Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-Ones for Scalable Pharmaceutical Production
Advanced Palladium-Catalyzed Synthesis of 1,2,4-Triazole-3-Ones for Scalable Pharmaceutical Production
The pharmaceutical industry continuously seeks robust synthetic methodologies for constructing nitrogen-containing heterocycles, which serve as the structural backbone for a vast array of bioactive molecules. Patent CN112538054B discloses a groundbreaking preparation method for 1,2,4-triazole-3-one compounds, a privileged scaffold known for its diverse pharmacological properties ranging from antifungal and anti-inflammatory activities to antitumor and anticonvulsant effects. This innovation addresses critical bottlenecks in the supply chain of complex pharmaceutical intermediates by introducing a transition metal palladium-catalyzed carbonylation tandem cyclization reaction. By utilizing cheap and readily available chlorohydrazones and sodium azide as starting materials, this technology significantly enhances reaction efficiency and substrate compatibility. The ability to synthesize variously substituted derivatives through rational substrate design underscores the versatility of this approach, making it a highly attractive option for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios.
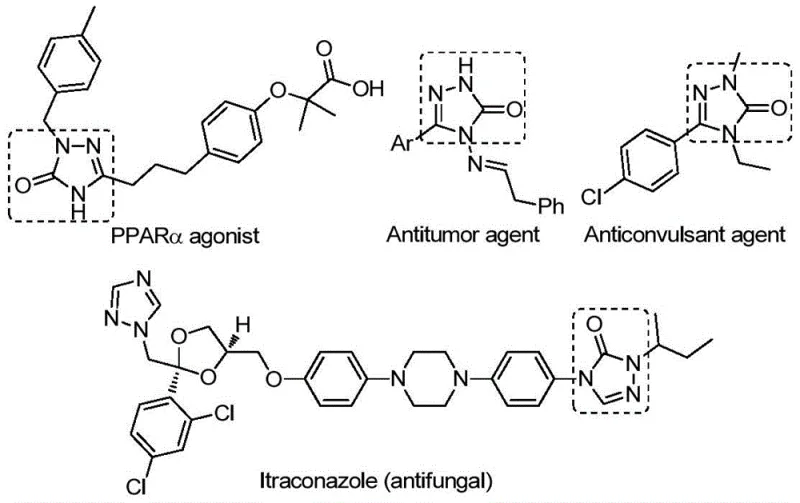
The strategic importance of this patent lies in its potential to streamline the manufacturing of high-value drug candidates. Traditional methods often suffer from limitations that hinder commercial viability, such as the need for hazardous reagents or multi-step sequences that erode overall yield. In contrast, the methodology described in CN112538054B offers a direct, one-pot pathway that aligns perfectly with the principles of green chemistry and process intensification. For R&D directors and procurement managers, understanding the mechanistic nuances and operational advantages of this palladium-catalyzed system is essential for evaluating its integration into existing production lines. This report provides a deep technical analysis of the process, highlighting how it delivers substantial cost savings and supply chain reliability for the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,4-triazole-3-one compounds has relied on several classical routes that present significant challenges for modern industrial application. Common traditional methods include the cyclization of benzoyl hydrazide with urea under basic conditions, the tandem cyclization of hydrazides with isocyanates, or the condensation of thioamides with hydrazines at elevated temperatures. These legacy processes are frequently plagued by harsh reaction conditions that require extreme pH levels or high thermal energy, leading to increased operational costs and safety risks. Furthermore, many of these routes necessitate the pre-activation of reaction substrates, adding unnecessary synthetic steps that reduce atom economy and generate excessive waste. The narrow substrate scope of these conventional methods often results in low yields when attempting to introduce diverse functional groups, limiting the chemical space available for drug discovery teams. Consequently, the reliance on these outdated techniques creates a bottleneck in the cost reduction in API manufacturing, as the cumulative inefficiencies translate directly into higher production expenses and longer lead times.
