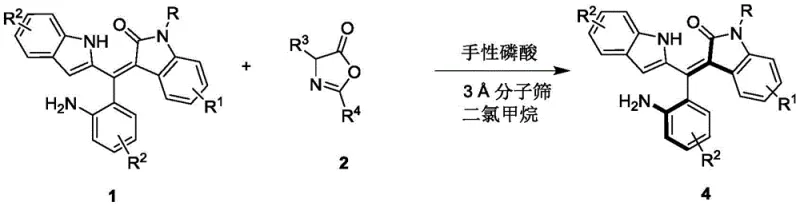 . This novel pathway not only simplifies the operational complexity by eliminating metal catalysts but also demonstrates broad substrate tolerance, accommodating various substituents on the indole and phenyl rings without compromising enantioselectivity, thus offering a robust platform for generating structural diversity.
. This novel pathway not only simplifies the operational complexity by eliminating metal catalysts but also demonstrates broad substrate tolerance, accommodating various substituents on the indole and phenyl rings without compromising enantioselectivity, thus offering a robust platform for generating structural diversity.
Mechanistic Insights into Chiral Phosphoric Acid Catalyzed Resolution
The success of this resolution strategy lies in the precise molecular recognition and activation facilitated by the chiral phosphoric acid catalyst, which acts as a bifunctional organocatalyst. The catalyst likely engages the substrate through a dual hydrogen-bonding network, simultaneously activating the electrophilic and nucleophilic components while imposing a rigid chiral environment that dictates the trajectory of the bond formation. This supramolecular assembly ensures that the reaction proceeds through a highly organized transition state, effectively discriminating between the enantiomers of the racemic starting material or controlling the facial selectivity of the addition. The use of binaphthyl-derived phosphoric acids (Formula 3), particularly those with bulky 2-naphthyl groups at the 3,3'-positions, creates a confined chiral pocket that sterically shields one face of the reacting species, leading to the observed high levels of enantiomeric excess (up to 96% ee). Such mechanistic precision is critical for R&D teams aiming to replicate these results, as it underscores the importance of catalyst loading and steric bulk in achieving optimal stereochemical outcomes.
Beyond the primary resolution, the utility of the resulting axially chiral styrenes is further amplified through their derivatization into functional chiral catalysts, demonstrating a cascade of value creation from a single synthetic operation. The resolved compound (Formula 4a) can be efficiently transformed into a chiral tertiary amine catalyst (Formula 5) via a two-step sequence involving thiocarbonylation and amidation. 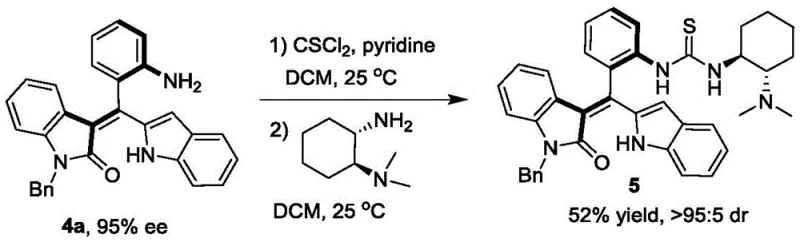 . This derivative retains the axial chirality of the parent scaffold and introduces a basic nitrogen center capable of activating substrates through non-covalent interactions. When applied to the asymmetric [4+2] cyclization of o-methylenebenzoquinones and malononitrile, this new catalyst exhibits superior performance compared to commercially available alternatives, achieving yields of 90% and 91% ee. This mechanistic versatility highlights the dual role of the patented compounds as both valuable intermediates and potent catalysts, offering a compelling value proposition for supply chain managers looking to consolidate vendor relationships.
. This derivative retains the axial chirality of the parent scaffold and introduces a basic nitrogen center capable of activating substrates through non-covalent interactions. When applied to the asymmetric [4+2] cyclization of o-methylenebenzoquinones and malononitrile, this new catalyst exhibits superior performance compared to commercially available alternatives, achieving yields of 90% and 91% ee. This mechanistic versatility highlights the dual role of the patented compounds as both valuable intermediates and potent catalysts, offering a compelling value proposition for supply chain managers looking to consolidate vendor relationships.
