Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Imidazoles for Commercial Pharmaceutical Applications
The landscape of modern pharmaceutical synthesis is constantly evolving to meet the demands for higher purity, safer processes, and more efficient access to complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN111423381A, which discloses a novel preparation method for 2-trifluoromethyl substituted imidazole compounds. These fluorinated heterocycles are critical structural motifs found in numerous bioactive molecules, including antihistamines like Alcaftadine and various kinase inhibitors, where the trifluoromethyl group enhances metabolic stability and lipophilicity. The disclosed technology leverages a transition metal palladium-catalyzed carbonylation cascade reaction, utilizing cheap and readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salts. This approach represents a paradigm shift from traditional methodologies, offering a robust pathway for the reliable pharmaceutical intermediate supplier seeking to optimize their production lines for next-generation drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of a trifluoromethyl group into nitrogen-containing heterocycles has been fraught with synthetic challenges and safety hazards. Conventional literature often relies on the use of highly unstable and explosive synthons like trifluorodiazoethane, which poses severe risks during storage and handling on a large scale. Furthermore, many existing protocols require harsh reaction conditions, such as elevated temperatures or the use of toxic carbon monoxide gas supplied from high-pressure cylinders, which complicates reactor design and increases operational costs. The substrate scope in these older methods is frequently limited, struggling to tolerate diverse functional groups without significant degradation or side reactions. Consequently, the cost reduction in API manufacturing is often hindered by low yields, difficult purification processes, and the need for specialized safety infrastructure to manage hazardous reagents, making the commercial viability of such routes questionable for large-scale production.
The Novel Approach
In stark contrast, the methodology described in patent CN111423381A introduces a remarkably mild and versatile strategy that circumvents these historical bottlenecks. By employing trifluoroethylimidoyl chloride as a stable trifluoromethyl synthon, the process eliminates the need for dangerous diazo compounds while maintaining high reactivity. The reaction proceeds efficiently at a mild temperature of 30°C, utilizing a formic acid and acetic anhydride mixture as a safe carbon monoxide surrogate, thereby removing the logistical burden of handling toxic CO gas. This novel approach demonstrates exceptional substrate compatibility, allowing for the synthesis of diversified imidazole derivatives with various substituents on the aryl rings, as illustrated by the successful preparation of compounds (I-1) through (I-5). 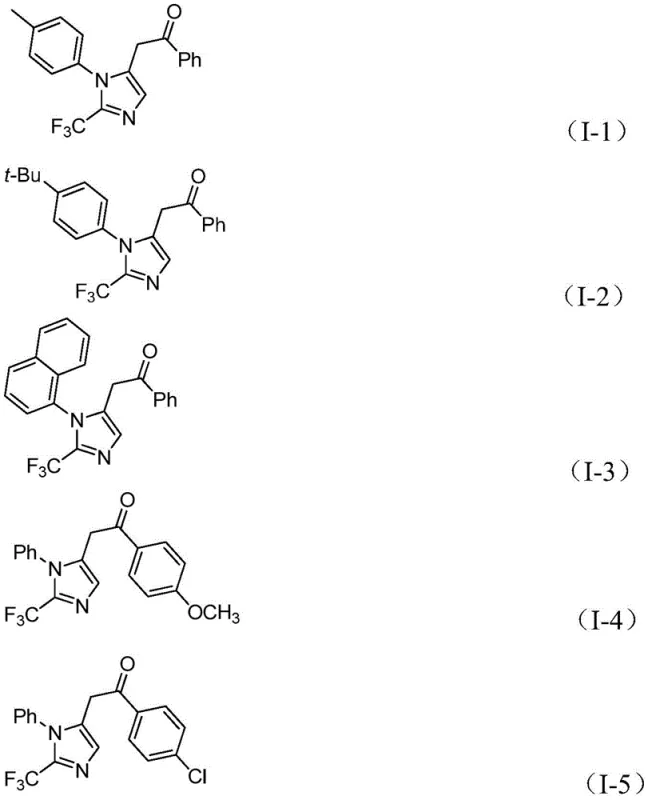 This flexibility enables medicinal chemists to rapidly explore structure-activity relationships (SAR) without being constrained by synthetic limitations, ultimately accelerating the drug discovery timeline and enhancing the utility of this method for producing high-purity OLED material precursors or pharmaceutical intermediates.
This flexibility enables medicinal chemists to rapidly explore structure-activity relationships (SAR) without being constrained by synthetic limitations, ultimately accelerating the drug discovery timeline and enhancing the utility of this method for producing high-purity OLED material precursors or pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The elegance of this synthesis lies in its intricate yet efficient catalytic cycle, which orchestrates multiple bond-forming events in a single pot. The reaction initiates with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and propargylamine, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization, setting the stage for the pivotal palladium-catalyzed steps. The palladium catalyst, typically generated in situ from PdCl2 and PPh3, facilitates the aminopalladation of the alkyne moiety to form an alkenyl-palladium intermediate, which further isomerizes to a more stable alkyl-palladium species. 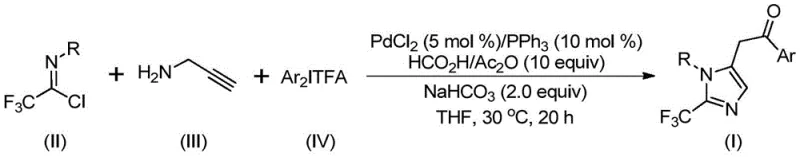 Crucially, the carbon monoxide released from the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond, forming an acyl-palladium intermediate. This is followed by oxidative addition of the diaryliodonium salt, generating a high-valent tetravalent palladium species that finally undergoes reductive elimination to release the desired 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst.
