Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanone Scaffolds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 4-Acyl-2(5H)-Furanone Scaffolds for Commercial Scale-Up
The landscape of organic synthesis for bioactive heterocycles is constantly evolving, driven by the need for more efficient and scalable routes to complex molecular architectures. A significant advancement in this domain is detailed in patent CN112239436A, which discloses a robust preparation method for 4-acyl-2(5H)-furanone compounds. These specific furanone derivatives are not merely academic curiosities; they serve as critical structural backbones in a wide array of natural products and pharmacologically active molecules. For instance, related structures have demonstrated significant antimicrobial activity, potent analgesic properties, and even utility as antifouling agents in marine ecosystems, highlighting their immense value across the pharmaceutical and agrochemical sectors.
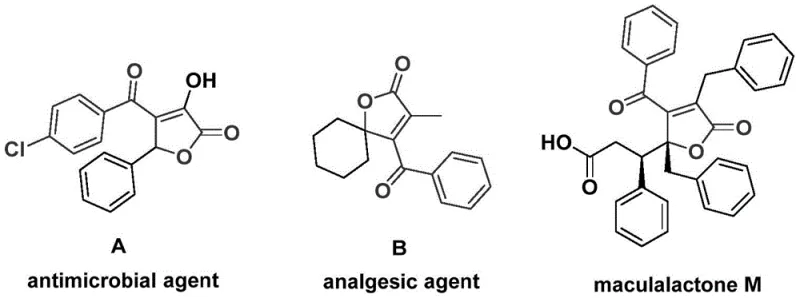
For R&D directors and process chemists, the ability to access these scaffolds efficiently is paramount. The traditional reliance on multi-step sequences or harsh reaction conditions often creates bottlenecks in drug discovery pipelines. However, the methodology outlined in the referenced patent offers a streamlined, one-pot solution that leverages palladium-catalyzed bis-carbonylation. By utilizing readily available starting materials such as propiolic alcohols and aryl trifluoromethanesulfonates, this process achieves high reaction efficiency and broad substrate compatibility, positioning it as a superior alternative for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4-acyl-2(5H)-furanone skeleton has been fraught with synthetic challenges that hinder large-scale adoption. Conventional pathways often require the use of gaseous carbon monoxide under high pressure, which introduces severe safety hazards and necessitates specialized high-pressure reactor equipment that is not universally available in standard pilot plants. Furthermore, many existing protocols suffer from poor atom economy, requiring multiple protection and deprotection steps to manage the reactivity of sensitive functional groups, which inevitably drives up the cost of goods sold (COGS). The lack of regioselectivity in some traditional cyclization reactions can also lead to difficult-to-separate isomeric mixtures, complicating the purification process and reducing the overall yield of the desired bioactive isomer.
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in patent CN112239436A utilizes a sophisticated palladium-catalyzed bis-carbonylation strategy that operates under relatively mild conditions. This method ingeniously employs chromium hexacarbonyl as a solid carbon monoxide surrogate, effectively bypassing the logistical and safety nightmares associated with handling toxic CO gas. The reaction proceeds in a single pot, directly coupling propiolic alcohols with aryl triflates to forge the complex furanone ring system with remarkable precision. As illustrated in the general reaction scheme below, this transformation is catalytic and highly convergent, allowing for the rapid assembly of diverse molecular libraries.
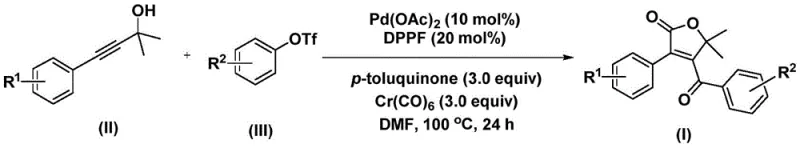
The operational simplicity of this new route cannot be overstated; it tolerates a wide range of substituents, including halogens and alkoxy groups, without the need for extensive optimization for each new substrate. This flexibility means that process chemists can rapidly iterate on lead compounds without being constrained by synthetic feasibility. Moreover, the use of common solvents like DMF and standard heating conditions (100 °C) ensures that the transition from milligram-scale discovery to kilogram-scale production is seamless, addressing a critical pain point in cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Bis-Carbonylation
To fully appreciate the robustness of this synthesis, one must delve into the intricate catalytic cycle that drives the formation of the furanone core. The reaction initiates with the oxidative addition of the palladium catalyst into the aryl trifluoromethanesulfonate bond, generating a reactive aryl-palladium intermediate. Subsequently, carbon monoxide, which is slowly liberated from the chromium carbonyl source, inserts into this palladium-carbon bond to form an acyl-palladium species. This step is crucial as it installs the first carbonyl functionality required for the final lactone structure, setting the stage for the subsequent cyclization event.
