Advanced Organocatalytic Strategy for Scalable Chiral Spiro Indolone Manufacturing
Introduction to Breakthrough Chiral Synthesis Technology
The pharmaceutical industry continuously seeks robust methodologies for constructing complex chiral frameworks, particularly those found in bioactive natural products and drug candidates. Patent CN113980028A introduces a significant advancement in this domain by disclosing a preparation method for chiral spiro indolone compounds. This technology leverages an intramolecular carbonyl α-asymmetric oxidative coupling reaction, utilizing indolone derivatives bearing active hydrogen functional groups as substrates. The core innovation resides in the employment of a cinchona alkaloid-derived quaternary ammonium iodide salt as a chiral organocatalyst, paired with a peroxide oxidant. For a reliable pharmaceutical intermediate supplier, understanding such novel synthetic routes is paramount, as they offer pathways to high-value scaffolds with exceptional stereocontrol. This report analyzes the technical merits and commercial viability of this approach, highlighting its potential to streamline the manufacturing of critical API intermediates.
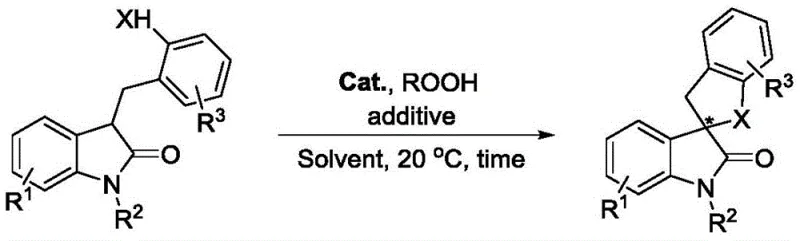
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral spiro indolone skeletons has relied heavily on transition metal catalysis or harsh Lewis acid-mediated transformations. Traditional strategies, such as 1,3-dipolar cycloadditions catalyzed by Lewis acids or nucleophilic phosphine-catalyzed cyclizations, often suffer from significant drawbacks. These conventional methods frequently necessitate stringent anhydrous conditions, expensive chiral ligands, and toxic heavy metals, which pose severe challenges for regulatory compliance in pharmaceutical manufacturing. Furthermore, the removal of trace metal residues from the final active pharmaceutical ingredient (API) requires additional purification steps, increasing both production costs and environmental waste. The sensitivity of these reactions to moisture and oxygen often limits their operational window, making large-scale implementation risky and economically inefficient for cost reduction in API manufacturing.
The Novel Approach
In stark contrast, the methodology described in CN113980028A offers a transformative solution through metal-free organocatalysis. By employing a chiral quaternary ammonium iodide salt derived from cinchona alkaloids, the process achieves high enantioselectivity under remarkably mild conditions. The reaction proceeds efficiently at 20°C in common organic solvents like n-propyl acetate, eliminating the need for cryogenic temperatures or high-pressure equipment. The use of peroxides, specifically cumene hydroperoxide, as the terminal oxidant ensures a clean oxidation profile. This novel approach not only simplifies the operational protocol but also enhances the safety profile of the synthesis. The broad substrate scope, accommodating various substituents on the indolone and benzene rings, underscores the versatility of this method for generating diverse libraries of high-purity chiral spiro indolones.
Mechanistic Insights into Organocatalytic Asymmetric Oxidative Coupling
The success of this transformation hinges on a sophisticated catalytic cycle involving in situ generation of hypervalent iodine species. The quinine-derived quaternary ammonium iodide salt catalyst reacts with the peroxide oxidant to generate a reactive hypoiodite or hypoiodate species. This active iodine species subsequently oxidizes the substrate to form a key N-I intermediate. The chirality is induced during the subsequent intramolecular SN2 reaction, where the rigid quaternary ammonium unit engages in tight ion pairing with the indolone enol anion. Additionally, hydrogen bonding interactions between the catalyst's secondary alcohol moiety and the substrate's sulfonyl group further reinforce stereochemical control. This dual activation mode ensures that the nucleophilic attack occurs with high facial selectivity, resulting in the formation of the chiral spirocyclic center with excellent enantiomeric ratios.
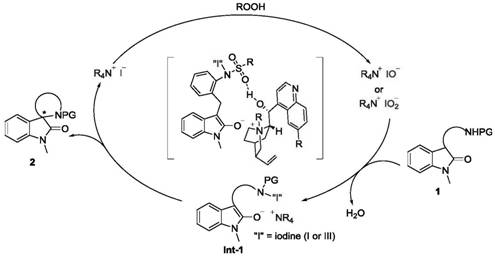
A critical aspect of this mechanism is the management of reaction byproducts, specifically water. As the oxidative coupling progresses, water molecules are generated, which can potentially hydrolyze the active iodine species or slow down the reaction equilibrium. The patent strategically incorporates molecular sieves (5Å MS) as an additive to address this issue. These molecular sieves act as efficient desiccants, continuously absorbing the produced water molecules. By removing water from the reaction medium, the equilibrium is driven towards the product side, significantly enhancing reaction efficiency and yield. Furthermore, the solid nature of the molecular sieves allows them to settle at the bottom of the reactor without interfering with the homogeneous liquid-phase catalysis, ensuring a smooth and scalable process suitable for commercial scale-up of complex heterocycles.
