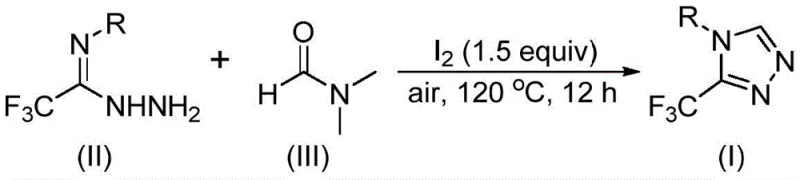
Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles via DMF Carbon Source Activation for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand efficient access to fluorinated heterocycles, particularly 3-trifluoromethyl substituted 1,2,4-triazole compounds, due to their profound impact on metabolic stability and bioactivity. A significant technological breakthrough in this domain is documented in patent CN114920707B, which discloses a novel preparation method utilizing the ubiquitous organic solvent DMF as a carbon source. This innovation represents a paradigm shift from traditional multi-step syntheses to a more atom-economical tandem cyclization approach. By leveraging molecular iodine as a promoter under aerobic conditions, this method circumvents the need for expensive transition metal catalysts or harsh anhydrous environments. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding this technology is crucial as it offers a pathway to high-purity OLED material precursors and API intermediates with substantially reduced manufacturing complexity. The ability to synthesize these core skeletons efficiently directly translates to enhanced supply chain resilience and cost competitiveness in the global fine chemical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted 1,2,4-triazole scaffolds has been plagued by significant synthetic challenges that hinder large-scale commercial adoption. Traditional routes often rely on pre-functionalized building blocks that are costly to procure and require stringent storage conditions to prevent degradation. Furthermore, many established protocols necessitate the use of heavy metal catalysts, which introduce severe downstream purification burdens to meet regulatory limits on residual metals in active pharmaceutical ingredients. The requirement for strictly anhydrous and oxygen-free environments in conventional methods further escalates operational expenditures, demanding specialized reactor setups and inert gas manifolds that are not always available in standard multipurpose plants. These factors collectively contribute to extended lead times and volatile pricing structures, creating bottlenecks for supply chain heads who prioritize continuity and predictability in raw material sourcing for complex polymer additives or specialty chemical production.
The Novel Approach
In stark contrast, the methodology outlined in CN114920707B introduces a streamlined strategy that fundamentally simplifies the reaction architecture by dual-utilizing DMF as both the reaction medium and the carbon synthon. This approach eliminates the dependency on exotic reagents, relying instead on commercially abundant starting materials like trifluoroethyliminohydrazide and molecular iodine. The reaction proceeds smoothly under air atmosphere at moderate temperatures ranging from 110°C to 130°C, removing the logistical burden of maintaining inert conditions. This operational simplicity is a game-changer for cost reduction in electronic chemical manufacturing and pharmaceutical sectors, as it allows for the use of standard glass-lined reactors without specialized modifications. The robustness of this system ensures that the process is not only scientifically elegant but also industrially viable, offering a scalable solution that aligns perfectly with the needs of a reliable agrochemical intermediate supplier seeking to optimize production throughput.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic elegance of this transformation lies in the versatile reactivity of DMF, which can participate as a carbon source through either its formyl or N-methyl group depending on the specific reaction trajectory. When the formyl group acts as the carbon donor, it undergoes condensation with trifluoroethyliminohydrazide to form a hydrazone intermediate, which subsequently cyclizes intramolecularly with the elimination of dimethylamine to yield the target triazole. Alternatively, the N-methyl pathway involves the activation of DMF by molecular iodine to generate an amine salt species, followed by nucleophilic addition and oxidative aromatization. This dual-pathway capability underscores the chemical flexibility of the system, allowing for fine-tuning of reaction conditions to maximize yield and minimize byproduct formation. Understanding these mechanistic nuances is vital for process chemists aiming to replicate high-purity pharmaceutical intermediates, as it provides the theoretical foundation for troubleshooting and optimization during technology transfer.
Furthermore, the impurity profile of this reaction is inherently cleaner compared to metal-catalyzed alternatives, primarily because the promoter is molecular iodine which is easily removed during aqueous workup. The absence of transition metals means there is no risk of metal-ligand complexation side reactions that often complicate the purification of bioactive molecules like Factor IXa inhibitors. The oxidative aromatization step ensures the formation of a stable aromatic triazole ring, driving the equilibrium towards the product and enhancing overall conversion rates. For quality assurance teams, this mechanistic clarity translates to more predictable analytical results and easier validation of the manufacturing process. The ability to control the reaction pathway through stoichiometry and temperature allows manufacturers to consistently deliver materials that meet stringent purity specifications required for clinical trial applications and commercial drug substance production.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and thermal parameters to ensure optimal performance. The general procedure involves charging a reaction vessel with molecular iodine, the hydrazide substrate, and excess DMF, followed by heating to the specified range of 110-130°C for a duration of 10-15 hours. Post-reaction processing is straightforward, involving filtration to remove inorganic salts, followed by standard extraction and purification techniques such as column chromatography or recrystallization. This operational simplicity makes it an ideal candidate for rapid scale-up from laboratory benchtop to pilot plant operations. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results with high fidelity.