Following the initial carbonylation, the acyl-palladium intermediate undergoes a migratory insertion with the triple bond of the propiolic alcohol, leading to the formation of a five-membered ring palladium species. This cyclization is highly regioselective, driven by the electronic and steric properties of the ligand system, specifically the 1,1'-bis(diphenylphosphino)ferrocene (DPPF). A second molecule of carbon monoxide then inserts into this cyclic intermediate, expanding the coordination sphere before a final reductive elimination releases the 4-acyl-2(5H)-furanone product and regenerates the active palladium catalyst. This elegant mechanism ensures that impurities arising from alternative insertion pathways are minimized, resulting in a clean reaction profile that simplifies downstream processing.
How to Synthesize 4-Acyl-2(5H)-Furanone Efficiently
Implementing this synthesis in a practical setting requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process is designed to be user-friendly, utilizing reagents that are commercially available and stable under ambient conditions, which reduces the barrier to entry for contract manufacturing organizations. The reaction typically runs for 24 hours at 100 °C, providing ample time for the slow release of CO from the chromium source to drive the bis-carbonylation to completion without the risk of runaway exotherms. Detailed standardized operating procedures for this transformation are provided below to guide your technical teams.
- Combine palladium acetate, DPPF ligand, chromium carbonyl, p-toluquinone, propiolic alcohol, and aryl triflate in DMF solvent.
- Heat the reaction mixture to 100 °C and stir for 24 hours under inert atmosphere to facilitate bis-carbonylation and cyclization.
- Filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the high-purity 4-acyl-2(5H)-furanone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards this novel synthetic route represents a strategic opportunity to optimize vendor relationships and reduce dependency on complex, multi-step supply chains. The primary advantage lies in the consolidation of the synthetic sequence; by achieving the target scaffold in a single operational step from cheap starting materials, the number of unit operations is drastically reduced. This reduction directly correlates to lower labor costs, decreased solvent consumption, and a smaller physical footprint required for production, all of which contribute to substantial cost savings in fine chemical manufacturing without compromising on quality.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and hazardous reagents, specifically high-pressure carbon monoxide gas. By substituting this with solid chromium hexacarbonyl, the facility requirements are downgraded from high-pressure rated vessels to standard glass-lined or stainless steel reactors, representing a significant capital expenditure saving. Furthermore, the high conversion rates observed (often exceeding 90% for optimized substrates) mean that raw material waste is minimized, directly improving the margin profile for the final API intermediate.
- Enhanced Supply Chain Reliability: The starting materials for this reaction, namely substituted propiolic alcohols and aryl triflates, are derived from commodity chemicals that are widely produced globally. This abundance ensures that supply disruptions are unlikely, providing a stable foundation for long-term production planning. Additionally, the robustness of the reaction conditions means that batch-to-batch variability is low, ensuring consistent quality delivery to downstream customers and reducing the risk of rejected shipments due to specification failures.
- Scalability and Environmental Compliance: From an environmental perspective, the use of a solid CO source mitigates the risk of atmospheric emissions associated with gas leaks, aligning with stricter global environmental regulations. The workup procedure, which involves simple filtration and silica gel treatment, avoids the generation of large volumes of aqueous waste streams typical of acid/base extractions. This streamlined purification process facilitates easier scale-up from pilot plant to commercial tonnage, ensuring that the technology remains viable as demand for the bioactive furanone derivatives increases.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed furanone synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, aiming to clarify the operational feasibility for potential partners. Understanding these nuances is essential for evaluating the technology transfer potential and integration into existing manufacturing portfolios.
Q: How does this method address safety concerns regarding carbon monoxide usage?
A: The process utilizes chromium hexacarbonyl (Cr(CO)6) as a solid carbon monoxide substitute, eliminating the need for handling hazardous high-pressure CO gas cylinders, thereby significantly enhancing operational safety in standard laboratory or plant settings.
Q: What is the substrate compatibility for this furanone synthesis?
A: The methodology demonstrates excellent functional group tolerance, successfully accommodating electron-donating groups like methoxy and alkyl chains, as well as electron-withdrawing halogens such as fluorine, chlorine, and bromine on both the alkyne and aryl triflate components.
Q: Is the purification process scalable for industrial production?
A: Yes, the post-treatment involves simple filtration followed by standard silica gel column chromatography, which is a widely established and scalable unit operation in fine chemical manufacturing, avoiding complex crystallization or distillation steps that often limit throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acyl-2(5H)-Furanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into industrial reality. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4-acyl-2(5H)-furanone intermediate meets the exacting standards required for pharmaceutical applications.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic advantages of switching to this route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring your supply chain is optimized for efficiency, cost, and reliability.
