Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)-one Derivatives for Commercial Scale-up
Introduction to Next-Generation Quinolinone Synthesis
The landscape of heterocyclic chemistry is continually evolving, driven by the demand for more efficient and sustainable pathways to bioactive scaffolds. A significant breakthrough in this domain is detailed in patent CN113045489B, which discloses a novel preparation method for 3-arylquinoline-2(1H)one derivatives. These compounds are not merely academic curiosities; they represent a critical class of pharmacophores found in numerous natural products and therapeutic agents, ranging from antibiotics and antiplatelet drugs to potent antitumor candidates and endothelin receptor antagonists. The traditional reliance on complex multi-step syntheses often hampers the rapid development of these valuable intermediates. However, this new methodology revolutionizes the approach by utilizing benzisoxazole as a dual-purpose reagent, acting simultaneously as the nitrogen source and the formyl source. This innovation simplifies the synthetic architecture significantly, offering a streamlined route that is highly attractive for industrial application.
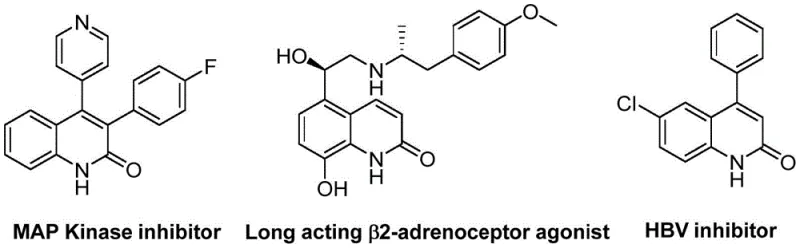
For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the versatility of this scaffold is paramount. As illustrated in the structural diversity of known bioactive molecules, the 3-arylquinolin-2(1H)one core is a privileged structure in medicinal chemistry. The ability to access this core efficiently opens doors to accelerating drug discovery pipelines. The patent highlights that this method is not limited to a narrow subset of substrates but boasts a wide functional group tolerance, allowing for the synthesis of a broad array of derivatives tailored to specific biological targets. This adaptability ensures that the supply chain for these critical building blocks remains robust and responsive to the changing needs of the pharmaceutical industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied on classical methodologies such as the Vilsmeier-Haack, Knorr, and Friedlander reactions. While these methods have served the community well for decades, they are increasingly viewed through the lens of modern green chemistry and process efficiency as being suboptimal. Conventional routes often necessitate harsh reaction conditions, including strong acids or bases, elevated temperatures that can degrade sensitive functional groups, and the use of toxic reagents. Furthermore, many traditional pathways require pre-functionalized starting materials that are themselves expensive or difficult to procure, adding layers of complexity and cost to the overall manufacturing process. Transition metal-catalyzed improvements and ring-closure metathesis have been explored, yet they frequently suffer from limitations regarding substrate scope or the requirement for hazardous carbon monoxide gas sources, posing significant safety and logistical challenges for large-scale operations.
The Novel Approach
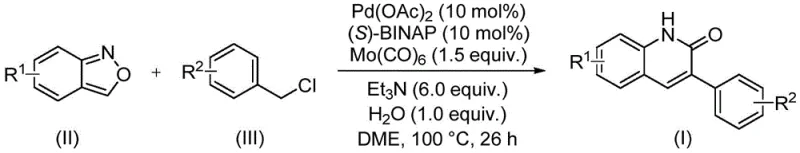
In stark contrast, the methodology described in CN113045489B offers a paradigm shift by employing a palladium-catalyzed aminocarbonylation strategy that is both operationally simple and chemically elegant. By leveraging benzisoxazole and benzyl chloride compounds as the primary starting materials, this novel approach circumvents the need for external carbon monoxide gas, utilizing molybdenum hexacarbonyl as a safe, solid CO surrogate instead. This substitution drastically reduces the safety risks associated with high-pressure CO handling, making the process far more amenable to standard reactor setups. The reaction proceeds under relatively mild conditions at 100°C, demonstrating exceptional efficiency with reaction times optimized around 26 hours. This new route not only simplifies the operational workflow but also expands the practical utility of the synthesis, enabling the production of diverse derivatives that were previously challenging to access via conventional means.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this technological advancement lies in the sophisticated interplay between the palladium catalyst and the unique reactivity of the benzisoxazole ring. The reaction mechanism initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species, generated in situ from palladium acetate and the chiral ligand (S)-1,1'-binaphthyl-2,2'-bisdiphenylphosphine ((S)-BINAP). This step forms a key organopalladium intermediate that is poised for carbonyl insertion. Uniquely, the carbonyl moiety is derived from the decomposition of molybdenum hexacarbonyl, which releases CO under the reaction conditions. The subsequent coordination and insertion of this CO into the palladium-carbon bond create an acyl-palladium complex. Simultaneously, the benzisoxazole undergoes ring opening, likely facilitated by the basic environment provided by triethylamine and the presence of water, releasing the necessary nitrogen nucleophile.