Crucially, the carbon monoxide released from the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond, forming an acyl-palladium intermediate. This is followed by oxidative addition of the diaryliodonium salt, generating a high-valent tetravalent palladium species that finally undergoes reductive elimination to release the desired 2-trifluoromethyl substituted imidazole product and regenerate the active catalyst.
From a quality control perspective, understanding this mechanism is vital for managing the impurity profile of the final product. The use of diaryliodonium salts as oxidants is particularly advantageous because it avoids the formation of halogenated byproducts often associated with traditional oxidative coupling reagents. The mild reaction temperature of 30°C minimizes thermal decomposition pathways, ensuring that sensitive functional groups on the aromatic rings remain intact. Furthermore, the stoichiometric balance of reagents, specifically the slight excess of trifluoroethylimidoyl chloride relative to propargylamine, drives the reaction to completion while suppressing potential polymerization of the alkyne starting material. This precise control over the reaction kinetics results in a cleaner crude reaction mixture, simplifying the downstream purification process and ensuring that the final active pharmaceutical ingredient meets stringent purity specifications required by global regulatory bodies.
How to Synthesize 2-Trifluoromethyl Imidazole Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly attractive for both laboratory-scale optimization and pilot plant operations. The procedure involves charging a reaction vessel with the palladium catalyst system, additives, and the three key organic components in a suitable aprotic solvent such as tetrahydrofuran (THF). The reaction mixture is stirred at a controlled temperature for a defined period, after which standard workup procedures involving filtration and silica gel chromatography yield the pure product. For detailed operational parameters and specific stoichiometric ratios optimized for different substrates, please refer to the standardized synthesis guide below.
- Mix palladium chloride, triphenylphosphine, sodium bicarbonate, and a formic acid/acetic anhydride mixture in an organic solvent like THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and diaryliodonium salt to the reaction vessel under stirring.
- Maintain the reaction at 30°C for 16 to 24 hours, then filter and purify via column chromatography to isolate the target imidazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift towards this methodology addresses several critical pain points in the sourcing of complex heterocyclic building blocks, particularly regarding cost stability and supply continuity. By utilizing starting materials that are commercially available and inexpensive, such as aromatic amines and propargylamine, the dependency on exotic or custom-synthesized reagents is significantly reduced. This accessibility translates directly into a more resilient supply chain, mitigating the risks associated with raw material shortages that often plague the fine chemical industry. Moreover, the operational simplicity of the process reduces the barrier to entry for contract manufacturing organizations, fostering a more competitive supplier landscape.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the elimination of expensive and hazardous reagents. Traditional methods often require specialized equipment for handling high-pressure gases or cryogenic conditions, which entails significant capital expenditure and maintenance costs. In contrast, this palladium-catalyzed method operates at ambient pressure and mild temperatures, allowing for the use of standard glass-lined or stainless steel reactors without modification. The catalyst system itself, based on palladium chloride and triphenylphosphine, is cost-effective compared to exotic ligand systems, and the high turnover efficiency ensures that catalyst loading remains low. Additionally, the avoidance of toxic CO gas cylinders removes the need for costly safety monitoring systems and specialized gas delivery infrastructure, leading to substantial cost savings in facility operations and compliance.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the stability and shelf-life of the key reagents involved in this synthesis. Trifluoroethylimidoyl chlorides and diaryliodonium salts are solid or stable liquid compounds that can be stored and transported under standard conditions, unlike gaseous reagents or unstable diazo compounds that require immediate use or specialized cold chain logistics. This stability allows manufacturers to maintain strategic stockpiles of raw materials, buffering against market volatility and ensuring consistent production schedules. The robustness of the reaction conditions also means that production is less susceptible to disruptions caused by minor fluctuations in utility supplies, such as cooling water temperature variations, further enhancing the predictability of delivery timelines for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this methodology is inherently designed for scalability. The use of THF as a solvent is well-established in the industry, with mature recovery and recycling protocols that minimize waste generation. The reaction produces minimal hazardous byproducts, and the post-treatment involves straightforward filtration and chromatography, avoiding complex aqueous workups that generate large volumes of wastewater. From an environmental compliance standpoint, the replacement of toxic carbon monoxide gas with a liquid CO surrogate significantly reduces the facility's risk profile and simplifies permitting processes. This green chemistry advantage aligns with the increasing regulatory pressure on pharmaceutical manufacturers to reduce their environmental footprint, making this route a future-proof choice for sustainable commercial scale-up of complex polymer additives or drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this chemistry into their portfolio. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the key advantages of this palladium-catalyzed method over traditional trifluoromethylation?
A: Unlike traditional methods that often rely on hazardous trifluorodiazoethane or harsh conditions, this protocol utilizes stable trifluoroethylimidoyl chlorides and mild temperatures (30°C), significantly improving operational safety and substrate compatibility.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the patent explicitly demonstrates that the method can be expanded to the gram level with high efficiency. The use of commercially available catalysts and simple post-processing suggests strong potential for commercial scale-up of complex pharmaceutical intermediates.
Q: What is the role of the formic acid and acetic anhydride mixture in this reaction?
A: This mixture serves as a safe and effective carbon monoxide (CO) surrogate. It releases CO in situ under the reaction conditions, eliminating the need for handling toxic high-pressure CO gas cylinders while driving the carbonylation step of the catalytic cycle.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Imidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111423381A for the production of high-value fluorinated heterocycles. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Our expertise in palladium catalysis and carbonylation chemistry allows us to troubleshoot and optimize this specific route, guaranteeing consistent quality and yield for your critical projects.
We invite you to collaborate with us to leverage this cutting-edge technology for your upcoming drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We encourage you to reach out today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules, ensuring that your supply chain is built on a foundation of innovation, reliability, and scientific excellence.