How to Synthesize Axially Chiral Oxindole-Substituted Styrene Efficiently
The practical implementation of this resolution technology is designed to be straightforward and scalable, aligning with the needs of industrial process chemistry where reproducibility and safety are paramount. The standard protocol involves dissolving the racemic precursor and the coupling partner in dry dichloromethane, followed by the addition of activated 3A molecular sieves to sequester trace water that could inhibit the catalyst. The chiral phosphoric acid is then introduced at a loading of 10 mol%, and the mixture is stirred at a controlled low temperature of 0°C to ensure maximum stereocontrol. Detailed standardized synthesis steps see the guide below.
- Combine racemic Formula 1 compound and Formula 2 compound in dichloromethane with 3A molecular sieves.
- Add 10 mol% chiral phosphoric acid catalyst (Formula 3) and stir at 0°C to 10°C.
- Monitor reaction via TLC, then filter, concentrate, and purify via silica gel column chromatography to obtain Formula 4.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic resolution method offers tangible strategic advantages that extend beyond mere technical feasibility. By shifting away from transition metal-dependent processes, manufacturers can significantly reduce the complexity of their waste streams and eliminate the costly purification steps associated with heavy metal removal. This transition not only lowers the overall cost of goods sold but also mitigates regulatory risks associated with residual metal limits in active pharmaceutical ingredients. Furthermore, the mild reaction conditions (0°C to 10°C) imply lower energy consumption for heating or cooling compared to processes requiring cryogenic temperatures or high-pressure reactors, contributing to a more sustainable and cost-efficient manufacturing footprint. The robustness of the catalyst system across a wide range of substrates ensures supply continuity, as variations in raw material specifications are less likely to cause batch failures, thereby enhancing the reliability of the supply chain for critical chiral intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or titanium catalysts directly reduces raw material costs, while the simplified workup procedure minimizes solvent usage and processing time. The absence of metal scavengers and the ability to use standard silica gel chromatography for purification streamline the production workflow, leading to substantial operational savings. Additionally, the high atom economy of the resolution process ensures that a greater proportion of the input mass is converted into valuable product, reducing waste disposal costs and improving overall process efficiency.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including the chiral phosphoric acid and the organic substrates, are derived from commercially available feedstocks, reducing dependency on specialized or single-source suppliers. The stability of the reaction under ambient pressure and moderate temperatures allows for execution in standard glass-lined reactors, facilitating easy scale-up from laboratory to commercial production without the need for specialized equipment. This accessibility ensures that production schedules can be maintained consistently, reducing lead times for high-purity pharmaceutical intermediates and safeguarding against supply disruptions.
- Scalability and Environmental Compliance: The use of dichloromethane as a solvent, while common, is managed effectively within closed systems, and the potential for solvent recovery further enhances the environmental profile of the process. The organocatalytic nature of the reaction aligns with green chemistry principles by avoiding toxic heavy metals, making the resulting waste easier to treat and dispose of in compliance with increasingly stringent environmental regulations. The demonstrated scalability of similar organocatalytic processes suggests that this method can be readily adapted for multi-kilogram or ton-scale production, supporting the long-term growth demands of the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology, based on the detailed experimental data provided in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing pipelines. The answers reflect the specific conditions and outcomes reported in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of this resolution method over traditional metal-catalyzed couplings?
A: This organocatalytic method eliminates the need for expensive and toxic transition metals like palladium or titanium, operating under mild conditions (0°C) with high enantioselectivity (up to 96% ee).
Q: Can the resolved styrene compounds be further derivatized?
A: Yes, the resolved axially chiral styrenes (Formula 4) can be efficiently converted into chiral tertiary amine catalysts (Formula 5) which demonstrate superior performance in asymmetric [4+2] cyclization reactions.
Q: What represents the optimal reaction temperature for this resolution process?
A: The patent specifies an optimal reaction temperature range of 0°C to 10°C, with 0°C being preferred to maximize enantioselectivity and control reaction kinetics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Axially Chiral Oxindole-Substituted Styrene Supplier
As the global demand for enantiomerically pure intermediates continues to surge, NINGBO INNO PHARMCHEM stands ready to support your development goals with our expertise in complex organic synthesis. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including advanced chiral HPLC analysis to guarantee the optical purity of every batch we deliver. By leveraging the innovative strategies outlined in patents like CN111848322B, we can offer customized synthesis solutions that optimize both cost and quality for your specific project requirements.
We invite you to engage with our technical procurement team to discuss how we can tailor this resolution technology to your unique supply chain needs. Whether you require a Customized Cost-Saving Analysis for your current route or need to evaluate the feasibility of scaling this organocatalytic process, our experts are prepared to provide comprehensive support. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