The Novel Approach
The novel approach detailed in patent CN112538054B represents a paradigm shift by employing a palladium-catalyzed carbonylation tandem cyclization strategy. This method utilizes chlorohydrazones and sodium azide as inexpensive and accessible starting materials, reacting them in the presence of a palladium catalyst, a specialized ligand, and a carbon monoxide surrogate known as TFBen. The reaction proceeds smoothly in an organic solvent at moderate temperatures, typically between 100°C and 120°C, eliminating the need for the extreme conditions associated with older protocols. A key advantage of this system is its exceptional functional group tolerance, allowing for the synthesis of 1,2,4-triazole-3-one compounds substituted with various alkyl, aryl, and halogen groups without compromising yield. The use of TFBen as a solid CO source is particularly advantageous for commercial scale-up of complex pharmaceutical intermediates, as it avoids the logistical and safety hazards of handling toxic carbon monoxide gas. This streamlined process not only simplifies the operational workflow but also ensures high reaction efficiency, making it an ideal solution for producing high-purity intermediates required in stringent pharmaceutical applications.

Mechanistic Insights into Palladium-Catalyzed Carbonylation Cyclization
The success of this synthetic route hinges on a sophisticated catalytic cycle driven by the palladium complex. The reaction is initiated by the oxidative addition of the palladium catalyst into the carbon-chlorine bond of the chlorohydrazone substrate, forming a reactive divalent palladium intermediate. Simultaneously, the carbon monoxide surrogate, TFBen, undergoes thermal decomposition to release carbon monoxide in situ. This generated CO then inserts into the carbon-palladium bond, creating an acyl-palladium species that serves as the precursor for ring closure. Subsequently, the acyl-palladium intermediate reacts with sodium azide to generate an acyl azide compound, which spontaneously undergoes a Curtius rearrangement to form an isocyanate intermediate. The final step involves an intramolecular nucleophilic addition where the hydrazine nitrogen attacks the electrophilic carbon of the isocyanate group, closing the five-membered triazole ring and releasing the final 1,2,4-triazole-3-one product. This intricate sequence demonstrates the power of transition metal catalysis in orchestrating multiple bond-forming events within a single reaction vessel, thereby maximizing efficiency and minimizing waste generation.
From an impurity control perspective, the choice of ligand and reaction conditions plays a pivotal role in ensuring product quality. The patent specifies the use of Xantphos as a ligand, which stabilizes the palladium center and facilitates the necessary coordination geometry for efficient CO insertion and reductive elimination. By maintaining precise control over the reaction temperature and time, typically 16 to 30 hours, the process minimizes the formation of side products such as unreacted starting materials or over-carbonylated byproducts. The use of aprotic solvents like 1,4-dioxane further enhances the reaction rate and selectivity, preventing unwanted hydrolysis or solvolysis of sensitive intermediates. For R&D teams focused on purity and impurity profiles, this mechanistic understanding allows for fine-tuning of the process parameters to achieve stringent quality specifications. The robustness of the catalytic system ensures consistent batch-to-batch reproducibility, which is a critical factor for regulatory compliance in the manufacturing of active pharmaceutical ingredients.
How to Synthesize 1,2,4-Triazole-3-One Efficiently
The practical implementation of this synthesis involves a straightforward protocol that balances reaction kinetics with operational simplicity. To achieve optimal results, the molar ratio of chlorohydrazone to sodium azide is typically maintained at 1:2.5, ensuring that the azide is present in excess to drive the reaction to completion without requiring difficult purification steps later. The catalyst loading is kept low, often around 2.5 mol% for the palladium source, which helps in reducing the residual metal content in the final product, a key concern for pharmaceutical applications. The reaction mixture is heated to 100°C in a sealed vessel to maintain the integrity of the volatile components while providing sufficient energy for the catalytic cycle to proceed. Detailed standardized synthesis steps see the guide below.
- Combine palladium catalyst, ligand, CO surrogate, chlorohydrazone, and sodium azide in an organic solvent.