- Combine molecular iodine, trifluoroethyliminohydrazide, and DMF solvent in a reaction vessel under air atmosphere.
- Heat the reaction mixture to 110-130°C and maintain stirring for 10-15 hours to ensure complete conversion.
- Perform post-treatment including filtration, washing, and column chromatography to isolate the pure triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that address critical pain points in the global chemical supply chain. The substitution of expensive, specialized reagents with commodity chemicals like DMF and iodine drastically lowers the bill of materials, providing substantial cost savings without compromising product quality. Moreover, the tolerance for air and moisture significantly reduces the capital expenditure required for facility upgrades, allowing existing infrastructure to be utilized for production. This flexibility enhances supply chain reliability by mitigating risks associated with the sourcing of sensitive reagents that may have long lead times or limited vendor availability. For procurement managers, this translates to a more resilient sourcing strategy capable of withstanding market fluctuations and geopolitical disruptions.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of costly transition metal catalysts and the use of DMF as a dual-purpose solvent and reactant. By removing the need for expensive ligands and specialized anhydrous solvents, the overall raw material cost is significantly reduced. Additionally, the simplified workup procedure reduces labor hours and solvent consumption during purification, further driving down the cost of goods sold. These efficiencies allow suppliers to offer more competitive pricing structures while maintaining healthy margins, making it an attractive option for cost-sensitive projects in generic drug manufacturing.
- Enhanced Supply Chain Reliability: The reliance on readily available, commodity-grade starting materials ensures a stable and continuous supply of inputs for production. Unlike specialized reagents that may be subject to allocation or single-source restrictions, DMF and molecular iodine are produced globally in vast quantities. This abundance minimizes the risk of production stoppages due to raw material shortages. Furthermore, the robust nature of the reaction conditions means that manufacturing can proceed without interruption even if minor variations in environmental controls occur, ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling this process to industrial levels is facilitated by the absence of hazardous pyrophoric reagents or high-pressure equipment requirements. The reaction operates at atmospheric pressure and moderate temperatures, aligning well with standard safety protocols in chemical plants. From an environmental standpoint, the reduced solvent waste and absence of heavy metal contamination simplify waste stream management and disposal. This compliance with green chemistry principles not only reduces environmental fees but also enhances the corporate sustainability profile of the manufacturing entity, appealing to eco-conscious partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific beneficial effects and background art analysis within the patent documentation to provide clarity on feasibility and application. Understanding these details helps stakeholders make informed decisions about integrating this method into their existing production portfolios.
Q: What are the primary advantages of using DMF as a carbon source in this synthesis?
A: Using DMF eliminates the need for specialized carbon sources, leveraging a common solvent as a reactant which significantly simplifies the supply chain and reduces raw material costs while maintaining high reaction efficiency.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, the patented process operates effectively under air atmosphere without the need for anhydrous conditions, drastically reducing operational complexity and equipment requirements for commercial production.
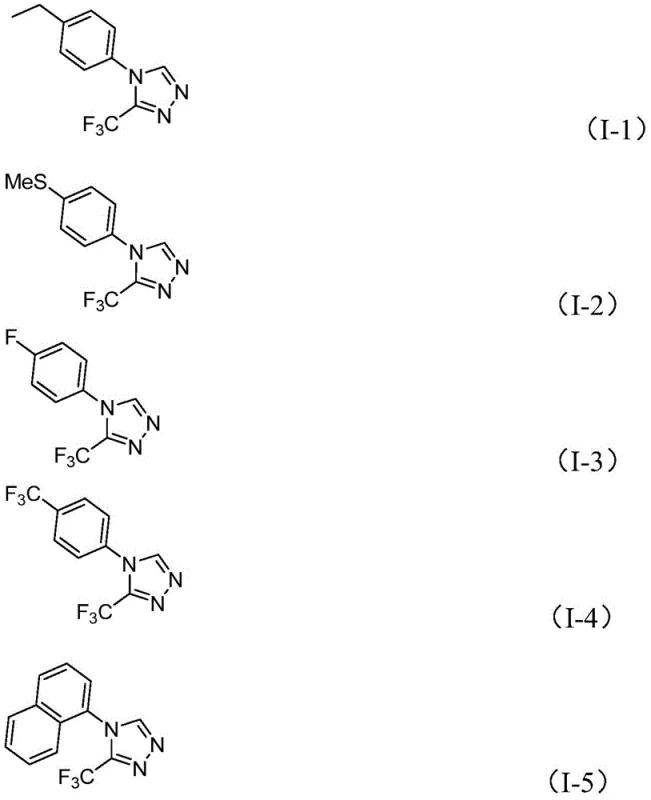
Q: What is the substrate scope for the R-group in this triazole synthesis?
A: The method demonstrates broad compatibility with various substituents including alkyl, alkoxy, halogen, and trifluoromethyl groups on the phenyl ring, allowing for diverse derivative production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN114920707B for developing next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from concept to market. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-trifluoromethyl-1,2,4-triazole intermediate meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this DMF-based route for your specific needs, delivering high-quality materials that accelerate your drug development timelines.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of adopting this technology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to build a more efficient and cost-effective future for your chemical manufacturing needs.