This nitrogen species then attacks the acyl-palladium intermediate, followed by reductive elimination to forge the new carbon-nitrogen bond and close the quinolinone ring, regenerating the active palladium catalyst to complete the cycle. The inclusion of water as a co-reagent is critical, potentially assisting in the hydrolysis or activation steps required for the benzisoxazole ring cleavage. From an impurity control perspective, the use of a well-defined catalytic system with specific ligands like (S)-BINAP helps minimize side reactions such as homocoupling of the benzyl chloride or non-selective carbonylation. The high yields reported, often exceeding 90% for unsubstituted or electron-rich substrates, indicate that the catalytic cycle is highly efficient and that competing degradation pathways are effectively suppressed. This mechanistic robustness is essential for ensuring the high purity required for pharmaceutical applications, reducing the burden on downstream purification processes.
How to Synthesize 3-Arylquinolin-2(1H)-one Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific protocols to maximize yield and reproducibility. The process is designed to be straightforward, utilizing commercially available reagents that do not require specialized synthesis prior to use. The standard procedure involves charging a sealed reaction vessel with the catalyst system, comprising palladium acetate and (S)-BINAP, along with the carbonyl source Mo(CO)6. To this mixture, the substrates—benzisoxazole and the specific benzyl chloride derivative—are added alongside the base triethylamine and a stoichiometric amount of water. The choice of solvent, ethylene glycol dimethyl ether (DME), is crucial for solubilizing the reactants and facilitating the reaction kinetics at the optimal temperature of 100°C. For a detailed breakdown of the standardized synthesis steps, please refer to the guide below.
- Charge a sealed tube with palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound.
- Add ethylene glycol dimethyl ether (DME) as the solvent and stir the mixture to ensure homogeneity.
- Heat the reaction mixture to 100°C for 26 hours, then filter, mix with silica gel, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the significant simplification of the raw material portfolio. By utilizing benzisoxazole and benzyl chlorides, which are commodity chemicals widely available in the global market, manufacturers can mitigate supply chain risks associated with exotic or custom-synthesized precursors. This accessibility ensures a stable supply of starting materials, reducing the likelihood of production delays due to raw material shortages. Furthermore, the elimination of gaseous carbon monoxide from the process removes the need for specialized high-pressure equipment and rigorous safety protocols associated with toxic gas handling, thereby lowering capital expenditure and operational overheads.
- Cost Reduction in Manufacturing: The economic implications of this method are profound, primarily driven by the use of inexpensive and readily available starting materials. Benzisoxazole and benzyl chloride derivatives are generally low-cost commodities compared to the complex intermediates required for traditional Knorr or Friedlander condensations. Additionally, the use of a solid carbonyl source (Mo(CO)6) instead of pressurized CO gas eliminates the infrastructure costs for gas storage and delivery systems. The high reaction efficiency and yields, often reaching up to 97% for optimized substrates, mean that less raw material is wasted, and the throughput per batch is maximized. This combination of cheap inputs and high conversion rates translates directly into a lower cost of goods sold (COGS), providing a competitive edge in the pricing of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the commercial availability of all reagents. The tolerance for a wide range of functional groups allows for the sourcing of diverse benzyl chloride derivatives without the need for extensive protection-deprotection strategies, which often complicate supply chains. The reaction operates at a moderate temperature of 100°C, which is easily achievable with standard heating mantles or oil baths, avoiding the need for cryogenic cooling or extreme high-temperature reactors. This operational simplicity ensures that the manufacturing process can be easily transferred between different facilities or scaled up without significant re-engineering, guaranteeing continuity of supply even in fluctuating market conditions.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns well with modern green chemistry principles. The avoidance of toxic CO gas reduces the environmental footprint and simplifies regulatory compliance regarding hazardous air pollutants. The workup procedure is equally streamlined, involving simple filtration followed by silica gel treatment and column chromatography, which are standard unit operations in fine chemical manufacturing. The high atom economy, where the benzisoxazole contributes both nitrogen and carbon to the final product, minimizes waste generation. This efficiency facilitates the commercial scale-up of complex pharmaceutical intermediates, allowing producers to move from gram-scale discovery to multi-ton commercial production with confidence in the process's environmental and economic sustainability.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific applications, we have compiled answers to common questions regarding the reaction parameters and scope. These insights are derived directly from the experimental data provided in the patent documentation, ensuring accuracy and relevance for process development. Understanding these nuances is critical for assessing the feasibility of adopting this route for your specific target molecules.
Q: What is the role of benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl (carbonyl) source, eliminating the need for external carbon monoxide gas.
Q: What are the optimal reaction conditions for high yield?
A: The optimal conditions involve reacting at 100°C for 26 hours in DME solvent with a Pd(OAc)2/(S)-BINAP catalyst system and Mo(CO)6 as the carbonyl source.
Q: Does this method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent tolerance for substituents such as methoxy, chloro, cyano, trifluoromethyl, and tert-butyl groups on both aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the Pd-catalyzed aminocarbonylation described in CN113045489B. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We maintain stringent purity specifications across all our output, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch of 3-arylquinolin-2(1H)-one derivatives we produce.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your drug development programs. Whether you require custom synthesis of specific derivatives or large-scale supply of the core scaffold, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your project. We encourage you to reach out to our technical procurement team to obtain specific COA data and discuss route feasibility assessments that can accelerate your path to market while optimizing your manufacturing costs.