How to Synthesize Chiral Spiro Indolone Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction parameters to maximize stereochemical outcomes. The process begins with the dissolution of the indolone substrate in a dry organic solvent, followed by the addition of the chiral catalyst and molecular sieves. The oxidant is introduced last to initiate the catalytic cycle. Maintaining the temperature at 20°C is crucial for balancing reaction rate and selectivity. Detailed standard operating procedures regarding stoichiometry, mixing rates, and workup protocols are essential for reproducibility. For comprehensive technical guidance on executing this transformation, the detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining the indolone-derived substrate containing active hydrogen functional groups with a cinchona alkaloid-derived quaternary ammonium iodide salt catalyst in an organic solvent such as n-propyl acetate.
- Add molecular sieves as an additive to absorb water generated during the reaction, followed by the addition of a peroxide oxidant like cumene hydroperoxide.
- Stir the reaction mixture at a mild temperature of 20°C for approximately 12 hours, then quench, extract, and purify the resulting chiral spiro indolone product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this organocatalytic method presents compelling advantages over traditional metal-catalyzed routes. The elimination of precious transition metals removes the need for expensive scavenging resins and extensive metal testing, directly contributing to cost reduction in API manufacturing. The starting materials, including the indolone derivatives and the cinchona-based catalysts, are industrial commodities with wide availability, ensuring a stable supply chain. The mild reaction conditions reduce energy consumption and allow for the use of standard glass-lined or stainless steel reactors without specialized corrosion-resistant linings. These factors collectively enhance the economic feasibility of producing these high-value intermediates on a multi-ton scale.
- Cost Reduction in Manufacturing: The absence of heavy metal catalysts significantly lowers the raw material costs and simplifies the downstream purification process. Traditional methods often require costly ligands and rigorous metal removal steps to meet ppm-level specifications, whereas this organocatalytic route generates organic byproducts that are easier to separate. The use of inexpensive oxidants like cumene hydroperoxide and common solvents further drives down the variable cost of goods sold (COGS). Additionally, the high yields reported in the patent examples suggest minimal material waste, optimizing atom economy and reducing the overall cost burden associated with raw material procurement and waste disposal.
- Enhanced Supply Chain Reliability: The reliance on readily available industrial commodities mitigates the risk of supply disruptions often associated with specialized reagents. Cinchona alkaloids are abundant natural products, and their derivatives are commercially accessible from multiple global suppliers. The robustness of the reaction conditions, tolerating ambient temperature and standard atmospheric pressure, means that production is less susceptible to utility failures or equipment limitations. This reliability is crucial for reducing lead time for high-purity intermediates, ensuring that downstream drug development timelines are met without unexpected delays caused by raw material shortages or complex synthesis bottlenecks.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, characterized by simple operation and easy expansion of reaction scale. The use of molecular sieves as a heterogeneous additive simplifies the reaction control, as they can be easily filtered off during workup. From an environmental standpoint, the metal-free nature of the catalyst aligns with green chemistry principles, reducing the ecological footprint of the manufacturing process. The mild conditions minimize energy usage for heating or cooling, and the simplified workup reduces solvent consumption. These attributes facilitate regulatory approval and support sustainable manufacturing practices, which are increasingly demanded by global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral spiro indolone synthesis technology. These insights are derived directly from the experimental data and technical disclosures within patent CN113980028A. Understanding these nuances is vital for R&D teams evaluating the feasibility of this route for their specific pipeline projects. The answers provided reflect the current state of the art as described in the intellectual property documentation.
Q: What is the primary advantage of this organocatalytic method over traditional Lewis acid catalysis?
A: The primary advantage lies in the mild reaction conditions and the avoidance of transition metals. Unlike traditional Lewis acid methods which often require strict anhydrous conditions and complex metal removal steps, this method utilizes a cinchona alkaloid-derived catalyst at 20°C, significantly simplifying downstream processing and reducing heavy metal contamination risks in pharmaceutical intermediates.
Q: How does the addition of molecular sieves impact the reaction efficiency?
A: Molecular sieves play a critical dual role in this process. Firstly, they act as a desiccant to absorb water molecules produced as a byproduct of the oxidative coupling, thereby shifting the chemical equilibrium towards product formation according to Le Chatelier's principle. Secondly, they function as solid particles that settle without interfering with the liquid phase, ensuring consistent reaction kinetics and improved overall yields.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is highly amenable to scale-up. The patent explicitly highlights characteristics such as simple operation, mild conditions (20°C), and the use of inexpensive, readily available industrial commodities as starting materials. These factors collectively lower the barrier for commercial scale-up of complex heterocycles, making it a viable route for manufacturing high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro Indolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced synthetic methodologies like the one described in CN113980028A for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at adapting laboratory-scale organocatalytic processes to industrial manufacturing environments, ensuring that the high stereoselectivity and yields observed in the patent are maintained at scale. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of chiral spiro indolone meets the exacting standards required for clinical and commercial applications.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this innovative technology for their drug discovery programs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, evaluating how this metal-free route can optimize your budget. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a reliable supply chain capable of delivering high-quality intermediates with speed and precision, accelerating your path to market.