- Heat the reaction mixture to 100-120°C and maintain for 16-30 hours to ensure complete conversion.
- Perform post-treatment including filtration and column chromatography to isolate the pure 1,2,4-triazole-3-one product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend beyond mere chemical efficiency. The primary value proposition lies in the significant simplification of the raw material supply chain. By utilizing chlorohydrazones and sodium azide, which are commodity chemicals available from multiple global vendors, manufacturers can mitigate the risk of supply disruptions often associated with specialized or proprietary reagents. This diversification of the supplier base enhances supply chain resilience, ensuring continuous production even in volatile market conditions. Furthermore, the elimination of pre-activation steps reduces the overall number of unit operations required, which directly translates to lower capital expenditure on equipment and reduced labor costs. The ability to source cheap and easy-to-obtain starting materials means that the cost of goods sold (COGS) can be drastically optimized, providing a competitive edge in pricing negotiations with downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound due to the elimination of expensive and hazardous reagents traditionally used in triazole synthesis. By replacing toxic isocyanates or unstable acyl chlorides with stable chlorohydrazones and a solid CO surrogate, the process removes the need for specialized containment systems and extensive safety protocols, thereby lowering overhead costs. Additionally, the high reaction efficiency and yield reported in the patent mean that less raw material is wasted, improving the overall material balance and reducing the cost per kilogram of the final product. The use of a palladium catalyst, while a precious metal, is employed at low loadings and can potentially be recovered and recycled, further enhancing the economic viability of the process. These factors collectively contribute to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures a stable and predictable supply of critical intermediates. Since the reaction tolerates a wide range of functional groups, manufacturers can produce a diverse library of derivatives from a common set of building blocks, allowing for rapid response to changing market demands. The scalability of the process, demonstrated from milligram to gram scales in the patent data, suggests a clear path to ton-scale production with minimal re-optimization. This scalability is crucial for meeting the rigorous delivery schedules of multinational pharmaceutical companies. Moreover, the use of standard organic solvents and common laboratory equipment means that the technology can be easily transferred between different manufacturing sites, reducing the lead time for high-purity intermediates and ensuring business continuity across the global supply network.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this method aligns well with modern sustainability goals. The replacement of gaseous carbon monoxide with a solid surrogate significantly reduces the risk of accidental releases and simplifies waste gas treatment systems. The reaction generates fewer byproducts compared to traditional condensation methods, resulting in a cleaner crude product that requires less intensive purification, thus reducing solvent consumption and waste generation. The simplified post-treatment process, involving filtration and standard chromatography, minimizes the environmental footprint of the manufacturing operation. For companies striving to meet strict environmental regulations and corporate social responsibility targets, adopting this greener synthetic route demonstrates a commitment to sustainable chemistry practices while maintaining high production standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects described in patent CN112538054B, providing clarity on the practical aspects of the technology. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this method into their existing manufacturing frameworks.
Q: What are the advantages of this Pd-catalyzed method over traditional synthesis?
A: This method avoids harsh conditions and pre-activation steps required by traditional routes like benzoyl hydrazide cyclization, offering better substrate compatibility and higher yields.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the use of stable solid reagents like TFBen instead of toxic CO gas and the robustness of the catalyst system make it highly amenable to commercial scale-up.
Q: What is the role of TFBen in the reaction?
A: TFBen acts as a safe and efficient carbon monoxide surrogate, releasing CO in situ under heating conditions to facilitate the carbonylation step without requiring high-pressure gas equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,4-Triazole-3-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN112538054B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1,2,4-triazole-3-one derivatives meets the highest industry standards. We are committed to leveraging our technical expertise to optimize this palladium-catalyzed process for maximum efficiency and cost-effectiveness, helping our partners accelerate their drug development timelines.
We invite you to collaborate with us to explore the full capabilities of this innovative synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating how this technology can enhance your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global pharmaceutical market through superior chemical manufacturing solutions.
